Revolutionizing Asymmetric Hydrogenation with Novel Chiral Spiro Phosphine-Nitrogen-Phosphine Iridium Catalysts
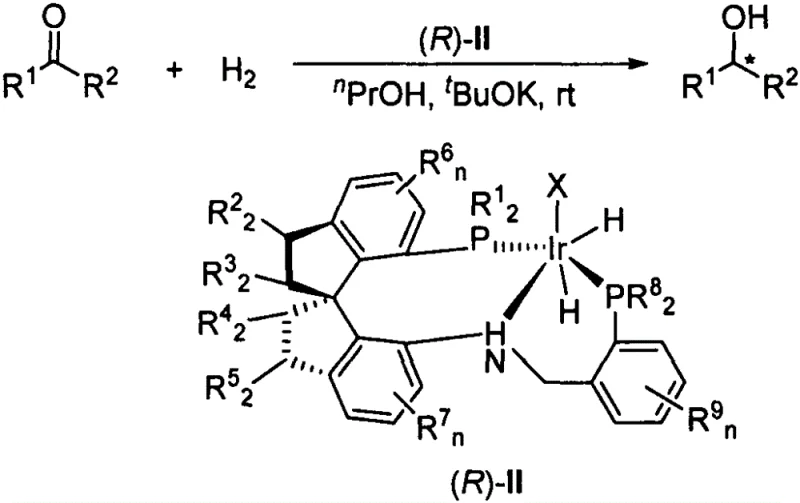
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in Chinese Patent CN111484533B, which introduces a novel class of chiral spiro phosphine-nitrogen-phosphine tridentate ligands, designated as SpiroPNP, and their corresponding iridium complexes. This technology addresses one of the most persistent challenges in organic synthesis: the highly enantioselective hydrogenation of simple dialkyl ketones. Unlike aryl-alkyl ketones, dialkyl ketones possess minimal steric and electronic differentiation between the two alkyl groups flanking the carbonyl, making chiral discrimination exceptionally difficult for conventional catalysts. The invention provides a robust solution by leveraging a unique chiral spiroindane skeleton coupled with sterically demanding phosphine substituents, achieving yields exceeding 99% and enantiomeric excess (ee) values up to 99.8%. For R&D directors and process chemists seeking reliable pharmaceutical intermediates supplier partnerships, this technology represents a critical advancement in accessing difficult-to-synthesize chiral alcohols efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of ketones has been dominated by ruthenium-BINAP type catalysts and various rhodium or iridium systems utilizing bidentate phosphine ligands. While these systems excel with functionalized ketones, such as beta-keto esters or aryl-alkyl ketones, they often fail when confronted with simple dialkyl ketones like methyl ethyl ketone. The fundamental limitation lies in the catalyst's inability to distinguish between the small steric difference of a methyl group versus an ethyl or propyl group. Traditional bidentate ligands often lack the necessary three-dimensional rigidity and the specific steric environment required to force the substrate into a single productive orientation. Consequently, reactions with these challenging substrates typically result in low enantioselectivity, necessitating costly and wasteful downstream purification steps like chiral resolution or recrystallization, which drastically impact the overall process mass intensity and cost reduction in fine chemical manufacturing.
The Novel Approach
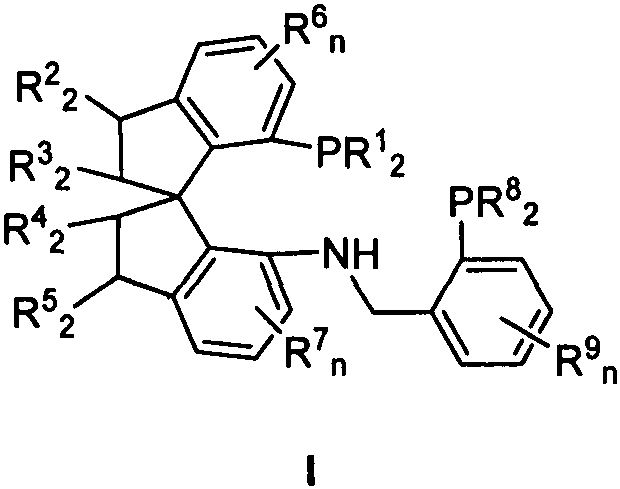
The innovation described in CN111484533B overcomes these barriers through the design of a tridentate P-N-P ligand system anchored on a chiral spiro[4.4]nonane or spiroindane framework. By introducing a nitrogen donor atom alongside two phosphine donors, the ligand forms a more rigid and defined coordination geometry around the iridium center. Crucially, the phosphine arms are substituted with bulky groups such as di-tert-butylphenyl (DTB) or di-tert-butylmethoxyphenyl (DTBM). These massive substituents create a highly congested chiral pocket that effectively amplifies the minute steric differences between the alkyl groups of the ketone substrate. This structural design allows the catalyst to act almost enzymatically, locking the substrate in a specific conformation that favors the formation of one enantiomer over the other. This approach not only solves the scientific challenge of dialkyl ketone hydrogenation but also offers a practical pathway for the commercial scale-up of complex pharmaceutical intermediates that were previously inaccessible via direct asymmetric hydrogenation.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The exceptional performance of the Ir-SpiroPNP catalyst stems from the synergistic interplay between the rigid spiro backbone and the flexible yet bulky phosphine arms. Mechanistically, the tridentate coordination ensures that the iridium center remains stable under hydrogenation conditions, preventing ligand dissociation which often leads to background racemic catalysis. The spiro-indane scaffold imposes a fixed chirality that is transmitted directly to the metal center. When the ketone substrate approaches the active site, the bulky tert-butyl groups on the phosphine ligands exert strong repulsive forces. In the case of a dialkyl ketone, the catalyst forces the smaller alkyl group (e.g., methyl) and the larger alkyl group (e.g., ethyl) into distinct spatial quadrants defined by the ligand architecture. This precise positioning lowers the activation energy for hydride transfer to one face of the carbonyl plane while raising it for the other. Furthermore, the presence of the amine linker (-NH-CH2-) provides a potential secondary interaction site, possibly through hydrogen bonding or hemilabile coordination, which further stabilizes the transition state. This level of mechanistic control is essential for maintaining high purity specifications in the production of active pharmaceutical ingredients.
Impurity control is another critical aspect where this catalyst system excels. In traditional hydrogenation processes, over-reduction or isomerization of double bonds can occur as side reactions. However, the specific electronic properties of the SpiroPNP ligand tune the electrophilicity of the iridium-hydride species, making it highly chemoselective for the carbonyl group. The patent data indicates that even in the presence of other reducible functionalities, the catalyst maintains high selectivity for the ketone. This chemoselectivity minimizes the formation of by-products, simplifying the workup procedure and reducing the burden on quality control laboratories. For supply chain heads, this translates to more predictable batch outcomes and reduced risk of batch failure due to impurity profiles exceeding regulatory limits. The ability to achieve turnover numbers (TON) as high as 4300 further underscores the efficiency of the catalytic cycle, ensuring that the precious iridium metal is utilized to its maximum potential before deactivation.
How to Synthesize SpiroPNP Ligands Efficiently
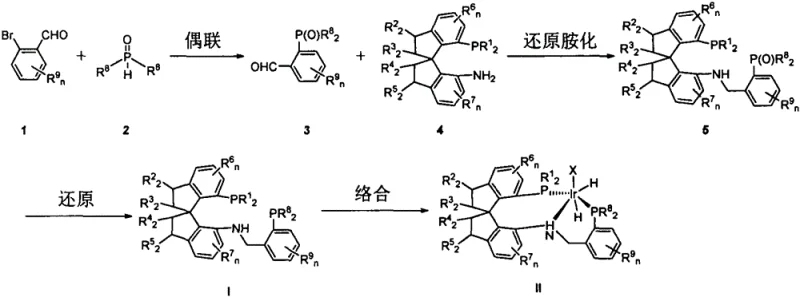
The preparation of these high-performance ligands follows a logical and scalable synthetic sequence that avoids exotic reagents or extreme conditions. The process begins with the construction of the phosphine oxide precursor via palladium-catalyzed cross-coupling, followed by a reductive amination step to install the chiral spiroindane amine moiety. A final reduction step converts the phosphine oxide to the active phosphine, which is then complexed with an iridium precursor. This modular approach allows for the easy variation of the phosphine substituents to fine-tune the catalyst for specific substrates. The detailed standardized synthetic steps for producing these ligands and the subsequent iridium complexes are outlined in the guide below.
- Perform palladium-catalyzed coupling of bromo-aldehydes with secondary phosphine oxides to generate phosphine oxide intermediates.
- Conduct reductive amination between the phosphine oxide intermediate and chiral spiroindane amines using sodium cyanoborohydride.
- Reduce the phosphine oxide moiety to phosphine using silane reagents, followed by complexation with iridium precursors like [Ir(COD)Cl]2 under hydrogen pressure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the SpiroPNP iridium catalyst technology offers substantial strategic advantages beyond mere technical performance. The primary value driver is the drastic simplification of the synthesis of chiral alcohols, which are ubiquitous building blocks in drug discovery and development. By enabling direct asymmetric hydrogenation of simple ketones, this technology eliminates the need for multi-step sequences involving stoichiometric chiral auxiliaries or resolution agents. This reduction in step count inherently lowers the consumption of raw materials, solvents, and energy, leading to significant cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the high enantioselectivity reduces the reliance on expensive chiral chromatography for purification, a process that is often a bottleneck in large-scale production due to low throughput and high solvent usage.
- Cost Reduction in Manufacturing: The high catalytic activity and turnover numbers associated with Ir-SpiroPNP complexes mean that significantly lower catalyst loadings are required to achieve complete conversion. Since iridium is a precious metal with volatile pricing, minimizing the loading directly impacts the bill of materials. Additionally, the mild reaction conditions (often room temperature to moderate heating and moderate hydrogen pressure) reduce energy consumption and allow for the use of standard glass-lined or stainless-steel reactors without the need for specialized high-pressure equipment. The elimination of resolution steps also recovers yield that would otherwise be lost as the unwanted enantiomer, effectively doubling the theoretical output from the same amount of starting material.
- Enhanced Supply Chain Reliability: The synthetic route for the ligands relies on robust chemical transformations such as reductive amination and silane reduction, which are well-understood and easily scaled in multipurpose chemical plants. The starting materials, including spiroindane amines and substituted phosphine oxides, are becoming increasingly accessible as the technology matures. This accessibility mitigates the risk of supply disruptions associated with niche, single-source reagents. Moreover, the stability of the catalyst under reaction conditions ensures consistent batch-to-batch performance, allowing for tighter production scheduling and reduced lead time for high-purity chiral intermediates. This reliability is crucial for maintaining continuous supply to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this catalytic method aligns well with green chemistry principles. The atom economy of hydrogenation is 100%, as hydrogen is the only reagent added to the substrate. The high selectivity minimizes waste generation, reducing the volume of hazardous waste requiring disposal. The ability to operate at lower pressures and temperatures also enhances process safety, lowering the risk profile of the manufacturing operation. As regulatory scrutiny on solvent emissions and waste disposal intensifies globally, adopting such efficient catalytic technologies positions companies favorably for long-term compliance and sustainability goals, facilitating smoother regulatory approvals for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology in industrial settings. These answers are derived from the experimental data and scope defined within the patent documentation, providing a realistic view of the technology's capabilities and limitations for potential adopters.
Q: What is the primary advantage of SpiroPNP ligands over conventional diphosphines?
A: SpiroPNP ligands feature a rigid chiral spiroindane backbone combined with bulky phosphine substituents, enabling superior chiral recognition for challenging substrates like simple dialkyl ketones where steric differences are minimal.
Q: What enantioselectivity can be achieved with Ir-SpiroPNP catalysts?
A: The patented Ir-SpiroPNP catalysts demonstrate exceptional enantioselectivity, reaching up to 99.8% ee in the asymmetric hydrogenation of simple dialkyl ketones, significantly outperforming many traditional systems.
Q: Are these catalysts suitable for large-scale industrial production?
A: Yes, the synthesis utilizes robust reaction conditions and commercially available starting materials, and the high turnover numbers (TON) reduce the required catalyst loading, making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced asymmetric catalysis in accelerating drug development timelines. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs like the SpiroPNP ligand system can be seamlessly translated into robust manufacturing processes. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of handling sensitive organometallic catalysts and verifying enantiomeric excess with high precision. We understand that the successful deployment of such catalysts requires not just the material, but the process expertise to handle air-sensitive compounds and optimize reaction parameters for maximum efficiency.
We invite you to collaborate with our technical team to evaluate the feasibility of this hydrogenation technology for your specific pipeline projects. Whether you require custom synthesis of the ligand, the pre-formed iridium catalyst, or full process development services, we are prepared to support your needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current synthetic routes. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about integrating this high-performance catalytic system into your supply chain.
