Advanced Synthesis of Ropivacaine Hydrochloride: A Green Route for Commercial Scale-up
The global demand for high-quality local anesthetics continues to drive innovation in pharmaceutical manufacturing, particularly for long-acting agents like ropivacaine. A significant technological breakthrough in this sector is detailed in patent CN113735760A, which discloses a novel preparation method for ropivacaine hydrochloride. This patent addresses critical bottlenecks in existing industrial processes by introducing a streamlined synthetic route that prioritizes stereochemical integrity and operational safety. Unlike conventional methods that rely on cumbersome resolution steps or hazardous reagents, this new approach leverages a strategic reductive amination of single-chiral (S)-piperidine-2-carboxylic acid. For R&D directors and procurement managers alike, this represents a pivotal shift towards more sustainable and economically viable production of this essential API intermediate. The method promises not only superior purity levels exceeding 99.99% but also a drastic simplification of the workflow, making it an ideal candidate for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of ropivacaine has been plagued by inefficiencies and safety concerns inherent to older chemical pathways. Several prior art routes, such as those utilizing triphosgene for acid chloride formation, present severe hazards due to the toxicity and instability of the reagents involved. Triphosgene, while effective, requires rigorous safety protocols for storage and post-treatment, increasing the operational burden and risk profile of the manufacturing facility. Furthermore, many traditional strategies begin with racemic starting materials, necessitating a resolution step to isolate the pharmacologically active (S)-enantiomer. This resolution process is inherently wasteful, theoretically discarding up to 50% of the starting material unless dynamic kinetic resolution is employed, which adds further complexity and cost. Additionally, alternative routes involving late-stage alkylation often suffer from the formation of quaternary ammonium salt impurities, which are difficult to remove and can compromise the safety profile of the final drug product. These cumulative issues result in higher production costs, longer lead times, and a larger environmental footprint.
The Novel Approach
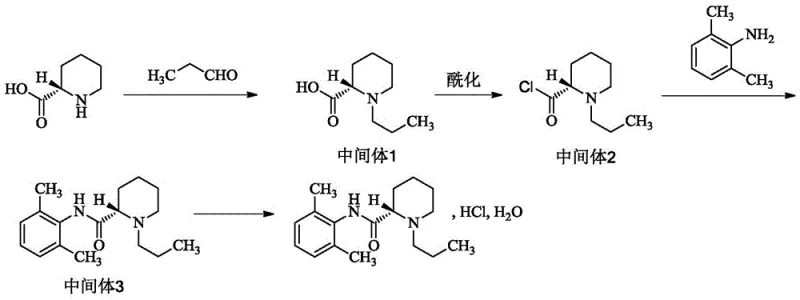
The methodology outlined in patent CN113735760A offers a transformative solution by reordering the synthetic sequence to maximize efficiency and minimize risk. The core innovation involves initiating the synthesis with single-chiral (S)-piperidine-2-carboxylic acid and immediately subjecting it to a reductive amination reaction with propionaldehyde. This early introduction of the N-propyl group eliminates the need for amino protection and deprotection cycles that characterize other routes. By establishing the chiral center and the N-substituent at the very beginning, the process ensures that all subsequent steps proceed with high stereochemical fidelity. The use of mild reducing agents like sodium cyanoborohydride or sodium borohydride acetate allows the reaction to proceed at moderate temperatures between 25°C and 55°C, avoiding the harsh conditions that often degrade sensitive intermediates. This strategic reordering not only simplifies the purification workflow but also inherently suppresses the formation of problematic byproducts, delivering a crude product of exceptional quality that requires minimal downstream processing.
Mechanistic Insights into Reductive Amination and Amidation
The success of this novel pathway hinges on the precise control of chemoselectivity during the initial reductive amination step. In this mechanism, the secondary amine of the piperidine ring reacts with propionaldehyde to form an iminium ion intermediate, which is subsequently reduced by the hydride source to yield the tertiary amine, (S)-1-n-propylpiperidine-2-carboxylic acid. The choice of solvent, typically dichloromethane or similar aprotic solvents, is critical to maintaining the solubility of the zwitterionic starting material while facilitating the iminium formation. Crucially, the carboxylic acid moiety remains intact and unreactive under these specific reduction conditions, preserving the functionality needed for the subsequent acylation step. This orthogonality is key; it allows the nitrogen to be functionalized without affecting the carbon skeleton or the stereocenter at the 2-position. Following this, the conversion to the acid chloride using thionyl chloride is catalyzed by trace amounts of DMF, generating the highly reactive acyl chloride intermediate in situ. This activation is performed under reflux at a relatively low temperature of 40°C, ensuring that the thermal stress on the chiral center is minimized, thereby preventing racemization.
Impurity control is another cornerstone of this mechanistic design. In traditional routes where alkylation is performed last, the nucleophilic attack of the amide nitrogen on the alkyl halide can lead to over-alkylation, producing quaternary ammonium salts that are structurally similar to the target molecule and difficult to separate. By reversing the order—alkylating the piperidine nitrogen first—the nucleophilicity of that nitrogen is significantly reduced once the amide bond is formed in the final condensation step with 2,6-dimethylaniline. The resulting amide nitrogen is far less nucleophilic due to resonance delocalization, effectively shutting down the pathway for quaternary salt formation. Furthermore, the final salification step with concentrated hydrochloric acid is conducted in isopropanol, which promotes the crystallization of the hydrochloride salt while keeping organic impurities in solution. The final recrystallization from an isopropanol-ethanol mixture acts as a powerful polishing step, leveraging the differential solubility of the target compound versus any remaining trace impurities to achieve the reported purity of greater than 99.99%.
How to Synthesize Ropivacaine Hydrochloride Efficiently
The synthesis of ropivacaine hydrochloride via this patented route is designed for scalability and robustness, making it highly suitable for industrial implementation. The process begins with the reductive amination of the chiral starting material, followed by activation of the carboxylic acid and coupling with the aniline derivative. Each step has been optimized to balance reaction rate with product stability, ensuring high yields without compromising safety. The detailed standardized operating procedures, including specific molar ratios, temperature profiles, and workup protocols, are essential for reproducing the high purity and yield reported in the patent data. Manufacturers looking to adopt this technology should pay close attention to the stoichiometry of the reducing agent and the control of temperature during the acid chloride formation to prevent side reactions.
- Perform reductive amination of (S)-piperidine-2-carboxylic acid with propionaldehyde using sodium cyanoborohydride to form the N-propyl intermediate.
- Convert the carboxylic acid intermediate to an acid chloride using thionyl chloride under mild reflux conditions.
- Condense the acid chloride with 2,6-dimethylaniline in the presence of a base to form the ropivacaine free base.
- Salify the free base with hydrochloric acid and purify via recrystallization in isopropanol and ethanol to achieve >99.99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical elegance. The elimination of the resolution step is perhaps the most significant economic driver, as it effectively doubles the theoretical yield from the chiral starting material compared to racemic routes. This efficiency translates directly into a lower cost of goods sold (COGS) and reduced raw material consumption. Moreover, the avoidance of hazardous reagents like triphosgene simplifies the regulatory compliance landscape, reducing the costs associated with specialized waste disposal and safety monitoring. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. These factors combined create a supply chain that is not only more cost-effective but also more resilient and easier to scale.
- Cost Reduction in Manufacturing: The primary cost advantage stems from the atom economy and step efficiency of the new route. By bypassing the resolution step, manufacturers avoid the substantial loss of material inherent in separating enantiomers, which traditionally caps the maximum yield at 50% unless recycling loops are implemented. Additionally, the reagents used, such as propionaldehyde and thionyl chloride, are commodity chemicals with stable pricing and widespread availability, unlike specialized chiral catalysts or dangerous reagents that command premium prices. The simplified purification process, which relies on standard recrystallization rather than complex chromatography or distillation, further reduces operational expenditures related to solvent usage and equipment time.
- Enhanced Supply Chain Reliability: Supply continuity is critical for API production, and this method enhances reliability by reducing dependency on scarce or regulated materials. The starting material, (S)-piperidine-2-carboxylic acid, is commercially available from multiple sources, mitigating the risk of single-supplier bottlenecks. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in utility supplies or environmental conditions, leading to more consistent batch-to-batch performance. This predictability allows for tighter production scheduling and shorter lead times, enabling suppliers to respond more agilely to market demand fluctuations without the need for excessive safety stock.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the mild nature of this synthesis facilitates a smoother transition from pilot plant to commercial production. The absence of high-pressure or high-temperature requirements reduces the engineering complexity of the reactors needed, allowing for the use of standard glass-lined or stainless steel equipment. From an environmental perspective, the process generates less hazardous waste, particularly by avoiding phosgene-related byproducts. This simplifies wastewater treatment and reduces the liability associated with hazardous waste disposal, ensuring that the manufacturing facility remains in good standing with increasingly stringent environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does this new synthesis route improve upon traditional methods involving resolution?
A: Traditional methods often start with racemic mixtures requiring costly and yield-limiting resolution steps to isolate the active (S)-enantiomer. This patented route utilizes single chiral (S)-piperidine-2-carboxylic acid as the starting material, completely eliminating the need for resolution. This not only theoretically doubles the yield relative to racemic starts but also removes the complexity and waste associated with separating enantiomers, leading to a more efficient and cost-effective process.
Q: What are the safety advantages of avoiding triphosgene in ropivacaine production?
A: Previous synthetic routes, such as those reported in earlier literature, utilized triphosgene to generate acid chlorides. Triphosgene is highly toxic, releases phosgene gas upon decomposition, and poses significant storage and handling hazards. The new method replaces this dangerous reagent with thionyl chloride under controlled, mild conditions. This substitution drastically reduces occupational health risks, simplifies waste treatment protocols, and aligns the manufacturing process with modern green chemistry and environmental compliance standards.
Q: Why is the order of reaction steps critical in this specific synthesis?
A: The innovation lies in performing the reductive amination to install the N-propyl group before forming the amide bond with 2,6-dimethylaniline. By alkylating the piperidine nitrogen first, the process avoids the need to protect and deprotect the amine functionality, which is required in other routes. Furthermore, this sequence inhibits the formation of quaternary ammonium salt impurities that can occur if alkylation is attempted on the final amide product, thereby ensuring a cleaner reaction profile and higher final purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ropivacaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the route disclosed in CN113735760A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific reductive amination and acylation steps under stringent purity specifications. Our rigorous QC labs ensure that every batch of ropivacaine hydrochloride meets the highest international standards, with impurity profiles tightly controlled to support regulatory filings in major markets.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific supply needs. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your organization. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing process is seamless, compliant, and commercially successful.
