Revolutionizing C-S Bond Construction: Scalable Synthesis of Arylthionaphthols and Indole Derivatives
Revolutionizing C-S Bond Construction: Scalable Synthesis of Arylthionaphthols and Indole Derivatives
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective methods for constructing carbon-heteroatom bonds, particularly carbon-sulfur (C-S) linkages which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN113816884B, which discloses a novel synthetic method for arylthionaphthols, naphthylamine compounds, and phenylthioindole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed cross-coupling reactions to a greener, metal-free aerobic oxidation protocol. By utilizing atmospheric oxygen as the sole oxidant and operating under basic conditions without the need for precious metal catalysts, this invention addresses critical pain points regarding cost, environmental safety, and product purity. For R&D directors and procurement managers alike, understanding the implications of this patent is vital for optimizing supply chains and reducing the cost of goods sold (COGS) for complex sulfur-containing intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-S bonds has relied heavily on transition-metal catalyzed cross-coupling reactions, such as those involving palladium or copper catalysts reacting aryl halides with thiols or disulfides. While effective, these conventional methodologies suffer from several inherent drawbacks that hinder large-scale commercial viability. Firstly, the reliance on expensive transition metal catalysts significantly inflates raw material costs, creating volatility in pricing tied to the global markets for precious metals. Secondly, and perhaps more critically for pharmaceutical manufacturing, is the issue of residual metal contamination. Stringent regulatory limits on heavy metals in active pharmaceutical ingredients (APIs) necessitate complex and costly purification steps, often involving specialized scavengers or repeated recrystallizations, which inevitably lower overall process yield. Furthermore, these reactions typically require stoichiometric amounts of chemical oxidants or inert atmospheres, generating substantial chemical waste and increasing the burden on environmental health and safety (EHS) protocols. The cumulative effect is a manufacturing process that is economically inefficient and environmentally burdensome.
The Novel Approach
In stark contrast, the methodology presented in patent CN113816884B offers a streamlined, high-efficiency alternative that circumvents the limitations of metal catalysis. This novel approach utilizes a direct oxidative coupling between naphthylamines, naphthols, or indoles and thiophenols under alkaline conditions, driven simply by heating in an air environment. 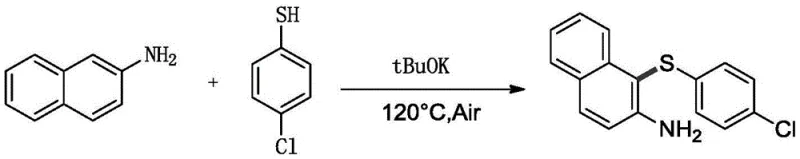 As illustrated in the reaction scheme above, the process eliminates the need for aryl halide precursors and transition metal catalysts entirely. Instead, it leverages the intrinsic reactivity of the substrates in the presence of a base like potassium tert-butoxide (tBuOK) and molecular oxygen. This results in a highly selective synthesis of monosulfide products with minimal byproduct formation—essentially water. The operational simplicity is remarkable; reactions can be conducted solvent-free or with minimal solvent, at temperatures ranging from 60°C to 150°C, drastically simplifying the work-up procedure to basic extraction and filtration. This transition from complex catalytic cycles to a direct oxidative coupling represents a significant leap forward in process chemistry, offering a robust pathway for the reliable arylthionaphthol supplier market.
As illustrated in the reaction scheme above, the process eliminates the need for aryl halide precursors and transition metal catalysts entirely. Instead, it leverages the intrinsic reactivity of the substrates in the presence of a base like potassium tert-butoxide (tBuOK) and molecular oxygen. This results in a highly selective synthesis of monosulfide products with minimal byproduct formation—essentially water. The operational simplicity is remarkable; reactions can be conducted solvent-free or with minimal solvent, at temperatures ranging from 60°C to 150°C, drastically simplifying the work-up procedure to basic extraction and filtration. This transition from complex catalytic cycles to a direct oxidative coupling represents a significant leap forward in process chemistry, offering a robust pathway for the reliable arylthionaphthol supplier market.
Mechanistic Insights into Metal-Free Aerobic Oxidative Coupling
To fully appreciate the technical elegance of this invention, one must delve into the mechanistic underpinnings that enable such high efficiency without metal mediation. The reaction proceeds through a radical-mediated pathway initiated by the base and molecular oxygen. 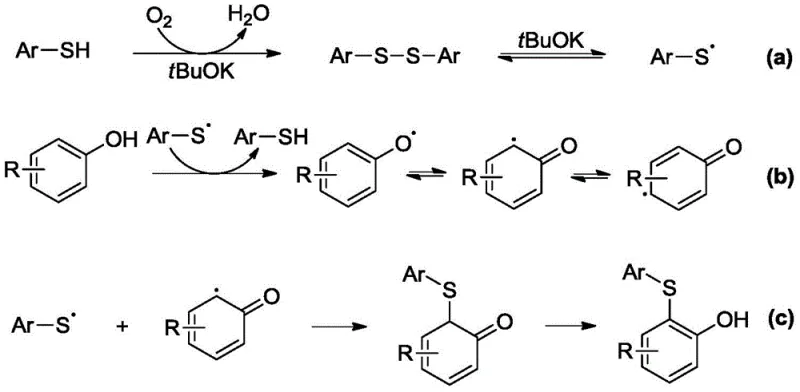 As depicted in the mechanistic diagram, the process begins with the deprotonation of the thiophenol by the base to form a thiolate anion. Under the influence of atmospheric oxygen, this thiolate is oxidized to generate a thiyl radical (Ar-S•), with water being the only stoichiometric byproduct. Concurrently, the electron-rich aromatic substrate (such as a naphthol or naphthylamine) undergoes single-electron oxidation to form a resonance-stabilized radical intermediate. The key bond-forming step involves the coupling of the thiyl radical with the carbon-centered radical on the aromatic ring. This radical-radical coupling is highly exothermic and drives the reaction forward. Subsequent tautomerization and rearomatization yield the final stable aryl thioether product. This mechanism explains the high regioselectivity observed, as the radical stability dictates the position of substitution, typically favoring the positions ortho or para to the activating hydroxyl or amino groups.
As depicted in the mechanistic diagram, the process begins with the deprotonation of the thiophenol by the base to form a thiolate anion. Under the influence of atmospheric oxygen, this thiolate is oxidized to generate a thiyl radical (Ar-S•), with water being the only stoichiometric byproduct. Concurrently, the electron-rich aromatic substrate (such as a naphthol or naphthylamine) undergoes single-electron oxidation to form a resonance-stabilized radical intermediate. The key bond-forming step involves the coupling of the thiyl radical with the carbon-centered radical on the aromatic ring. This radical-radical coupling is highly exothermic and drives the reaction forward. Subsequent tautomerization and rearomatization yield the final stable aryl thioether product. This mechanism explains the high regioselectivity observed, as the radical stability dictates the position of substitution, typically favoring the positions ortho or para to the activating hydroxyl or amino groups.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways. Because the reaction avoids strong nucleophilic substitutions that can lead to polysubstitution or ether formation, the impurity profile is remarkably clean. The primary side reaction is the formation of disulfides from the thiophenol, but the patent data indicates that the coupling with the aromatic substrate is kinetically favored under the optimized conditions. Furthermore, the absence of metal catalysts means there are no metal-ligand complexes or metal-induced decomposition products to worry about during storage or downstream processing. This purity profile is crucial for R&D teams aiming to accelerate IND-enabling studies, as it reduces the analytical burden required to characterize and qualify impurities. The ability to achieve high purity (>98% in many examples) directly from column chromatography without extensive recrystallization underscores the robustness of this chemical transformation.
How to Synthesize Arylthionaphthols Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and safety. The general protocol involves mixing the aromatic substrate (first reactant) and the thiophenol derivative (second reactant) with a suitable base, such as potassium tert-butoxide, potassium carbonate, or DBU. The molar ratio is typically optimized around 1:1.5 to 1:3 for the reactants, with a catalytic to stoichiometric amount of base. The mixture is then heated, typically between 100°C and 150°C, under an open air atmosphere or with oxygen sparging to ensure sufficient oxidant supply. The reaction progress is conveniently monitored by thin-layer chromatography (TLC), and upon completion, the crude product is isolated via simple aqueous workup and organic extraction. This straightforward procedure makes the technology accessible for rapid process development and scale-up.
- Mix the first reactant (naphthylamine, naphthol, or indole derivative) with the second reactant (thiophenol derivative) and a base such as potassium tert-butoxide in a dry reaction vessel.
- Heat the mixture to a temperature between 60°C and 150°C under an air or oxygen atmosphere while stirring continuously to facilitate the radical oxidation process.
- Monitor reaction progress via TLC, then perform extraction with ethyl acetate and water, followed by drying and column chromatography purification to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that go beyond mere technical novelty. The shift away from transition metal catalysis fundamentally alters the cost structure and risk profile of manufacturing sulfur-containing intermediates. By eliminating the need for palladium or copper catalysts, companies can avoid the price volatility associated with these commodities and remove the costly step of metal scavenging from the downstream processing train. This simplification of the purification workflow not only reduces the consumption of auxiliary materials like silica gel and solvents but also shortens the overall production cycle time. Consequently, this leads to substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in the final API market.
- Cost Reduction in Manufacturing: The economic impact of this metal-free protocol is profound. Traditional methods incur hidden costs related to catalyst recovery, metal testing, and waste disposal of heavy metal residues. By utilizing air as the oxidant and inexpensive inorganic or organic bases, the raw material cost is drastically reduced. Moreover, the solvent-free or low-solvent nature of the reaction minimizes the volume of waste solvent that needs to be treated or incinerated, further lowering operational expenditures. The high atom economy of the reaction, where the only byproduct is water, ensures that the majority of the input mass ends up in the desired product, maximizing resource efficiency and driving down the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commodity-grade reagents. Thiophenols and naphthols are bulk chemicals with established supply chains, unlike specialized ligands or pre-catalysts which may have single-source suppliers or long lead times. The robustness of the reaction conditions—tolerating air and moisture to a degree—reduces the risk of batch failures due to minor deviations in process control. This reliability ensures consistent delivery schedules, which is critical for maintaining continuous API production lines. Additionally, the simplified logistics of handling non-hazardous oxidants (air) versus compressed oxygen cylinders or hazardous chemical oxidants reduces transportation and storage risks.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this technology has demonstrated excellent scalability. The patent data confirms that yields remain high even when scaling from millimole to gram quantities, suggesting that heat and mass transfer issues are manageable in larger reactors. From an environmental standpoint, the process aligns perfectly with green chemistry principles. The elimination of heavy metals simplifies wastewater treatment and ensures compliance with increasingly strict environmental regulations regarding metal discharge. This 'green' credential is not just a regulatory checkbox but a market differentiator, appealing to end-clients who prioritize sustainability in their supply chain audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aerobic oxidation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free C-S bond formation method?
A: The primary advantages include the elimination of expensive transition metal catalysts, the use of air as a green oxidant producing only water as a byproduct, and the ability to operate under solvent-free or minimal solvent conditions, significantly reducing environmental impact and purification costs.
Q: Can this synthesis method be scaled up for industrial production?
A: Yes, the patent data explicitly demonstrates successful scale-up from millimole scales to gram-level reactions (e.g., 5mmol scale) with maintained or improved yields (up to 97%), indicating robust scalability for commercial manufacturing.
Q: What types of substrates are compatible with this aerobic oxidation protocol?
A: The method exhibits broad substrate scope, successfully reacting various naphthylamines, naphthols, and indole derivatives with diverse thiophenol derivatives, including those with electron-withdrawing groups like chlorine and nitro groups, as well as heterocyclic thiols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylthionaphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods disclosed in CN113816884B for the production of high-value sulfur-containing intermediates. As a leading CDMO partner, we possess the technical expertise to adapt and optimize these green chemistry protocols for your specific project needs. Our facilities are equipped to handle diverse synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that moving from a patent example to a commercial process requires rigorous optimization, and our team is dedicated to ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities.
We invite you to collaborate with us to leverage this innovative technology for your next project. Whether you require custom synthesis of complex arylthionaphthols or scale-up of phenylthioindole derivatives, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can enhance your supply chain efficiency and reduce your overall manufacturing costs.
