Scaling High-Purity Urokinase Inhibitor Intermediates via Aromatic Sulfonic Acid Precipitation
The pharmaceutical industry's relentless pursuit of potent oncology therapeutics has placed significant emphasis on the urokinase plasminogen activator (UPA) system as a critical target for inhibiting tumor metastasis. Patent CN101039928A introduces a groundbreaking methodology for the preparation and purification of 3-hydroxyamidinophenylalanine derivatives, which serve as highly specific and selective urokinase inhibitors. Unlike traditional synthetic routes that struggle with low yields and difficult separations, this invention leverages the unique crystallization properties of aromatic sulfonic acid salts to achieve exceptional purity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding this technological shift is paramount. The process not only addresses the chronic issue of amide impurity contamination but also streamlines the manufacturing workflow, offering a robust pathway for producing oral bioavailable drug candidates. By integrating enzymatic resolution with a novel precipitation technique, the patent outlines a comprehensive strategy that balances stereochemical integrity with industrial feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amidinophenylalanine derivatives, such as the potent inhibitor WX-UK1, has been plagued by significant downstream processing challenges. Conventional methodologies often rely on the hydrolysis of protected intermediates, which inadvertently generates hydrolyzed TIPPS-OH as a persistent and undesired by-product. The separation of the target reaction product from these by-products typically necessitates time-consuming and laborious chromatographic techniques, which are inherently difficult to scale and costly to operate. Furthermore, prior art methods, including those described in PCT/EP03/08230, frequently yield amidoxime intermediate compounds that are not obtained in pure forms, often containing substantial proportions of amide impurities. These impurities can account for up to 30% of the crude mixture, severely compromising the quality of the final API and necessitating complex purification protocols that erode overall process efficiency. The reliance on chromatography not only increases the cost reduction in API manufacturing but also introduces bottlenecks that threaten supply chain continuity and batch-to-batch consistency.
The Novel Approach
The innovative process disclosed in the patent data fundamentally reimagines the purification landscape by replacing chromatographic separation with a selective precipitation strategy using aromatic sulfonic acids. This novel approach involves adding an aromatic sulfonic acid, most preferably naphthalene-1,5-disulfonic acid (Armstrong acid), to a solution of the optionally contaminated 3-hydroxyamidinophenylalanine derivative. This addition induces the formation of a specific salt that precipitates out of the solution, effectively leaving behind soluble impurities such as amides and unreacted starting materials. The precipitate can then be isolated through simple filtration, a unit operation that is far more amenable to large-scale industrial application than column chromatography. Following isolation, the free 3-hydroxyamidinophenylalanine derivative is recovered from the salt using a base stronger than the derivative itself, such as sodium bicarbonate. This method allows for the production of amidoximes with purity levels exceeding 95%, and optimally greater than 99%, thereby ensuring that the subsequent conversion to the final amidino-end product proceeds with significantly higher yields and minimal side reactions.
Mechanistic Insights into Aromatic Sulfonic Acid Salt Precipitation
The core mechanistic advantage of this technology lies in the differential solubility and crystallization kinetics of the sulfonic acid salts formed during the purification stage. When an aromatic sulfonic acid is introduced to the reaction mixture containing the crude amidoxime, it interacts with the basic amidino or hydroxyamidino functionality to form an ionic salt. The choice of the sulfonic acid is critical; disulfonic acids like Armstrong acid possess a strong tendency to crystallize, which drives the equilibrium towards the solid phase. This precipitation acts as a powerful thermodynamic sink, pulling the desired product out of the solution while leaving structurally similar impurities, particularly the troublesome amide by-products, dissolved in the mother liquor. The process is remarkably selective, capable of distinguishing between the target hydroxyamidino species and its amidino analogues or hydrolyzed variants. This selectivity is crucial for maintaining the stereochemical integrity of the L-configuration, as the mild conditions of salt formation and recovery prevent racemization, ensuring the production of enantiomerically pure intermediates essential for biological activity.
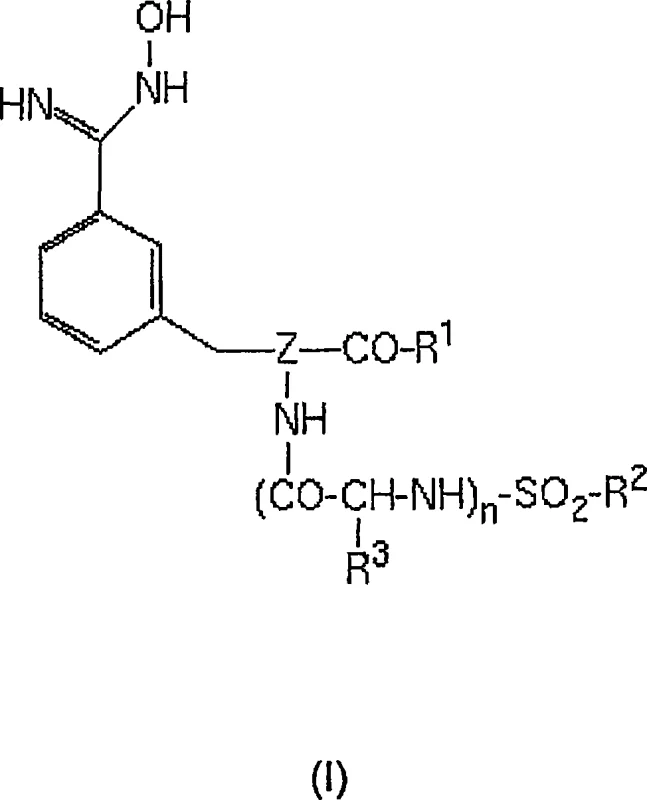
Furthermore, the mechanism extends to the broader synthetic sequence, where the purity of the intermediate directly influences the efficacy of the final reduction step. The conversion of the hydroxyamidino group to the amidino group, typically achieved via catalytic hydrogenation, is highly sensitive to catalyst poisoning by impurities. By ensuring the input material is of high purity through the sulfonic acid precipitation method, the lifespan and activity of the hydrogenation catalyst are preserved, leading to more consistent reaction outcomes. The structural versatility of the derivatives, as defined by variables R1, R2, and R3 in the general formula, allows this purification mechanism to be applied across a range of analogues. Whether the substituent is an ethoxycarbonyl-piperazine moiety or various aryl groups, the fundamental principle of salt formation remains effective. This robustness makes the technology a versatile platform for the commercial scale-up of complex peptide mimetics, providing a standardized purification module that can be adapted to various stages of drug development.
How to Synthesize 3-Hydroxyamidinophenylalanine Derivatives Efficiently
The synthesis of these high-value intermediates begins with the preparation of the chiral backbone, often utilizing enzymatic resolution to establish the correct stereochemistry early in the sequence. As detailed in the patent examples, the process initiates with the alkylation and decarboxylation to form the racemic cyano-phenylalanine derivative, followed by a critical enzymatic resolution step using Acyltransferase I. This biocatalytic step is pivotal for enriching the L-enantiomer, which is then protected, coupled with a piperazine derivative, and deprotected to reveal the free amine. Subsequent sulfonylation with TIPPS-chloride installs the necessary steric bulk, followed by the conversion of the nitrile group to the hydroxyamidino functionality using hydroxylamine hydrochloride. The detailed standardized synthesis steps see the guide below for specific reaction conditions and stoichiometry.
- Perform enzymatic racemate resolution of acetyl-cyano-phenylalanine using Acyltransferase I to isolate the L-enantiomer.
- Protect the amino group with Boc, couple with ethoxycarbonylpiperazine, and deprotect to form the free amine intermediate.
- React with TIPPS-chloride to form the sulfonamide, convert the cyano group to hydroxyamidino, and purify using Armstrong acid precipitation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chromatographic purification to salt precipitation represents a transformative opportunity for cost optimization and risk mitigation. The elimination of large-scale chromatography columns removes a significant capital expenditure and operational expense, as it reduces the need for expensive silica gels, solvents, and specialized pumping systems. This simplification of the downstream processing train directly translates to substantial cost savings in manufacturing, allowing for more competitive pricing of the final active pharmaceutical ingredients. Moreover, the reliance on common industrial reagents like aromatic sulfonic acids and standard filtration equipment enhances the resilience of the supply chain, reducing dependency on niche consumables that might face availability constraints. The ability to produce high-purity intermediates with fewer processing steps also shortens the overall production cycle time, enabling faster response to market demands and reducing the inventory holding costs associated with long lead times.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the complete avoidance of preparative chromatography, which is notoriously expensive and inefficient at scale. By utilizing precipitation and filtration, the process drastically reduces solvent consumption and waste generation, leading to a leaner and more environmentally sustainable manufacturing footprint. The high chemical yields reported in the patent examples, combined with the high purity of the isolated product, minimize the loss of valuable chiral intermediates, ensuring that the cost of goods sold is optimized. This efficiency is particularly critical for complex molecules where the cost of raw materials and chiral building blocks constitutes a major portion of the total production cost.
- Enhanced Supply Chain Reliability: The robustness of the precipitation method ensures consistent batch quality, which is essential for maintaining regulatory compliance and avoiding costly batch rejections. The use of stable and readily available aromatic sulfonic acids, such as those commonly used in the dye industry, guarantees a secure supply of key reagents. This stability allows for better production planning and forecasting, as the process is less susceptible to the variabilities often encountered in chromatographic separations. Consequently, partners can rely on a steady flow of high-purity urokinase inhibitor precursors, supporting uninterrupted clinical trial material production and commercial launch schedules.
- Scalability and Environmental Compliance: Scaling a chromatographic process from grams to tons is a formidable engineering challenge that often requires disproportionate increases in facility size and complexity. In contrast, precipitation and filtration are unit operations that scale linearly and predictably, facilitating the commercial scale-up of complex pharmaceutical intermediates from pilot plant to full production. Additionally, the reduction in solvent usage and the potential for solvent recycling in the precipitation steps align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This alignment not only mitigates regulatory risk but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity on how this method compares to existing standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates. The answers reflect the practical realities of scaling this chemistry and the tangible benefits it offers over traditional approaches.
Q: What is the primary advantage of using Armstrong acid in this purification process?
A: Armstrong acid (naphthalene-1,5-disulfonic acid) facilitates the selective precipitation of the 3-hydroxyamidinophenylalanine derivative as a salt, allowing for the efficient removal of amide impurities and unreacted starting materials without the need for laborious chromatography.
Q: How does this method improve the purity compared to conventional synthesis?
A: Conventional methods often result in products contaminated with up to 30% amide impurities requiring chromatographic separation. This patented precipitation method achieves purity levels greater than 95%, and most preferably greater than 99%, through simple filtration and recrystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the replacement of chromatographic purification with salt precipitation and filtration significantly enhances scalability. It reduces equipment complexity and processing time, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxyamidinophenylalanine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the successful development of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 3-hydroxyamidinophenylalanine derivatives meets the exacting standards required for urokinase inhibitor synthesis. Our commitment to technical excellence means we can navigate the complexities of chiral synthesis and salt formation with precision, delivering materials that accelerate your drug development timeline.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production expenses. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate the tangible value of our supply chain solutions. Let us collaborate to bring life-saving urokinase inhibitors to the market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
