Scalable Synthesis of Novel Optically Pure Trost Ligands for Advanced Asymmetric Catalysis
Scalable Synthesis of Novel Optically Pure Trost Ligands for Advanced Asymmetric Catalysis
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN113135955B, which discloses a novel preparation method for optically pure Trost ligands. These ligands, characterized by their C2 symmetry and robust N,P-coordination capability, are pivotal for achieving high enantioselectivity in metal-catalyzed transformations. The invention addresses a critical gap in the prior art by providing a versatile synthetic strategy that not only ensures high optical purity but also expands the structural diversity of substituents on the phosphorus atom. This advancement is particularly relevant for R&D teams seeking reliable pharmaceutical intermediate suppliers who can deliver complex chiral building blocks with consistent quality. By leveraging a sequence of asymmetric addition, one-pot reduction-boration, and condensation reactions, this technology offers a streamlined pathway to access previously difficult-to-synthesize ligand architectures.
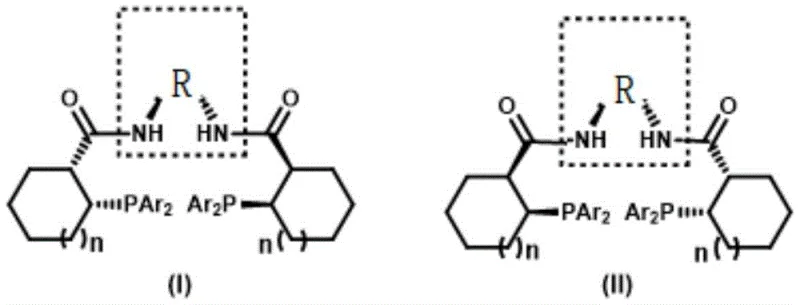
The limitations of conventional methods for synthesizing Trost ligands have long hindered the rapid development of new catalytic systems. Traditional approaches often rely on rigid synthetic routes that restrict the diversity of groups attached to the phosphorus centers, thereby limiting the tunability of the resulting catalysts for specific substrate interactions. Furthermore, existing methods frequently suffer from moderate yields and require harsh conditions that can compromise the integrity of sensitive functional groups. In contrast, the novel approach detailed in the patent utilizes a highly efficient addition reaction between methyl cycloalkenyl carboxylate and diarylphosphine oxides. This strategy allows for the introduction of various aryl groups, such as phenyl, p-tolyl, or naphthyl moieties, directly onto the phosphorus atom with excellent stereocontrol. The ability to modulate the steric and electronic properties of the ligand through simple changes in the starting phosphine oxide represents a paradigm shift, enabling the design of bespoke catalysts tailored for challenging asymmetric transformations in fine chemical manufacturing.
Mechanistic Insights into Bu2Mg/BINOL Catalyzed Asymmetric Addition
The core of this synthetic innovation lies in the initial asymmetric addition step, which sets the stereochemical foundation for the entire molecule. The reaction employs a catalytic system comprising dibutylmagnesium (Bu2Mg) and chiral H8-BINOL in tetrahydrofuran at low temperatures (-20°C). This specific combination generates a chiral magnesium species that activates the diarylphosphine oxide, facilitating its nucleophilic attack on the cycloalkenyl ester with high facial selectivity. The mechanistic precision of this step is evidenced by the achievement of up to 95% ee and favorable diastereomeric ratios (dr 6.3:1) in the first intermediate. Following this, the process incorporates a sophisticated one-pot reduction and boronation sequence. By utilizing trichlorosilane and triethylenediamine followed by borane-THF complex, the ester functionality is efficiently converted into a boron-containing intermediate without isolating the alcohol. This tandem operation not only reduces solvent consumption and processing time but also minimizes the risk of racemization, ensuring that the optical purity established in the first step is preserved throughout the synthesis.
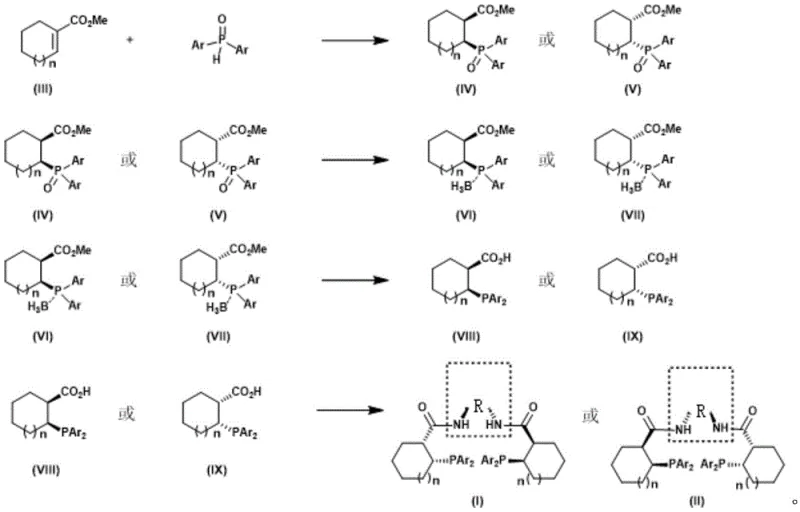
Impurity control is another critical aspect where this methodology excels, particularly for applications requiring high-purity OLED material precursors or API intermediates. The hydrolysis step, mediated by aluminum trichloride in dimethyl sulfide, cleanly converts the boron intermediate into the corresponding carboxylic acid without generating significant side products. The final condensation with chiral diamines, facilitated by EDC and DMAP, proceeds under mild conditions that prevent epimerization of the sensitive alpha-chiral centers. The rigorous purification protocols described, including specific eluent systems for column chromatography (e.g., n-pentane/ether/acetone), ensure that trace metal residues and organic impurities are reduced to negligible levels. This level of purity is essential for downstream catalytic applications where trace contaminants could poison the active metal center or alter the reaction pathway, thus guaranteeing the reliability of the ligand in commercial-scale asymmetric synthesis operations.
How to Synthesize Novel Optically Pure Trost Ligand Efficiently
The synthesis of these advanced ligands follows a logical four-step progression designed for maximum efficiency and scalability. The process begins with the activation of the phosphine oxide and subsequent addition to the cyclic ester, followed by a streamlined reduction-boration sequence that avoids unnecessary isolation steps. The third stage involves a controlled hydrolysis to reveal the carboxylic acid handle, which is then coupled with the chiral diamine backbone in the final step. Each stage has been optimized for solvent compatibility and reagent stoichiometry to maximize throughput. For detailed operational parameters, temperature controls, and specific workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform asymmetric addition of methyl cycloalkenyl carboxylate and diarylphosphine oxide using Bu2Mg/(R or S)-H8-BINOL catalyst at -20°C.
- Execute one-pot reduction using trichlorosilane/triethylenediamine followed by boronation with borane-THF complex to form the second intermediate.
- Hydrolyze the second intermediate using aluminum trichloride in dimethyl sulfide to obtain the carboxylic acid third intermediate.
- Condense the third intermediate with a chiral diamine using EDC/HOBt coupling agents to yield the final C2 symmetric Trost ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial advantages over legacy ligand synthesis routes. The primary benefit stems from the use of readily available and cost-effective starting materials, such as methyl cycloalkenyl carboxylates and common diarylphosphine oxides, which eliminates the dependency on scarce or prohibitively expensive chiral pool resources. This accessibility translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the raw material costs are significantly lower compared to routes requiring complex natural product derivatives. Furthermore, the synthetic route avoids the use of precious transition metal catalysts in the bond-forming steps, relying instead on abundant main-group elements like magnesium and boron. This not only lowers the direct material cost but also simplifies the downstream purification process by removing the need for expensive metal scavenging resins, thereby reducing the overall cost of goods sold (COGS) for the final ligand product.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of a one-pot reduction-boration strategy significantly streamline the production workflow. By combining two reaction steps into a single vessel operation, the process reduces solvent usage, energy consumption for heating and cooling cycles, and labor hours associated with intermediate isolation. This operational efficiency leads to substantial cost savings, making the commercial production of these high-value ligands more economically viable for large-scale applications without compromising on the quality or optical purity of the final product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as trichlorosilane, toluene, and dichloromethane ensures a robust and resilient supply chain. Unlike specialized reagents that may face geopolitical or logistical bottlenecks, these materials are widely produced and available from multiple global vendors. This diversification of the supply base mitigates the risk of production delays caused by raw material shortages. Additionally, the robustness of the reaction conditions, which tolerate standard industrial equipment and do not require extreme pressures or cryogenic temperatures beyond standard chilling, facilitates consistent batch-to-batch production, ensuring reducing lead time for high-purity chiral ligands delivery to customers.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been designed with potential commercial scale-up of complex polymer additives and fine chemicals in mind. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through standard distillation and wastewater treatment protocols common in modern chemical facilities. The avoidance of heavy metals reduces the environmental burden and regulatory compliance costs associated with hazardous waste disposal. This alignment with green chemistry principles not only supports corporate sustainability goals but also future-proofs the manufacturing process against tightening environmental regulations, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel Trost ligands. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating these ligands into their existing catalytic processes or for procurement specialists assessing the long-term viability of the supply source.
Q: What distinguishes this novel Trost ligand synthesis from conventional methods?
A: Unlike conventional strategies that offer limited diversity on the phosphorus atom, this patented method utilizes a unique cycloalkenyl backbone and allows for extensive modification of aryl substituents, significantly expanding the library of available C2 symmetric ligands for asymmetric catalysis.
Q: How does the new process improve optical purity and yield?
A: The process employs a highly stereoselective Bu2Mg/BINOL catalytic system for the initial addition step, achieving high ee values (up to 95%). Subsequent one-pot reduction and boronation steps minimize intermediate isolation losses, ensuring high overall purity and yield suitable for industrial applications.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the route relies on robust reagents like trichlorosilane and standard coupling agents (EDC/DMAP) rather than exotic precious metal catalysts. The use of common solvents like toluene and dichloromethane, combined with straightforward workup procedures like crystallization and column chromatography, facilitates easy scale-up from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trost Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation therapeutics and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Trost ligand meets the exacting standards required for sensitive asymmetric catalysis applications. Our infrastructure is designed to handle the specific solvent and reagent requirements of this patented process, ensuring consistent quality and availability for our global partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to this novel ligand synthesis route can optimize your overall production budget. We encourage you to contact our technical procurement team to request specific COA data for our current inventory or to discuss route feasibility assessments for custom derivatives. By partnering with us, you gain access to a reliable supply chain capable of supporting your most demanding chiral synthesis challenges with speed, precision, and economic efficiency.
