Optimizing Quinapril Hydrochloride Production: A Novel Tert-Butyl Ester Route for Commercial Scale
The pharmaceutical landscape for Angiotensin-Converting Enzyme (ACE) inhibitors continues to evolve, driven by the relentless demand for higher purity Active Pharmaceutical Ingredients (APIs) and more efficient manufacturing processes. Patent CN1747935A introduces a transformative methodology for the preparation of Quinapril Hydrochloride, addressing critical bottlenecks inherent in legacy synthetic routes. This innovation shifts the paradigm from traditional benzyl ester protection strategies, which are plagued by significant yield losses and safety hazards, to a robust tert-butyl ester approach. By leveraging N-carboxyanhydride chemistry coupled with mild acid-mediated deprotection, this process not only enhances the chemical integrity of the final product but also streamlines the supply chain for this vital cardiovascular medication. For global procurement teams and R&D directors, understanding this technological leap is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality at scale.
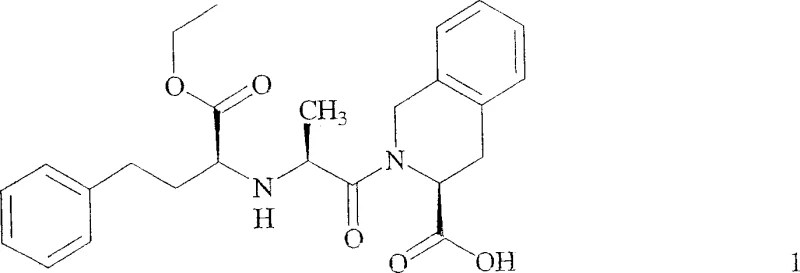
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Quinapril has relied heavily on routes involving (3S)-1,2,3,4-tetrahydro-isoquinoline-3-carboxylic acid benzyl esters. While these starting materials are readily accessible, the downstream processing presents severe challenges that impact both cost and quality. The primary defect lies in the necessity of a Pd/C catalyzed hydrogenolysis step to remove the benzyl protecting group. This step is not only operationally hazardous due to the use of hydrogen gas but also thermally stressful. Literature and patent data indicate that temperatures exceeding 45°C during the distillation of hydrogenolysis solvents promote the intramolecular cyclization of the peptide backbone. This side reaction leads to the formation of diketopiperazine impurities, resulting in catastrophic yield losses ranging from 20% to 40%. Furthermore, the removal of palladium residues requires additional purification stages, adding complexity and cost to the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology disclosed in CN1747935A circumvents these pitfalls by employing a tert-butyl protected tetrahydroisoquinoline derivative. This strategic modification allows for the removal of the protecting group using anhydrous hydrogen chloride in aprotic or protic solvents at ambient or slightly elevated temperatures, completely eliminating the need for hydrogenation. The reaction proceeds through the coupling of an N-carboxyanhydride with the tert-butyl ester, followed by a one-pot deprotection and salt formation. This route effectively suppresses the formation of diketopiperazine by avoiding the high-thermal stress conditions associated with solvent stripping after hydrogenolysis. Consequently, the process achieves a substantial increase in throughput, reported to be approximately three-fold when utilizing existing infrastructure, thereby offering a compelling value proposition for commercial scale-up of complex pharmaceutical intermediates.
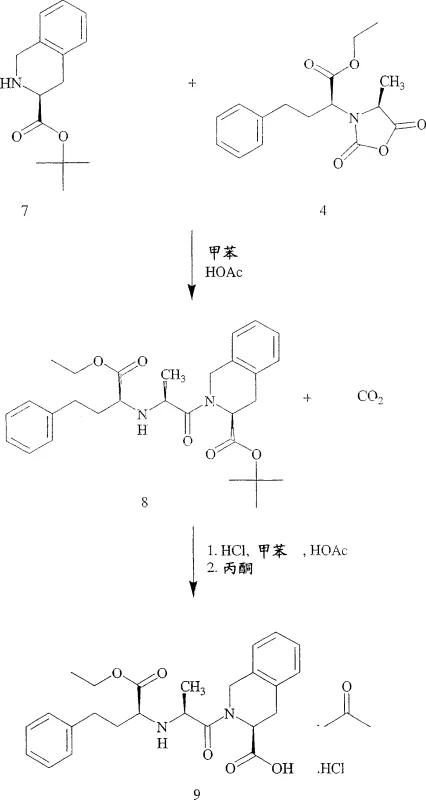
Mechanistic Insights into Acid-Catalyzed Deprotection and Coupling
The core of this synthetic advancement lies in the reactivity of the N-carboxyanhydride (NCA) intermediate, specifically (2S,4S)-2-(4-methyl-2,5-dioxo-oxazolidin-3-yl)-4-phenyl-butyric acid ethyl ester. In the presence of a catalytic amount of acid, such as acetic acid, the NCA undergoes nucleophilic attack by the secondary amine of the tetrahydroisoquinoline tert-butyl ester. This coupling reaction is highly efficient, typically reaching ≥99.9% conversion within 30 minutes at temperatures between 30°C and 35°C. The evolution of carbon dioxide during this step serves as a driving force, pushing the equilibrium towards the desired quinapril tert-butyl ester. Unlike traditional peptide coupling agents like DCC which generate difficult-to-remove urea byproducts, the NCA route generates gaseous CO2, simplifying the workup and enhancing the purity profile of the crude intermediate.
Following the coupling, the deprotection mechanism utilizes the lability of the tert-butyl carbamate under acidic conditions. By introducing anhydrous HCl into the reaction mixture, the tert-butyl group is cleaved to form isobutylene gas and the free amine, which immediately forms the hydrochloride salt. This transformation occurs rapidly at temperatures below 20°C, a critical parameter that preserves the stereochemical integrity of the chiral centers and prevents epimerization. The immediate precipitation of the product as an acetone solvate upon cooling further locks the molecule in its desired conformation, preventing the rearrangement into cyclic impurities. This precise control over reaction kinetics and thermodynamics is what enables the production of high-purity pharmaceutical intermediates with minimal downstream purification requirements.
How to Synthesize Quinapril Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from bench-scale optimization to industrial production. The process begins with the preparation of the tetrahydroisoquinoline tert-butyl ester free base from its p-toluenesulfonate salt, followed by the crucial coupling with the N-carboxyanhydride in toluene. The subsequent deprotection with HCl and isolation via acetone precipitation are designed to maximize recovery while minimizing solvent usage. Detailed standard operating procedures regarding stoichiometry, agitation rates, and temperature ramps are critical for replicating the reported 90% overall yield. For a comprehensive breakdown of the specific experimental conditions and safety protocols required for implementation, please refer to the standardized guide below.
- Couple (2S,4S)-2-(4-methyl-2,5-dioxo-oxazolidin-3-yl)-4-phenyl-butyric acid ethyl ester with (3S)-1,2,3,4-tetrahydro-isoquinoline-3-carboxylic acid tert-butyl ester in toluene with catalytic acetic acid.
- Treat the resulting quinapril tert-butyl ester with anhydrous hydrogen chloride in a mixed solvent system to effect deprotection and salt formation simultaneously.
- Precipitate the product as an acetone solvate, recrystallize from acetonitrile, and dry under vacuum to obtain the amorphous hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates directly into tangible operational efficiencies and risk mitigation. The elimination of the hydrogenolysis step removes a significant bottleneck in the production schedule, allowing for faster batch turnover and reduced dependency on specialized hydrogenation equipment. This simplification of the process flow not only accelerates time-to-market but also drastically reduces the capital expenditure required for facility upgrades. Furthermore, by avoiding the use of palladium catalysts, manufacturers can sidestep the stringent regulatory requirements and costs associated with heavy metal testing and clearance, ensuring a smoother path through quality control audits.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant improvement in chemical yield. By mitigating the formation of diketopiperazine impurities, the process recovers material that would otherwise be lost in conventional routes. This increase in mass efficiency means that less raw material is required to produce the same amount of final API, directly lowering the Cost of Goods Sold (COGS). Additionally, the removal of the hydrogenation step eliminates the costs associated with purchasing and disposing of precious metal catalysts, as well as the energy costs related to high-pressure hydrogen operations. The simplified solvent system, primarily utilizing toluene and acetone, further contributes to cost reduction in pharmaceutical intermediates manufacturing by using widely available and recyclable solvents.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses involving hazardous reagents. By replacing hydrogen gas and pyrophoric Pd/C with stable liquid reagents like anhydrous HCl and acetic acid, the process becomes inherently safer and less prone to shutdowns due to safety incidents. The starting materials, particularly the N-carboxyanhydride and the tert-butyl ester, are synthesized from commodity chemicals, reducing the risk of raw material shortages. This robustness ensures that a reliable pharmaceutical intermediates supplier can maintain consistent delivery schedules even during periods of market volatility, providing peace of mind to downstream drug manufacturers.
- Scalability and Environmental Compliance: Scaling peptide-based syntheses is notoriously difficult due to exothermic coupling reactions and purification challenges. This novel route addresses scalability by utilizing a coupling mechanism that evolves gas (CO2), which naturally agitates the mixture and aids in heat dissipation, making it easier to manage in large-scale reactors. From an environmental perspective, the process generates fewer hazardous waste streams. The absence of heavy metal waste and the reduction in solvent volume due to higher yields contribute to a smaller environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Quinapril Hydrochloride using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is crucial for technical teams evaluating potential technology transfers or sourcing strategies.
Q: How does the novel tert-butyl ester route improve yield compared to conventional benzyl ester methods?
A: Conventional methods utilizing benzyl esters often suffer from 20-40% yield loss due to intramolecular cyclization forming diketopiperazines, particularly during the hydrogenolysis and solvent removal steps. The novel tert-butyl ester route avoids hydrogenolysis entirely, using mild acid deprotection instead, which substantially increases yield by approximately 25% and minimizes thermal degradation.
Q: What are the safety advantages of eliminating the hydrogenolysis step in Quinapril production?
A: Removing the Pd/C catalyzed hydrogenolysis step eliminates the need for handling high-pressure hydrogen gas and pyrophoric catalysts. This significantly reduces operational hazards, simplifies the equipment requirements by removing the need for hydrogenation reactors, and lowers the overall cost of safety compliance and waste disposal associated with heavy metal catalyst removal.
Q: Why is the amorphous form of Quinapril Hydrochloride preferred in this synthesis?
A: The process specifically targets the amorphous form through a controlled recrystallization from acetonitrile followed by vacuum drying. This form offers superior solubility characteristics essential for bioavailability in the final drug product, and the described method achieves high purity (≥99.5% conversion) without the extensive purification steps often required for crystalline polymorphs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinapril Hydrochloride Supplier
The transition to more efficient synthetic routes like the one described in CN1747935A requires a manufacturing partner with deep technical expertise and proven scale-up capabilities. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this tert-butyl ester route, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to process optimization, we guarantee the supply of high-quality intermediates that adhere to global regulatory standards.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this superior technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and commercial reliability.
