Scalable Synthesis of Fluorinated Hydroxyquinoline-Aluminum Complexes for High-Performance Organic Photovoltaics
Scalable Synthesis of Fluorinated Hydroxyquinoline-Aluminum Complexes for High-Performance Organic Photovoltaics
The rapid evolution of the organic photovoltaic (OPV) sector demands materials that combine high electron mobility with exceptional batch-to-batch consistency. Patent CN110790760A introduces a groundbreaking class of fluorinated hydroxyquinoline-based metal-organic small molecule complexes bridged with aromatic groups of naphthalene diimides. Unlike traditional polymeric acceptors which often suffer from ill-defined molecular weights and polydispersity, these novel small molecule complexes utilize a metal core (such as Aluminum, Gallium, or Magnesium) coordinated with functionalized ligands. This structural precision allows for superior crystallization capabilities and enhanced electron affinity, directly addressing the reproducibility challenges faced in commercial organic solar cell manufacturing. The technology represents a significant leap forward for reliable electronic chemical suppliers aiming to provide high-purity n-type materials for next-generation energy devices.
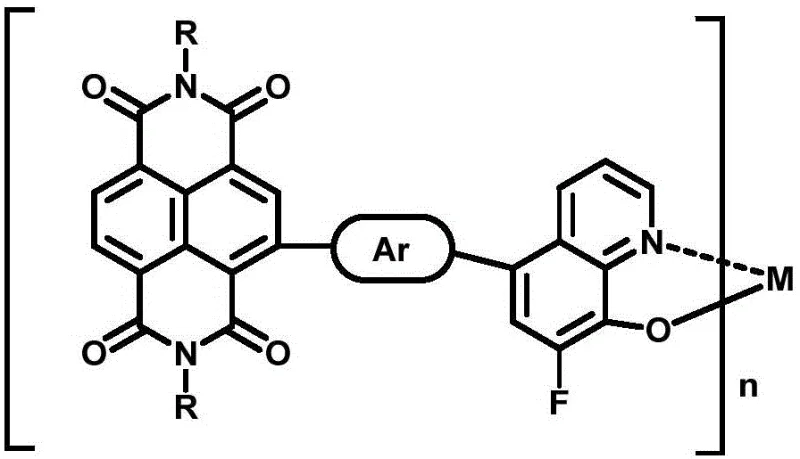
The core innovation lies in the synergistic combination of a fluorinated hydroxyquinoline unit and a naphthalene diimide (NDI) backbone. The hydroxyquinoline moiety acts as a potent electron-withdrawing group, a property that is further amplified by the strategic introduction of fluorine atoms. This modification not only strengthens the intramolecular charge transfer but also facilitates non-covalent interactions with the electron-donating bridging units, ensuring that charge carriers are transported efficiently within a planar molecular architecture. By grafting the NDI unit, the material achieves improved crystallinity, which is critical for forming high-quality thin films. For procurement teams evaluating cost reduction in display & optoelectronic materials manufacturing, this defined small-molecule approach eliminates the variability inherent in polymer synthesis, offering a more predictable and controllable supply chain for active layer components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic semiconductor materials has been dominated by polymeric systems. While polymers offer good film-forming properties, they present significant hurdles for industrial scale-up and quality control. The primary limitation is the inevitable batch-to-batch variation in molecular weight distribution and end-group functionality, which leads to inconsistent device performance and complicates the optimization of power conversion efficiency. Furthermore, the purification of high molecular weight polymers is notoriously difficult, often requiring extensive Soxhlet extraction processes that consume large volumes of solvents and time. These factors contribute to higher production costs and longer lead times, creating bottlenecks for commercial scale-up of complex polymer additives and active materials. Additionally, the amorphous nature of many polymeric acceptors can limit charge carrier mobility compared to their highly ordered small-molecule counterparts.
The Novel Approach
The methodology described in CN110790760A circumvents these issues by employing a discrete small-molecule architecture centered around a metal coordination complex. This approach ensures that every molecule in the batch is structurally identical, thereby eliminating polydispersity and guaranteeing consistent electronic properties across different production runs. The synthesis relies on robust palladium-catalyzed cross-coupling reactions followed by a straightforward metal coordination step, both of which are well-understood processes in fine chemical manufacturing. This transition from undefined polymers to defined metal-organic complexes simplifies the purification workflow, often allowing for standard recrystallization or column chromatography techniques. Consequently, this enables the production of high-purity OLED material precursors and OPV acceptors with significantly reduced impurity profiles, enhancing the overall reliability and lifespan of the final optoelectronic devices.
Mechanistic Insights into Stille Coupling and Metal Coordination
The synthesis of these advanced materials proceeds through a sophisticated yet scalable two-stage mechanism. The first critical stage involves the construction of the organic ligand framework via a Stille coupling reaction. In this step, a tin-substituted fluorohydroxyquinoline derivative reacts with a dibromo-naphthalenediimide core in the presence of a tetrakis(triphenylphosphine)palladium(0) catalyst. The reaction typically occurs in toluene at elevated temperatures around 105°C, facilitating the formation of a carbon-carbon bond between the thiophene bridge and the NDI core. This step is crucial for establishing the conjugated system necessary for electron transport. The use of the Stille protocol is particularly advantageous here due to its tolerance of various functional groups and its ability to proceed under relatively mild conditions compared to other cross-coupling methods, ensuring the integrity of the sensitive fluorine and hydroxyl functionalities.
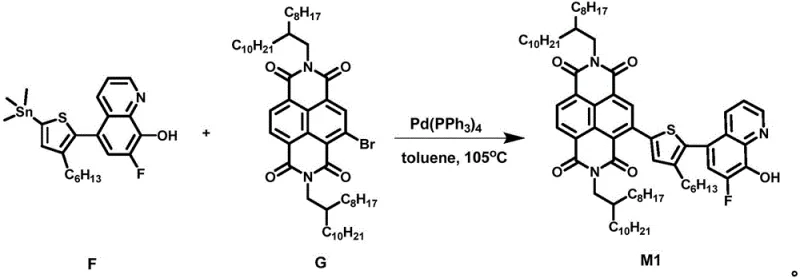
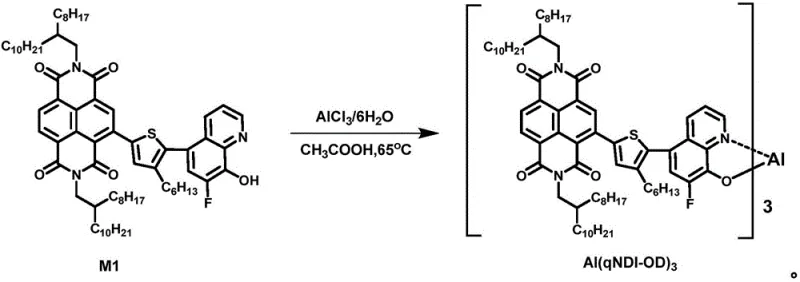
Following the formation of the organic ligand, the second stage involves the coordination of the metal center to complete the complex. This is achieved by reacting the purified ligand with a metal salt, such as aluminum chloride hexahydrate (AlCl3·6H2O), in a mixed solvent system of acetic acid and deionized water. The reaction is conducted under a nitrogen atmosphere at temperatures ranging from 65°C to 80°C. During this process, the hydroxyl group of the quinoline and the nitrogen atom coordinate with the metal ion, forming a stable five-membered chelate ring. The pH of the reaction mixture is carefully adjusted to between 7 and 8 to induce the precipitation of the final yellow solid product. This coordination step locks the molecular conformation, enhancing thermal stability and defining the electronic landscape of the molecule for optimal device performance.
How to Synthesize Fluorinated Hydroxyquinoline Complexes Efficiently
The preparation of these high-performance electron acceptors requires precise control over reaction stoichiometry and purification parameters to ensure maximum yield and purity. The process begins with the synthesis of the tin-substituted quinoline intermediate, followed by the pivotal Stille coupling with the NDI derivative. Detailed operational protocols, including specific molar ratios of catalysts and monomers, are essential for reproducing the high electron mobility reported in the patent literature. Manufacturers must adhere to strict anhydrous conditions during the coupling phase to prevent catalyst deactivation, while the subsequent metal coordination step benefits from controlled pH adjustment to maximize precipitation efficiency. For a comprehensive guide on executing this synthesis with industrial-grade reproducibility, please refer to the standardized procedure outlined below.
- Perform Stille coupling between a trimethyltin-substituted fluorohydroxyquinoline derivative and a dibromo-naphthalenediimide core using Pd(PPh3)4 catalyst in toluene at 105°C.
- Purify the resulting organic ligand intermediate via silica gel column chromatography using petroleum ether and dichloromethane eluents.
- Execute metal coordination by reacting the purified ligand with AlCl3·6H2O in an acetic acid and deionized water mixture at 65-80°C, adjusting pH to 7-8 to precipitate the final complex.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this small-molecule metal-organic complex technology offers substantial strategic benefits for supply chain managers and procurement officers in the organic electronics sector. By shifting away from polymeric systems with variable properties, companies can secure a more stable supply of active materials that meet rigorous specification standards. The synthetic route utilizes commercially available starting materials and standard catalytic systems, which mitigates the risk of raw material shortages and price volatility associated with exotic monomers. Furthermore, the simplified purification process reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals within the chemical industry.
- Cost Reduction in Manufacturing: The transition to a defined small-molecule synthesis eliminates the need for complex fractionation and extensive purification steps typically required for polymers. Since the molecular structure is fixed, quality control testing becomes faster and less resource-intensive, as there is no need to characterize molecular weight distributions. The use of standard palladium catalysts and common solvents like toluene and acetic acid ensures that reagent costs remain predictable and manageable. Additionally, the high thermal stability of the final products reduces the risk of material degradation during device fabrication, lowering scrap rates and improving overall manufacturing yield.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on robust chemical transformations that are easily scalable from laboratory to pilot plant volumes. The intermediates involved, such as the brominated quinolines and naphthalenediimides, are accessible through established supply chains, reducing dependency on single-source specialty vendors. This diversification of raw material sources enhances supply security and reduces lead times for high-purity electronic chemicals. Moreover, the solid-state nature of the final complexes facilitates easier storage and transportation compared to sensitive polymeric solutions, ensuring material integrity upon delivery to device fabrication facilities.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the metal coordination step in aqueous acetic acid, are inherently greener than many organic synthesis protocols that rely on harsh halogenated solvents throughout. The ability to precipitate the product by pH adjustment minimizes the need for energy-intensive distillation for product isolation. This process efficiency supports cost reduction in electronic chemical manufacturing by lowering utility costs and waste disposal fees. The scalability of the Stille coupling and coordination steps allows for seamless expansion to multi-kilogram production runs without significant re-engineering of the process, ensuring that supply can meet growing market demand for high-efficiency organic solar cells.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fluorinated hydroxyquinoline-based complexes. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy for R&D and procurement decision-making. Understanding these details is crucial for integrating these materials into existing device architectures and supply chains effectively.
Q: What are the thermal stability characteristics of these aluminum complexes?
A: According to patent CN110790760A, the synthesized complexes such as Al(qNDI-OD)3 exhibit excellent thermal stability, with decomposition temperatures exceeding 450°C at 5% mass loss, making them suitable for high-temperature device processing.
Q: How does the fluorine substitution affect device performance?
A: The introduction of fluorine atoms enhances the electron-withdrawing capability of the hydroxyquinoline unit. This promotes stronger intramolecular charge transfer and forms non-covalent bonds with the bridging unit, ensuring efficient carrier transport in a planar structure.
Q: Can these materials be processed using solution-based methods?
A: Yes, the patent highlights that these small molecule complexes possess good solubility in organic solvents like chlorobenzene and chloroform, allowing for film formation via spin-coating, which is compatible with low-cost solution processing techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Hydroxyquinoline Complexes Supplier
As the global demand for high-efficiency organic photovoltaics continues to surge, the need for precise, high-purity electron acceptor materials has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this technological shift, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive palladium-catalyzed reactions and metal coordination steps required for these advanced materials, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to process optimization, we deliver the consistency and reliability that top-tier device manufacturers require to maintain their competitive edge in the renewable energy sector.
We invite you to collaborate with us to explore how these novel small-molecule complexes can enhance your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and processing constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate the commercialization of next-generation organic electronic devices.
