Advanced Synthesis of Trifluoromethylfluorene Derivatives for Next-Generation OLED Displays
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer superior luminous efficiency and color purity, particularly in the challenging blue spectrum. Patent CN103666453A introduces a significant breakthrough in this domain by disclosing a novel blue fluorescent material containing trifluoromethylfluorene, specifically 2,7-bis-(3,5-bis-(trifluoromethyl)) phenyl-9,9-diethyl fluorene. This organic small molecule, characterized by the molecular formula C33H22F12, represents a strategic advancement for manufacturers seeking reliable electronic chemical suppliers. The patent details a robust preparation method that leverages microwave-assisted synthesis to achieve yields ranging from 66% to 90%, addressing the critical need for scalable and efficient production processes in the display sector. By integrating specific fluorine-containing substituents, this material not only enhances optical performance but also simplifies the manufacturing workflow, making it a highly attractive candidate for commercial scale-up of complex polymer additives and small molecule emitters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for high-performance fluorescent materials often suffer from cumbersome multi-step procedures that require stringent reaction conditions, such as extremely low temperatures or prolonged heating under strict inert atmospheres. These conventional methods frequently rely on expensive transition metal catalysts that are difficult to remove completely, leading to potential contamination issues that can degrade the performance of the final optoelectronic device. Furthermore, standard thermal heating methods often result in uneven energy distribution, causing side reactions that generate impurities and lower the overall yield, which drastically increases the cost of goods sold. The reliance on inert gas protection, such as nitrogen or argon, adds significant operational complexity and infrastructure costs to the manufacturing process, creating bottlenecks for procurement managers looking for cost reduction in electronic chemical manufacturing. Additionally, the purification of these complex aromatic structures often requires repeated recrystallization or extensive chromatography, consuming large volumes of solvents and generating substantial chemical waste.
The Novel Approach
In stark contrast, the methodology described in patent CN103666453A utilizes a microwave-assisted Suzuki-Miyaura coupling reaction that dramatically streamlines the production timeline and operational requirements. This innovative approach allows the reaction to proceed efficiently under air atmosphere, effectively removing the need for costly inert gas setups and simplifying the reactor configuration. The use of microwave irradiation ensures rapid and uniform heating, which accelerates the reaction kinetics to completion within merely 20 to 40 minutes, a fraction of the time required by traditional thermal methods. This technique not only boosts the productive rate to impressive levels of up to 90% but also minimizes the formation of by-products, thereby simplifying the downstream purification process. By employing readily available starting materials like 2,7-dibromo-9,9-diethylfluorene and 3,5-di(trifluoromethyl)phenylboronic acid, the process ensures a stable supply chain and reduces dependency on exotic precursors.
Mechanistic Insights into Microwave-Assisted Suzuki-Miyaura Coupling
The core of this synthesis lies in the palladium-catalyzed cross-coupling between the aryl halide and the aryl boronic acid, facilitated by the unique energy transfer mechanisms of microwave chemistry. The palladium catalyst, whether PdCl2 or Pd(OAc)2, undergoes an oxidative addition with the dibromo-fluorene derivative, followed by transmetallation with the boronic acid species activated by the potassium carbonate base. The microwave energy directly couples with the polar molecules and ions in the reaction mixture, creating superheating effects that lower the activation energy barrier for the catalytic cycle. This results in a highly efficient reductive elimination step that forms the carbon-carbon bond linking the trifluoromethylphenyl groups to the fluorene core. The presence of the trifluoromethyl groups is not merely structural; their strong electron-withdrawing nature stabilizes the intermediate species and influences the electronic properties of the final product, ensuring the desired blue shift in fluorescence.
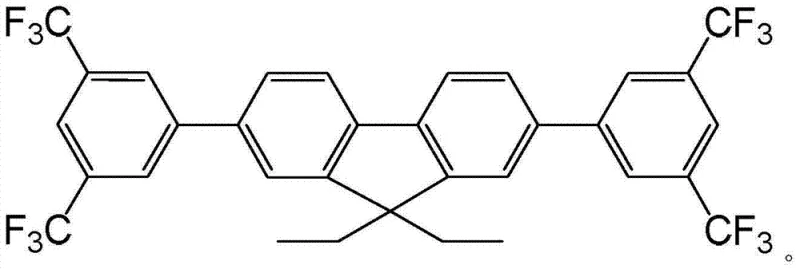
Controlling the impurity profile in such conjugated systems is paramount for achieving high-purity OLED material standards, and this process excels in that regard. The mild reaction conditions, operating between 65°C and 95°C, prevent thermal degradation of the sensitive fluorene backbone which can occur at higher temperatures. The specific stoichiometry, utilizing a molar ratio of reactants optimized to 1:2~5 for the boronic acid, ensures complete consumption of the dibromo precursor, minimizing the presence of mono-substituted intermediates. Furthermore, the workup procedure involving dichloromethane extraction and saturated brine washing effectively removes inorganic salts and polar by-products. The final purification via silica gel column chromatography using petroleum ether as an eluent guarantees the removal of trace palladium residues and homocoupling products, delivering a white powder product that meets the rigorous specifications required for high-end display applications.
How to Synthesize 2,7-bis-(3,5-bis-(trifluoromethyl)) phenyl-9,9-diethyl fluorene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. Operators begin by mixing the core fluorene derivative with the trifluoromethyl-substituted boronic acid in a solvent system that may include toluene, ethanol, or DMF mixed with water. The addition of a base like potassium carbonate and a precise amount of palladium catalyst initiates the coupling upon exposure to microwave radiation.
- Mix 2,7-dibromo-9,9-diethylfluorene, 3,5-di(trifluoromethyl)phenylboronic acid, potassium carbonate, and a palladium catalyst in a suitable solvent system.
- Subject the reaction mixture to microwave irradiation at temperatures between 65°C and 95°C for 20 to 40 minutes under air atmosphere.
- Perform standard workup including extraction with dichloromethane, washing with brine, drying over magnesium sulfate, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative benefits that extend beyond mere technical performance. The ability to conduct the reaction under air atmosphere eliminates the capital expenditure associated with inert gas generation and monitoring systems, leading to substantial cost savings in facility operations. The drastic reduction in reaction time from several hours to under an hour significantly increases reactor turnover rates, allowing existing infrastructure to produce higher volumes without additional investment. This efficiency translates directly into a more competitive pricing structure for the final high-purity electronic chemical, making it an attractive option for cost-sensitive mass production environments. Moreover, the use of common solvents and commercially available catalysts reduces supply chain risks associated with sourcing specialized reagents.
- Cost Reduction in Manufacturing: The elimination of inert atmosphere requirements and the reduction in energy consumption due to shorter reaction times contribute to a significantly lowered operational cost profile. By avoiding the need for expensive ligand systems or exotic catalysts, the raw material costs are kept manageable, allowing for better margin control. The high yield range of 66% to 90% ensures that raw material utilization is maximized, reducing the cost per kilogram of the active pharmaceutical ingredient or electronic material produced. Additionally, the simplified workup reduces solvent usage and waste disposal fees, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium carbonate, ethanol, and toluene ensures that the supply chain remains robust against market fluctuations. Since the process does not depend on single-source proprietary catalysts, procurement teams have the flexibility to source materials from multiple vendors, mitigating the risk of supply disruptions. The scalability of the microwave-assisted process from gram to kilogram scales ensures that lead times for high-purity OLED materials can be consistently met, even during periods of high demand. This reliability is crucial for maintaining continuous production lines in the fast-paced consumer electronics sector.
- Scalability and Environmental Compliance: The process generates less chemical waste due to higher selectivity and fewer side reactions, aligning with increasingly strict environmental regulations and sustainability goals. The ability to scale this reaction using continuous flow microwave reactors offers a pathway to ton-scale production while maintaining the safety and efficiency observed at the bench scale. Reduced solvent consumption and the potential for solvent recycling further minimize the environmental footprint of the manufacturing process. This compliance with green chemistry principles enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this trifluoromethylfluorene derivative. These insights are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance.
Q: What are the key advantages of using microwave irradiation for this fluorescent material synthesis?
A: Microwave irradiation significantly accelerates the reaction kinetics, reducing reaction times from hours to merely 20-40 minutes while maintaining high yields between 66% and 90%, thereby enhancing throughput efficiency.
Q: Why is the trifluoromethyl group critical for this blue fluorescent material?
A: The trifluoromethyl groups exert a strong electron-withdrawing effect that offsets the conjugated system electronics, causing a blue shift in absorption and modifying the LUMO energy levels essential for blue emission.
Q: Is an inert atmosphere required for this palladium-catalyzed coupling reaction?
A: Uniquely, this patented process operates effectively under air atmosphere, eliminating the need for expensive and cumbersome inert gas protection systems typically required for palladium catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-bis-(3,5-bis-(trifluoromethyl)) phenyl-9,9-diethyl fluorene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent materials play in the next generation of display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from R&D to full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of our electronic chemicals meets the exacting standards required for OLED applications. Our commitment to quality assurance means that you receive a product with a consistent impurity profile, essential for maintaining the longevity and efficiency of your final devices.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific product needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive innovation in your product line.
