Advanced Synthesis of 6-(Dibromomethyl)-2-Methyl Quinazoline-4(3H)-Ketone for Commercial Scale-Up
Advanced Synthesis of 6-(Dibromomethyl)-2-Methyl Quinazoline-4(3H)-Ketone for Commercial Scale-Up
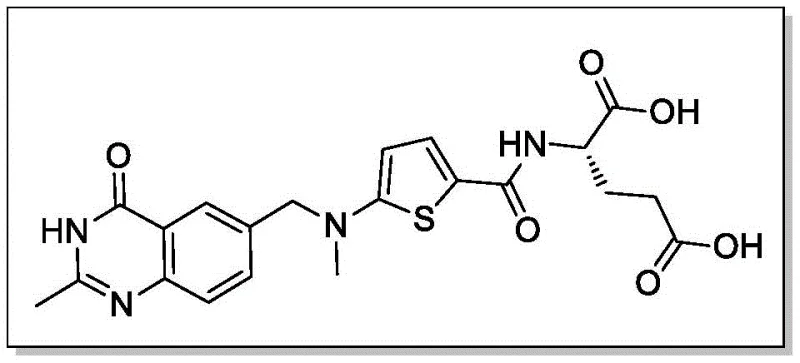
The pharmaceutical industry constantly seeks robust synthetic routes for critical oncology intermediates, particularly for thymidylate synthase inhibitors like Raltitrexed. Patent CN111995588A discloses a highly efficient synthesis method for 6-(dibromomethyl)-2-methyl quinazoline-4(3H)-ketone, a pivotal building block in this therapeutic class. This novel approach addresses longstanding challenges in regioselectivity and purity that have plagued traditional manufacturing processes. By introducing a strategic Boc-protection step prior to bromination, the inventors have created a pathway that significantly minimizes side reactions and simplifies purification. This technical breakthrough offers a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in producing high-quality reference standards and active drug substances. The following analysis details the mechanistic superiority and commercial viability of this protected route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalizing the quinazoline scaffold often suffer from poor regioselectivity during halogenation steps. When attempting to brominate 2,6-dimethyl quinazoline-4(3H)-one directly, chemists frequently encounter a complex mixture of products due to the similar reactivity of the methyl groups at the 2 and 6 positions. The absence of a directing group or protective mask allows radical species to attack multiple sites, leading to significant formation of 2-bromomethyl impurities alongside the desired 6-bromomethyl or 6-dibromomethyl species. This lack of selectivity necessitates rigorous and costly purification protocols, such as repeated recrystallization or preparative chromatography, which drastically reduce overall yield and increase production costs. Furthermore, the polar nature of the unprotected quinazolinone can lead to solubility issues in non-polar organic solvents preferred for radical reactions, complicating reaction homogeneity and heat transfer during scale-up operations.
The Novel Approach
The methodology described in the patent overcomes these deficiencies by employing a temporary Boc-protecting group on the quinazoline nitrogen. This strategic modification fundamentally alters the electronic and steric environment of the molecule, effectively reducing its polarity and enhancing solubility in common organic solvents like THF and dichloromethane. More critically, the bulky tert-butoxycarbonyl group exerts a steric influence that shields the adjacent 2-methyl position, thereby directing the brominating agent exclusively toward the more accessible 6-methyl group. This results in a highly selective transformation where the formation of unwanted 2-position isomers is virtually eliminated. The subsequent deprotection step is mild and efficient, yielding the target 6-(dibromomethyl) compound with exceptional purity, thus streamlining the entire manufacturing workflow for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Boc-Protection and Radical Bromination
The core of this synthetic innovation lies in the modulation of electron density through N-protection. In the first step, the acidic proton on the quinazoline nitrogen is deprotonated by a strong base such as sodium hydride or LDA, generating a nucleophilic anion. This anion attacks di-tert-butyl dicarbonate (Boc2O), installing the carbamate protecting group. This transformation converts the hydrogen-bond donating lactam into a neutral carbamate, which significantly lowers the dipole moment of the molecule. In the subsequent radical bromination step, the reaction proceeds via a free-radical chain mechanism initiated by benzoyl peroxide (BPO). The succinimidyl radical generated from NBS abstracts a hydrogen atom from the benzylic methyl group. Due to the electronic withdrawal and steric bulk of the Boc group at the N3 position, the bond dissociation energy of the C-H bonds at the 2-methyl group is effectively increased relative to the 6-methyl group. Consequently, the radical abstraction occurs preferentially at the 6-position, followed by bromine atom transfer to form the mono- and eventually the dibromomethyl species with high fidelity.
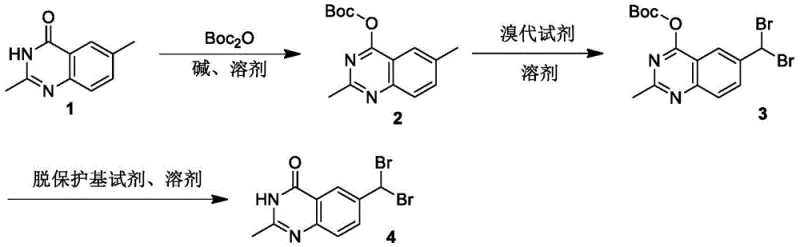
Impurity control is inherently built into this three-step sequence through the reversibility and specificity of the protecting group chemistry. By isolating the intermediate tert-butyl (6-(dibromomethyl)-2-methylquinazolin-4-yl) formate, manufacturers can verify the success of the selective bromination before committing to the final deprotection. Any unreacted starting material or mono-brominated intermediates can be removed at this stage if necessary, although the high selectivity usually renders this unnecessary. The final acidic hydrolysis using reagents like trifluoroacetic acid (TFA) or HCl cleaves the Boc group cleanly without affecting the sensitive dibromomethyl functionality, provided the temperature is controlled between 0°C and 40°C. This precise control prevents the hydrolysis of the dibromomethyl group into an aldehyde, a common degradation pathway in aqueous acidic conditions, ensuring the structural integrity of the high-purity pharmaceutical intermediates required for downstream coupling reactions.
How to Synthesize 6-(Dibromomethyl)-2-Methyl Quinazoline-4(3H)-Ketone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of stoichiometry and temperature control. The process begins with the dissolution of 2,6-dimethyl quinazoline-4(3H)-one in anhydrous THF under inert atmosphere, followed by the careful addition of base to generate the nucleophile. Once the Boc group is installed and the intermediate purified, the bromination is conducted under reflux conditions with a slight excess of NBS to drive the reaction to the dibromo stage. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and workup procedures required to achieve the reported yields and purity levels consistently.
- Protect the quinazoline nitrogen with a Boc group using base and Boc2O to reduce polarity and improve solubility.
- Perform selective radical bromination on the 6-methyl group using NBS and a radical initiator like BPO.
- Remove the Boc protecting group using an acidic reagent such as TFA or HCl to yield the final dibromomethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Boc-protected synthetic route translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the purification process; because the reaction selectivity is so high, the need for expensive and time-consuming chromatographic separations is minimized or eliminated. This reduction in downstream processing directly correlates to lower manufacturing costs and shorter batch cycle times, allowing for faster turnover and improved responsiveness to market demand. Furthermore, the reagents utilized—such as Boc2O, NBS, and common organic solvents—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialized synthetic routes relying on exotic catalysts.
- Cost Reduction in Manufacturing: The elimination of complex purification steps significantly lowers the cost of goods sold (COGS). By avoiding the loss of material associated with multiple recrystallizations or column chromatography, the overall mass balance of the process is improved. Additionally, the use of standard, non-precious metal reagents avoids the regulatory and financial burdens associated with heavy metal catalyst removal and validation, further driving down the total cost of production for this critical intermediate.
- Enhanced Supply Chain Reliability: The robustness of this three-step sequence ensures consistent batch-to-batch quality, which is paramount for maintaining regulatory compliance in pharmaceutical manufacturing. The high selectivity reduces the variance in impurity profiles, making the validation process smoother and more predictable. This reliability allows supply chain planners to forecast inventory needs with greater accuracy, minimizing the need for safety stock and reducing working capital tied up in intermediate inventory.
- Scalability and Environmental Compliance: The process utilizes solvents and conditions that are amenable to large-scale reactor operations, with exotherms that are manageable through standard cooling protocols. The waste streams generated are primarily organic salts and succinimide byproducts, which are easier to treat and dispose of compared to waste containing heavy metals or highly toxic reagents. This environmental profile facilitates easier permitting and aligns with modern green chemistry initiatives, ensuring long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method in a GMP environment. Understanding the nuances of the protection-deprotection strategy is essential for technology transfer teams aiming to replicate the high yields and purity reported in the patent literature. These insights are derived directly from the experimental data and mechanistic rationale provided in the source documentation.
Q: Why is Boc protection necessary for the bromination of 2,6-dimethyl quinazoline-4(3H)-one?
A: Boc protection significantly reduces the polarity of the starting material and increases steric hindrance at the nitrogen position. This electronic and steric modification effectively suppresses unwanted bromination at the 2-methyl group, ensuring high selectivity for the 6-position methyl group during the radical reaction.
Q: What are the optimal conditions for the radical bromination step?
A: The patent suggests using N-bromosuccinimide (NBS) with benzoyl peroxide (BPO) as an initiator in solvents like chloroform or dichloromethane. The reaction is typically heated to reflux (around 80°C) for 2 to 3 hours to ensure complete conversion while maintaining selectivity.
Q: How does this method improve impurity control compared to direct bromination?
A: Direct bromination often leads to a mixture of mono- and di-brominated products at both the 2 and 6 positions. By masking the nitrogen with a Boc group, the electron density is modulated, preventing over-bromination and positional isomers, which simplifies downstream purification and enhances overall product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(Dibromomethyl)-2-Methyl Quinazoline-4(3H)-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team has extensively analyzed the Boc-protection strategy disclosed in CN111995588A and possesses the expertise to implement this route with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 6-(dibromomethyl)-2-methyl quinazoline-4(3H)-ketone meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize your supply chain for Raltitrexed and related quinazoline derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your drug development goals.
