Advanced Chiral Spiro Ligands for High-Efficiency Asymmetric Hydrogenation and Commercial Scale-Up
The landscape of asymmetric catalysis has been fundamentally shifted by the innovations detailed in Chinese Patent CN102040625B, which introduces a novel class of chiral spiro pyridylamidophosphine ligand compounds. These sophisticated molecular architectures, characterized by a rigid chiral spirodihydroindane skeleton, represent a significant leap forward in the design of P-N ligands for transition metal catalysis. Unlike conventional ligands that often struggle to balance steric bulk with electronic flexibility, this patented technology offers a unique structural motif that enables exceptional control over stereochemistry during hydrogenation reactions. For R&D directors and process chemists seeking reliable solutions for synthesizing high-purity pharmaceutical intermediates, this development addresses the critical need for catalysts that combine high turnover numbers with robust enantioselectivity. The core innovation lies in the integration of a pyridine amine moiety onto a pre-existing chiral spiro phosphine backbone, creating a hemilabile ligand system that stabilizes the active iridium species while allowing substrate access.
Furthermore, the versatility of this ligand class is underscored by its ability to accommodate a wide range of substituents on both the phosphine and pyridine rings, allowing for fine-tuning of the catalyst's electronic and steric properties to match specific substrate requirements. This adaptability is crucial for the commercial scale-up of complex pharmaceutical intermediates where minor structural changes in the substrate can drastically affect reaction outcomes. By leveraging the synthetic methodologies outlined in this patent, manufacturers can access a library of ligands capable of driving asymmetric hydrogenation reactions with yields exceeding 90% and enantiomeric excess values reaching up to 99.9% ee. This level of performance is not merely an academic curiosity but a tangible asset for supply chain heads looking to secure consistent, high-quality production routes for chiral alcohols and amines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of carbonyl compounds has relied heavily on ruthenium-based catalysts, most notably the Noyori-type bisphosphine/diamine systems. While these catalysts revolutionized the field, they suffer from inherent limitations that complicate their industrial application. A primary drawback is the stringent requirement for precise matching between the chirality of the bisphosphine ligand and the diamine ligand; a mismatch often leads to catastrophic drops in both activity and enantioselectivity. Additionally, these traditional systems frequently require relatively high catalyst loadings to achieve acceptable conversion rates, which escalates the cost of goods due to the expense of ruthenium and the associated ligands. From a process safety and environmental perspective, the removal of residual heavy metals from the final API intermediate is a costly and time-consuming step, often requiring specialized scavenging resins or multiple crystallization cycles. Moreover, earlier generations of P-N ligands, such as those based on ferrocene or beta-amino alcohol scaffolds, often exhibited moderate enantioselectivity (typically below 80% ee) or limited substrate scope, failing to provide a universal solution for diverse ketone classes.
The Novel Approach
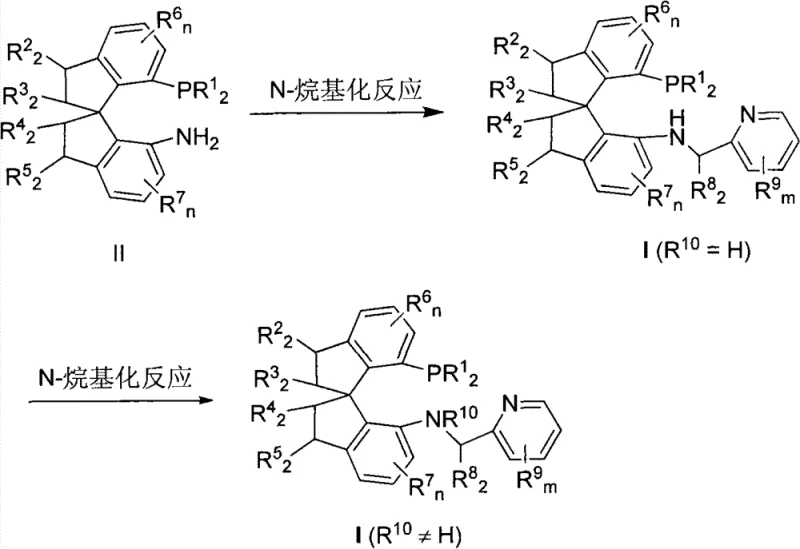
The technology disclosed in CN102040625B overcomes these hurdles by introducing a mono-dentate P-N ligand architecture built on a rigid spiro scaffold. This novel approach eliminates the need for complex bis-ligand matching, simplifying catalyst preparation and optimization. The spirodihydroindane backbone provides a fixed chiral environment that effectively transfers chirality to the substrate without the conformational flexibility that can lead to selectivity loss. Crucially, the iridium complexes derived from these ligands demonstrate remarkably high turnover numbers (TON), with effective catalyst loadings reported as low as 0.0001 mol%. This drastic reduction in catalyst usage directly translates to cost reduction in pharmaceutical intermediate manufacturing by minimizing precious metal consumption. The synthesis of the ligands themselves is also streamlined, utilizing a straightforward reductive amination of readily available chiral spiro aminophosphines with pyridine aldehydes. This operational simplicity ensures that the supply chain for these critical reagents remains robust and scalable, mitigating the risks associated with multi-step ligand syntheses.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The exceptional performance of these chiral spiro pyridylamidophosphine ligands stems from their unique coordination behavior with iridium centers. Upon complexation with iridium precursors such as [Ir(cod)Cl]2, the ligand forms a cationic or neutral active species where the phosphine atom binds strongly while the pyridine nitrogen acts as a labile coordination site. This hemilabile nature is key to the catalytic cycle; it allows the substrate to coordinate to the metal center more easily compared to rigid bidentate ligands that might create a sterically congested environment. During the hydrogenation cycle, the iridium-hydride species generated in situ transfers hydride to the carbonyl substrate in a highly stereoselective manner, dictated by the bulky substituents on the spiro backbone, such as the 3,5-di-tert-butylphenyl groups. These bulky groups create a well-defined chiral pocket that shields one face of the substrate, forcing the hydrogen addition to occur from the preferred trajectory. This mechanistic feature explains the high enantioselectivity observed across a broad range of substrates, including challenging unfunctionalized ketones and alpha,beta-unsaturated ketones.
Impurity control is another critical aspect where this mechanism offers advantages. The high specificity of the catalyst minimizes the formation of side products such as over-reduced species or racemic byproducts, which are common issues with less selective catalysts. The rigid spiro structure prevents the ligand from adopting non-productive conformations that could lead to background racemic catalysis. Furthermore, the stability of the iridium-ligand complex under hydrogenation conditions ensures that the catalyst remains active throughout the reaction duration, preventing the release of free iridium species that could catalyze non-selective background reactions. For quality control teams, this means a cleaner reaction profile and a simplified purification process, ultimately leading to higher purity specifications for the final chiral alcohol product. The ability to tune the electronic properties of the pyridine ring by introducing electron-withdrawing or electron-donating groups further allows chemists to optimize the reaction rate and selectivity for specific difficult substrates.
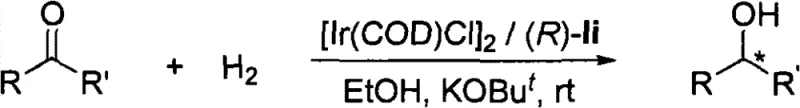
How to Synthesize Chiral Spiro Pyridylamidophosphine Ligands Efficiently
The preparation of these high-value ligands is designed for practicality and scalability, avoiding the need for cryogenic conditions or exotic reagents. The core transformation involves a reductive amination strategy that couples a chiral spiro aminophosphine precursor with various substituted pyridine carbaldehydes. This one-pot procedure is highly efficient, typically proceeding at room temperature in common solvents like dichloroethane or alcohols. The use of mild reducing agents such as sodium triacetoxyborohydride ensures compatibility with sensitive functional groups that might be present on the pyridine ring. Detailed standardized synthesis steps for specific analogues are provided in the guide below, illustrating the robustness of this methodology across different substituent patterns.
- Prepare the reaction vessel under inert atmosphere and dissolve the chiral spiro aminophosphine precursor (Formula II) in a suitable organic solvent such as dichloroethane.
- Add the substituted pyridine carbaldehyde and a reducing agent like sodium triacetoxyborohydride to the mixture.
- Stir the reaction at room temperature for 2 to 24 hours, then quench, extract, and purify via silica gel chromatography to obtain the target ligand (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this ligand technology offers compelling economic and operational benefits that extend beyond mere technical performance. The primary value driver is the drastic reduction in catalyst loading enabled by the high turnover frequency of the iridium complexes. By lowering the catalyst requirement to parts-per-million levels, manufacturers can achieve substantial cost savings on precious metal inputs, which is a significant portion of the variable cost in asymmetric hydrogenation processes. Additionally, the simplified ligand synthesis route, which relies on commercially available starting materials and standard organic transformations, reduces the risk of supply disruptions. This contrasts sharply with complex multistep ligand syntheses that are prone to yield losses and bottlenecks. The robustness of the catalyst also implies longer campaign runs and reduced downtime for reactor cleaning or catalyst recharge, enhancing overall plant throughput.
- Cost Reduction in Manufacturing: The ability to operate at ultra-low catalyst loadings directly impacts the bottom line by minimizing the consumption of expensive iridium and chiral ligands. Since the catalyst is highly active, the total mass of metal introduced into the process is negligible, which subsequently lowers the cost and complexity of metal scavenging and waste treatment. This efficiency allows for a more competitive pricing structure for the final chiral intermediates, providing a strategic advantage in price-sensitive markets. Furthermore, the high yields and selectivity reduce the amount of raw material wasted on byproducts, improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The synthetic route for these ligands is short and utilizes stable, readily accessible reagents, ensuring a consistent and reliable supply of the catalyst system. Unlike proprietary ligand systems that rely on single-source suppliers with long lead times, the chemistry described in the patent allows for flexible manufacturing options. This decentralization of supply risk is critical for maintaining continuous production schedules for key pharmaceutical ingredients. The stability of the ligands themselves also facilitates easier storage and transportation, reducing the logistical burden and potential for degradation during transit.
- Scalability and Environmental Compliance: The process operates under mild conditions, often at room temperature and moderate hydrogen pressures, which simplifies the engineering requirements for scale-up. This ease of scaling from gram to ton quantities ensures that process development timelines are shortened, accelerating time-to-market for new drug candidates. From an environmental standpoint, the high selectivity reduces solvent usage for purification and minimizes hazardous waste generation. The low metal content in the crude product stream simplifies compliance with strict regulatory limits on residual heavy metals in APIs, reducing the need for extensive downstream processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology in industrial settings. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this catalytic system into existing manufacturing workflows.
Q: What is the primary advantage of these spiro ligands over traditional Noyori catalysts?
A: Unlike traditional Noyori ruthenium-bisphosphine/diamine catalysts which require precise matching of chirality and steric effects, these novel spiro pyridylamidophosphine ligands offer greater flexibility and maintain high enantioselectivity (up to 99.9% ee) even with lower catalyst loadings.
Q: Can these ligands be used for the hydrogenation of unfunctionalized ketones?
A: Yes, the patent explicitly demonstrates that iridium complexes formed with these ligands achieve excellent yields and enantioselectivity in the asymmetric hydrogenation of simple aryl alkyl ketones, enones, and ketoesters.
Q: What is the typical catalyst loading required for industrial applications?
A: The technology allows for extremely low catalyst loadings, potentially as low as 0.0001 mol%, which significantly reduces the cost of precious metal usage and simplifies downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Pyridylamidophosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral spiro pyridylamidophosphine ligand technology in advancing asymmetric synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ligand or chiral intermediate meets the highest global standards. We understand that consistency is key in pharmaceutical manufacturing, and our robust quality management systems are designed to deliver that reliability.
We invite you to collaborate with our technical team to explore how this advanced catalytic system can optimize your specific synthesis routes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this low-loading catalyst system for your projects. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your target molecules. Contact our technical procurement team today to discuss how we can support your supply chain with high-performance chiral solutions.
