Advanced Synthesis of Bis-Thio-Hydrazide Amines for High-Efficacy Paclitaxel Adjuvants
The landscape of oncology treatment is constantly evolving, with a significant focus shifting towards combination therapies that maximize efficacy while minimizing the severe side effects associated with potent chemotherapeutic agents like paclitaxel. Patent CN1553893A presents a groundbreaking chemical methodology for the synthesis of specific thiohydrazide compounds, designated as formula (II), which serve as critical precursors to bis[thio-hydrazide amine] structures. These novel compounds have been identified as powerful enhancers of paclitaxel's anticancer activity, offering a potential pathway to reduce the dosage of the primary drug required for therapeutic effect. The core innovation lies in the strategic application of thionylation agents to convert hydrazide starting materials into their sulfur-containing analogues, followed by a precise amidation coupling process. This technical disclosure provides a robust framework for developing high-purity pharmaceutical intermediates that address the urgent clinical need for synergistic cancer treatments with improved safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to enhancing chemotherapy often involve the co-administration of distinct drug entities, which can lead to unpredictable pharmacokinetic interactions and exacerbated toxicity profiles such as neutropenia, neuropathy, and mucositis. In the context of synthesizing adjuvant molecules, conventional amide bond formation strategies frequently struggle with the incorporation of sulfur moieties, which are essential for the specific biological activity observed in these paclitaxel enhancers. Standard peptide coupling conditions may not be compatible with sensitive hydrazide functionalities or may fail to achieve the necessary thione substitution without degrading the molecular scaffold. Furthermore, older synthetic routes often rely on harsh conditions that generate significant impurity profiles, necessitating complex and costly purification steps that hinder commercial viability. The inability to efficiently introduce the thio-carbonyl group onto the hydrazide nitrogen backbone has historically limited the exploration of this chemical space for oncology applications.
The Novel Approach
The methodology outlined in the patent data introduces a sophisticated two-stage synthetic strategy that overcomes these historical barriers. By first isolating the thionylation step, the process ensures high conversion of the hydrazide starting compound (Formula I) into the reactive thiohydrazide intermediate (Formula II) using specialized reagents like Lawesson's reagent or phosphorus pentasulfide. 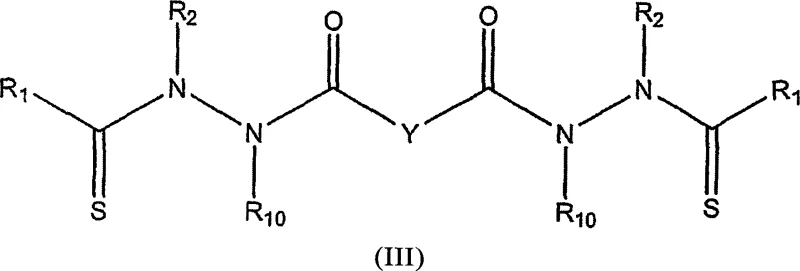 This intermediate is then coupled with dicarboxylic acid derivatives, such as malonyl chloride or activated esters, to form the final bis[thio-hydrazide amine] structure (Formula III). This modular approach allows for significant structural diversity by varying the R groups on the hydrazide and the linker Y, enabling the fine-tuning of physicochemical properties. The use of mild activation strategies, including in situ generation of activated esters via carbodiimides, ensures that the sensitive thioamide bonds remain intact during the coupling phase, resulting in superior product purity and yield compared to direct condensation methods.
This intermediate is then coupled with dicarboxylic acid derivatives, such as malonyl chloride or activated esters, to form the final bis[thio-hydrazide amine] structure (Formula III). This modular approach allows for significant structural diversity by varying the R groups on the hydrazide and the linker Y, enabling the fine-tuning of physicochemical properties. The use of mild activation strategies, including in situ generation of activated esters via carbodiimides, ensures that the sensitive thioamide bonds remain intact during the coupling phase, resulting in superior product purity and yield compared to direct condensation methods.
Mechanistic Insights into Thionylation and Amidation Coupling
The heart of this synthesis lies in the mechanistic precision of the thionylation reaction. When a hydrazide is treated with a thionylating agent such as Lawesson's reagent in an inert solvent like toluene or benzene, the oxygen atom of the carbonyl group is effectively replaced by a sulfur atom. This transformation typically proceeds under reflux conditions, ranging from 75°C to 125°C, ensuring complete conversion while minimizing side reactions. The patent highlights that stoichiometry is critical, with a slight excess of the thionylating agent (up to 1.1 equivalents) often preferred to drive the equilibrium forward without introducing excessive impurities that are difficult to remove. The resulting thiohydrazide exhibits enhanced nucleophilicity at the nitrogen center, which is pivotal for the subsequent coupling step. This specific electronic modification is what grants the final molecule its unique ability to interact synergistically with microtubule-stabilizing agents like paclitaxel.
Following thionylation, the amidation mechanism employs classic carboxylic acid activation principles adapted for this sensitive substrate. The process involves converting a dicarboxylic acid into a more reactive species, such as an acid chloride using thionyl chloride or an activated ester using DCC and HOBt. 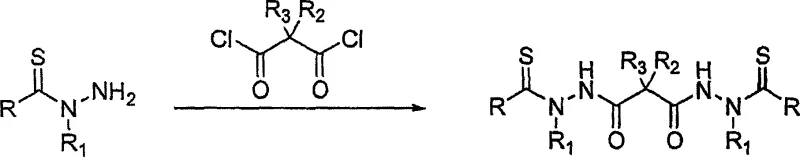 In the acid chloride route, the reaction is typically conducted at low temperatures (0°C) to control exothermicity and prevent degradation, followed by warming to room temperature to complete the coupling. Alternatively, the carbodiimide-mediated pathway allows for milder conditions, often proceeding at room temperature in polar aprotic solvents like DMF. The presence of additives like HOBt is crucial here, as they suppress the formation of N-acylurea byproducts and facilitate the attack of the thiohydrazide nitrogen on the activated carbonyl carbon. This dual-pathway flexibility allows manufacturers to select the most cost-effective and scalable route based on the specific substituents involved, ensuring robust impurity control throughout the synthesis.
In the acid chloride route, the reaction is typically conducted at low temperatures (0°C) to control exothermicity and prevent degradation, followed by warming to room temperature to complete the coupling. Alternatively, the carbodiimide-mediated pathway allows for milder conditions, often proceeding at room temperature in polar aprotic solvents like DMF. The presence of additives like HOBt is crucial here, as they suppress the formation of N-acylurea byproducts and facilitate the attack of the thiohydrazide nitrogen on the activated carbonyl carbon. This dual-pathway flexibility allows manufacturers to select the most cost-effective and scalable route based on the specific substituents involved, ensuring robust impurity control throughout the synthesis.
How to Synthesize Bis-Thio-Hydrazide Amines Efficiently
Executing this synthesis requires careful attention to solvent dryness and reagent quality to maximize yields, which the patent reports can reach up to 96% for intermediate steps and around 80% for final coupling reactions. The process begins with the protection of hydrazine groups if necessary, followed by the critical thionylation step in a refluxing aromatic solvent. Once the thio-intermediate is isolated and purified, typically via silica gel chromatography or recrystallization, it is ready for the final assembly. The detailed standardized synthesis steps below outline the precise conditions for reagent addition, temperature control, and workup procedures required to achieve pharmaceutical-grade quality consistently.
- Prepare the thiohydrazide intermediate by reacting a hydrazide starting compound with a thionylating agent such as Lawesson's reagent or phosphorus pentasulfide in an inert solvent.
- Activate the dicarboxylic acid linker using a coupling agent like DCC or convert it to an acid chloride using thionyl chloride.
- Couple the activated linker with two equivalents of the thiohydrazide intermediate to form the final bis[thio-hydrazide amine] structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere chemical elegance. The reliance on commercially available starting materials, such as substituted benzoic acids, hydrazines, and common dicarboxylic acids like malonic acid, ensures a stable and resilient supply chain. Unlike processes requiring exotic catalysts or rare earth metals, this methodology utilizes reagents that are produced at a global scale, mitigating the risk of supply disruptions. Furthermore, the elimination of transition metal catalysts in the key coupling steps removes the need for expensive and time-consuming heavy metal scavenging processes, which significantly streamlines downstream processing and reduces overall manufacturing costs.
- Cost Reduction in Manufacturing: The synthetic pathway described allows for significant cost optimization by utilizing high-yield reactions that minimize raw material waste. The ability to perform the thionylation and coupling steps in common industrial solvents like dichloromethane, toluene, and DMF means that solvent recovery and recycling systems already present in most fine chemical facilities can be leveraged immediately. Additionally, the avoidance of cryogenic conditions for the majority of the process reduces energy consumption associated with cooling, while the robust nature of the intermediates allows for simpler isolation techniques such as filtration and recrystallization rather than complex distillation or preparative HPLC.
- Enhanced Supply Chain Reliability: The modularity of the synthesis, where various R groups can be introduced without altering the core reaction conditions, provides immense flexibility in sourcing. If a specific substituted hydrazide becomes unavailable, the process can often accommodate alternative analogues with minimal re-validation, ensuring continuity of supply for the final active enhancer. The use of well-established coupling reagents like DCC and EDC, which have mature global supply chains, further de-risks the procurement process. This reliability is critical for maintaining the production schedules of downstream pharmaceutical formulations where delays can have significant clinical and financial impacts.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction parameters are highly favorable for commercial production, with exotherms that are manageable through standard jacketed reactor cooling systems. The process generates byproducts that are generally easier to handle and dispose of compared to those from heavy metal-catalyzed cross-couplings. For instance, the urea byproducts from DCC coupling can often be filtered off directly, simplifying the waste stream. This aligns well with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities and lowering the overall environmental footprint of the manufacturing operation, which is a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments and risk analysis. Understanding these nuances is essential for R&D teams planning pilot runs and procurement officers evaluating vendor capabilities.
Q: What is the primary advantage of the thionylation method described in CN1553893A?
A: The method allows for the efficient conversion of hydrazides to thiohydrazides using reagents like Lawesson's reagent, creating a unique chemical scaffold that enhances the anticancer activity of paclitaxel without increasing systemic toxicity.
Q: Which coupling agents are suitable for the amidation step in this synthesis?
A: The patent specifies several effective coupling agents including dicyclohexylcarbodiimide (DCC), 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC), and carbonyldiimidazole (CDI), often used with additives like HOBt to improve yield and purity.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the reaction conditions utilize common industrial solvents such as toluene, dichloromethane, and DMF, and operate at temperatures ranging from room temperature to reflux, making the process highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Thio-Hydrazide Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these bis[thio-hydrazide amine] compounds in the next generation of oncology therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reagent handling requirements of this chemistry, while our rigorous QC labs enforce stringent purity specifications to meet the demanding standards of the pharmaceutical industry. We understand that consistency is key in adjuvant therapy, and our process controls are designed to deliver batch-to-batch reproducibility that you can trust.
We invite you to engage with our technical team to explore how this synthesis can be optimized for your specific portfolio needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to verify specific COA data against your internal standards, our experts are ready to assist. Please contact our technical procurement team today to request route feasibility assessments and discuss how we can support your long-term supply goals for these critical anticancer enhancers.
