Advanced Silver-Catalyzed Cyanation for High-Purity Pharmaceutical Intermediates Manufacturing
Advanced Silver-Catalyzed Cyanation for High-Purity Pharmaceutical Intermediates Manufacturing
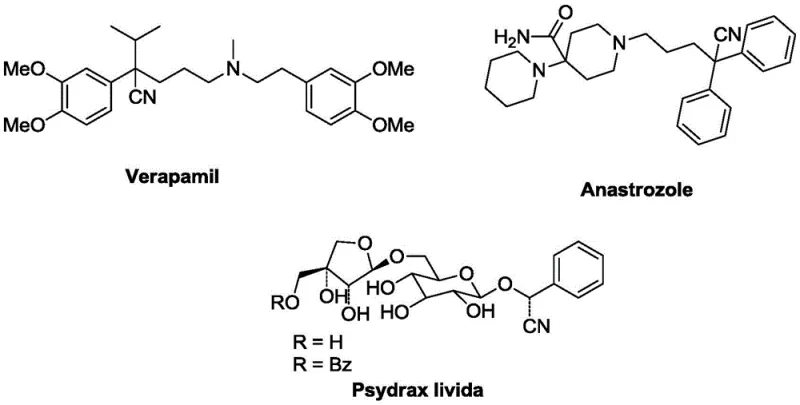
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for safer, more efficient, and environmentally benign methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN110240554B, which details a novel synthetic route for alpha-thioether aryl acetonitrile compounds. These structural motifs are not merely academic curiosities; they serve as critical scaffolds in the construction of complex bioactive molecules. For instance, the therapeutic landscape includes prominent drugs such as Verapamil, a coronary dilator, and Anastrozole, a cornerstone in breast cancer treatment, both of which rely on nitrile-containing precursors for their assembly. Furthermore, natural products like Psydrax livida demonstrate the ubiquity of these frameworks in biological systems. The patent introduces a transformative approach that leverages silver trifluoromethanesulfonate (AgOTf) catalysis to achieve direct cyanation, offering a compelling alternative to legacy methods that often suffer from harsh conditions or toxic reagent usage.
The development of robust synthetic routes for alpha-thioether aryl acetonitriles has historically been plagued by significant technical hurdles. Conventional methodologies, while effective in specific contexts, often impose severe limitations on process chemistry and supply chain flexibility. For example, earlier strategies reported by Zhao et al. relied on the nucleophilic substitution of alpha-bromophenylacetonitriles with sodium methanesulfonate. While chemically sound, this approach is fundamentally constrained by the difficulty in preparing the alpha-halo starting materials, which often require multi-step sequences and generate stoichiometric amounts of halogenated waste. Similarly, the method described by Beluakov utilizes alpha-(benzotriazolyl) methylphenyl sulfide reacting with TMSCN. Although this route offers moderate to excellent yields, the starting sulfide substrates are complex and not readily available off-the-shelf, creating a bottleneck for procurement teams seeking reliable raw material sources.
Further compounding the challenges of traditional synthesis is the reliance on expensive or hazardous reagents. The protocol developed by Okino employs tin tetrachloride (SnCl4) and trimethylsilyl cyanide (TMSCN). While TMSCN is a potent cyanating agent, it is relatively costly compared to simpler alternatives and poses hydrolysis risks that complicate large-scale handling. Additionally, the use of Lewis acids like SnCl4 necessitates rigorous anhydrous conditions and generates corrosive waste streams that increase environmental compliance costs. Another historical approach by Marchand involves the reaction of nitriles with disulfides under strongly basic conditions using anhydrous potassium hydroxide. This method often suffers from limited substrate scope and can lead to side reactions due to the harsh alkaline environment, potentially compromising the purity profile of the final intermediate. These cumulative drawbacks highlight the urgent need for a more streamlined, catalytic approach that bypasses pre-functionalized substrates.
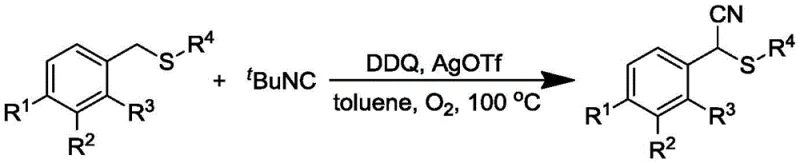
The novel methodology disclosed in patent CN110240554B represents a paradigm shift by employing a direct oxidative cyanation strategy. At the heart of this transformation is the use of tert-butyl isonitrile as the cyano group source, activated by a silver trifluoromethanesulfonate catalyst in the presence of 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as an oxidant. This system operates under an oxygen atmosphere in toluene at a mild reflux temperature of 100 °C. The mechanistic elegance lies in the ability of the silver catalyst to coordinate with the isonitrile and facilitate the insertion of the cyano group directly into the benzylic position of the sulfide substrate. This eliminates the need for pre-installed leaving groups like halides or benzotriazoles, thereby shortening the synthetic linear sequence. The reaction demonstrates remarkable tolerance for various substituents on the aromatic ring, including electron-donating groups like methyl and isopropyl, as well as electron-withdrawing groups such as fluoro, chloro, and bromo, ensuring broad applicability across different drug discovery programs.
From a mechanistic perspective, the synergy between the silver catalyst and the quinone oxidant is crucial for controlling the reaction pathway and minimizing impurities. The AgOTf likely acts as a Lewis acid to activate the isonitrile carbon, making it more susceptible to nucleophilic attack or radical coupling, while DDQ serves to regenerate the active catalytic species or participate in a hydrogen atom transfer (HAT) process to generate the reactive benzylic radical intermediate. This dual-activation system ensures that the reaction proceeds with high chemoselectivity, avoiding over-oxidation of the sulfide moiety to sulfoxides or sulfones, which are common byproducts in oxidative environments. The mild thermal conditions (100 °C) further contribute to a clean impurity profile by preventing thermal degradation of sensitive functional groups. For R&D directors, this means that downstream purification is significantly simplified, often requiring only standard column chromatography to achieve high-purity specifications suitable for subsequent coupling reactions in API synthesis.
How to Synthesize Alpha-Thioether Aryl Acetonitrile Efficiently
The operational simplicity of this new protocol makes it highly attractive for process development teams aiming to establish robust manufacturing procedures. The general procedure involves charging a reactor with the benzyl sulfide substrate, tert-butyl isonitrile, the silver catalyst, and DDQ in toluene, followed by heating under an oxygen atmosphere. The reaction progress is easily monitored by thin-layer chromatography (TLC), with typical completion times around 3 hours. Upon completion, the workup is straightforward: the solvent is removed, and the crude product is purified. This streamlined workflow reduces the operational burden on plant personnel and minimizes the risk of exposure to hazardous intermediates.
- Charge a reactor with benzyl sulfide substrate, tert-butyl isonitrile, silver trifluoromethanesulfonate catalyst, and DDQ oxidant in toluene solvent under an oxygen atmosphere.
- Heat the reaction mixture to reflux at 100 °C and maintain stirring until TLC analysis confirms the complete consumption of starting materials, typically requiring approximately 3 hours.
- Cool the system to room temperature, remove the solvent under reduced pressure, and purify the resulting crude residue via column chromatography to isolate the target nitrile compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silver-catalyzed cyanation route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the simplification of the raw material portfolio. By utilizing readily available benzyl sulfides and commercially sourced tert-butyl isonitrile, the process bypasses the need for custom-synthesized, multi-step precursors like alpha-halo nitriles or benzotriazole derivatives. This shift significantly reduces the complexity of the supply chain, mitigating the risk of delays associated with sourcing niche starting materials. Furthermore, the elimination of stoichiometric heavy metal reagents like tin tetrachloride reduces the burden on waste management infrastructure, leading to substantial cost savings in environmental compliance and disposal fees.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the catalytic nature of the silver system. Although silver salts are precious metals, they are used in low molar quantities (0.10-0.15 equivalents), and the overall atom economy is improved by avoiding the generation of stoichiometric salt waste typical of nucleophilic substitution reactions. Additionally, the use of toluene, a commodity solvent, instead of specialized anhydrous solvents or cryogenic media, drastically lowers the operational expenditure (OPEX) related to solvent procurement and recovery. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or cooling cycles, contributing to a more sustainable and cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining supply continuity in the pharmaceutical industry. The tolerance of the method towards various functional groups means that a single standardized protocol can be applied to a wide range of analogues, reducing the need for extensive process re-validation when switching between different intermediates. This flexibility allows manufacturers to respond more agilely to changing demand patterns for specific API precursors. Moreover, the stability of tert-butyl isonitrile compared to gaseous cyanide sources simplifies logistics and storage, removing a significant safety bottleneck from the supply chain.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the use of standard equipment and non-hazardous reagents. The absence of highly toxic cyanide salts or corrosive Lewis acids simplifies the engineering controls required for large-scale reactors. The oxidative nature of the reaction, utilizing molecular oxygen or air, is inherently greener than methods relying on stoichiometric oxidants that generate heavy metal waste. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the method's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using tert-butyl isonitrile over traditional cyanide sources?
A: Tert-butyl isonitrile serves as a safe, solid, and easy-to-handle cyano source, eliminating the severe safety hazards and toxicity associated with gaseous HCN or alkali metal cyanides often used in conventional nucleophilic substitution methods.
Q: How does the silver trifluoromethanesulfonate catalyst influence reaction selectivity?
A: The AgOTf catalyst works synergistically with the DDQ oxidant to facilitate a mild oxidative functionalization of the benzylic C-H bond, ensuring high regioselectivity for the alpha-position without requiring pre-functionalized halogenated starting materials.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the protocol utilizes common organic solvents like toluene and operates at standard reflux temperatures (100 °C), avoiding cryogenic conditions or ultra-high pressures, which makes the process highly amenable to scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Thioether Aryl Acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutics. Our technical team has extensively evaluated the silver-catalyzed cyanation pathway described in CN110240554B and confirmed its potential for delivering high-purity alpha-thioether aryl acetonitriles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from clinical trials to full-scale market supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you optimize your supply chain and accelerate your time to market with our reliable alpha-thioether aryl acetonitrile supply solutions.
