Advanced Synthesis of 3,4,5-Trimethoxytoluene for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 3,4,5-Trimethoxytoluene for Scalable Pharmaceutical Manufacturing
The global demand for high-purity pharmaceutical intermediates continues to surge, driven primarily by the expanding market for coenzyme Q10 and idebenone therapies. In response to the limitations of traditional extraction and synthesis methods, patent CN115368217A introduces a groundbreaking synthesis method for 3,4,5-trimethoxytoluene, a critical precursor in the production of these vital bioactive compounds. This innovative approach addresses the urgent need for a reliable pharmaceutical intermediate supplier capable of delivering consistent quality while adhering to stringent environmental standards. By utilizing 2,6-dibromo-4-methylphenol as a starting material and employing a clean gas-phase methylation strategy, this technology represents a significant leap forward in process chemistry. The structural integrity of the target molecule, as depicted below, is maintained through precise control of reaction conditions, ensuring minimal byproduct formation.
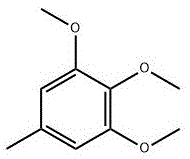
Furthermore, the patent outlines a robust pathway that not only enhances yield but also drastically simplifies the downstream processing requirements. For R&D directors and procurement managers alike, understanding the nuances of this synthesis is crucial for securing a stable supply chain for coenzyme Q10 raw materials. The shift away from biological extraction, which is often plagued by low quantities and high prices, towards this efficient chemical synthesis ensures long-term availability. Consequently, this technology positions manufacturers to meet the escalating market demands without compromising on cost-efficiency or ecological responsibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4,5-trimethoxytoluene has been fraught with significant environmental and operational challenges that hinder large-scale adoption. Traditional methods, such as those disclosed in Chinese patent CN1762949A, rely heavily on dimethyl sulfate as a methylating agent, a substance known for its high toxicity and the generation of substantial hazardous waste. Specifically, these legacy processes produce large volumes of high-salt wastewater containing sodium methyl sulfate, which necessitates complex and expensive treatment protocols before discharge. Additionally, alternative routes involving modified skeleton nickel catalysts, as seen in CN101531574A, suffer from prolonged reaction times and the prohibitive cost of noble metal catalysts. These factors collectively inflate the production cost and create bottlenecks in the supply chain, making it difficult to achieve the economies of scale required for mass production. Moreover, the use of bio-oil derived raw materials often introduces variability in feedstock quality, leading to inconsistent batch results and further complicating quality control measures.
The Novel Approach
In stark contrast to these outdated methodologies, the novel approach detailed in patent CN115368217A offers a streamlined, environmentally benign solution that redefines efficiency in intermediate manufacturing. This method employs a two-step sequence involving methylation followed by methoxylation, utilizing clean methyl chloride or methyl bromide gas instead of liquid alkylating agents. As illustrated in the reaction scheme below, this strategic shift eliminates the formation of sodium methyl sulfate wastewater entirely, thereby removing a major cost center associated with waste disposal. The process is designed as a one-pot operation where both methylation and subsequent methoxylation can be conducted in the same equipment, significantly reducing capital expenditure on reactor infrastructure. By optimizing reaction parameters such as temperature and pressure, the method ensures high conversion rates while maintaining mild operating conditions that are safer for personnel and equipment. This holistic improvement in process design makes it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
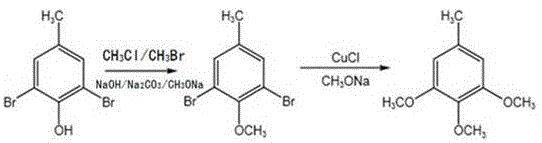
Mechanistic Insights into Copper-Catalyzed Methoxylation
The core of this synthetic breakthrough lies in the sophisticated application of copper-catalyzed nucleophilic aromatic substitution, a mechanism that allows for the precise installation of methoxy groups onto the aromatic ring. In the second stage of the synthesis, cuprous chloride acts as a potent catalyst, facilitating the displacement of bromine atoms on the 2,6-dibromo-4-methylanisole intermediate by methoxide ions. This reaction proceeds under elevated pressure (1.5-2.3 MPa) and temperature (110°C), conditions that are carefully calibrated to maximize the reaction kinetics without promoting thermal degradation of the sensitive aromatic core. The presence of the copper catalyst lowers the activation energy required for the substitution, enabling the reaction to proceed to completion within a relatively short timeframe of three hours. This mechanistic efficiency is critical for maintaining high throughput in a commercial setting, as it minimizes the residence time of materials in the reactor and maximizes equipment utilization rates.
From an impurity control perspective, the choice of reagents and the specific sequence of addition play a pivotal role in ensuring the final product meets rigorous purity specifications. The initial methylation step uses gaseous methylating agents which react cleanly with the phenolic hydroxyl group, avoiding the side reactions common with liquid esters. Furthermore, the use of sodium methoxide in the methoxylation step ensures that the resulting sodium bromide byproduct is easily separable via filtration, preventing salt contamination in the final organic phase. The patent data indicates that this meticulous control over the reaction environment yields a product with purity levels ranging from 98.6% to 99.4%, a testament to the selectivity of the catalytic system. For quality assurance teams, this implies a simplified purification workflow, as the need for extensive chromatographic separation is largely negated by the inherent cleanliness of the reaction pathway.
How to Synthesize 3,4,5-Trimethoxytoluene Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of specific thermodynamic conditions to ensure optimal yield and safety. The process begins with the methylation of 2,6-dibromo-4-methylphenol, where the choice of acid-binding agent—whether sodium hydroxide, sodium carbonate, or sodium methoxide—must be matched with the appropriate solvent system to facilitate efficient gas absorption. Following the initial alkylation, the reaction mixture is directly subjected to methoxylation conditions without the need for intermediate isolation, showcasing the power of the one-pot design. Detailed standard operating procedures regarding stoichiometry, pressure ramping, and distillation parameters are essential for replicating the high yields reported in the patent examples. The following guide outlines the critical operational phases necessary to achieve commercial-grade results.
- Perform methylation of 2,6-dibromo-4-methylphenol using methyl chloride or bromide gas at 130°C with an acid-binding agent.
- Conduct methoxylation by adding cuprous chloride catalyst and sodium methoxide solution, reacting at 110°C under pressure.
- Filter off catalyst and salts, then distill under reduced pressure to isolate the final 3,4,5-trimethoxytoluene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method translates into tangible strategic advantages that extend far beyond simple unit cost metrics. By fundamentally altering the chemical inputs and process flow, the technology addresses several chronic pain points associated with the sourcing of complex aromatic intermediates. The elimination of hazardous liquid methylating agents not only improves workplace safety but also simplifies logistics and storage requirements, as gaseous reagents can be managed more efficiently in bulk quantities. Furthermore, the reduction in wastewater generation means that facilities can operate with lower environmental compliance overheads, freeing up capital for other strategic investments. This alignment of technical innovation with operational efficiency creates a resilient supply chain capable of withstanding market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of dimethyl sulfate, a reagent that traditionally incurs high costs for both purchase and hazardous waste disposal. By switching to methyl chloride or bromide gas, the process eliminates the generation of sodium methyl sulfate wastewater, effectively removing the need for expensive effluent treatment systems. Additionally, the one-pot nature of the reaction reduces the requirement for multiple reactors and transfer lines, leading to substantial savings in equipment investment and maintenance. The high yield reported, exceeding 96%, ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: Securing a stable supply of 3,4,5-trimethoxytoluene is critical for downstream manufacturers of coenzyme Q10, and this method significantly de-risks the supply chain. The raw materials, specifically 2,6-dibromo-4-methylphenol and simple alkyl halides, are commodity chemicals with robust global availability, reducing the risk of shortages associated with specialized bio-extracts. The simplicity of the process also means that production can be scaled up rapidly across multiple sites without the need for highly specialized or scarce catalytic materials like modified nickel. This flexibility allows suppliers to respond quickly to spikes in demand, ensuring continuous availability for pharmaceutical clients who cannot afford production stoppages.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without significant re-engineering. The mild reaction conditions and the use of standard stainless-steel equipment make it accessible for a wide range of contract development and manufacturing organizations (CDMOs). From an environmental standpoint, the process aligns perfectly with green chemistry principles by minimizing waste at the source rather than treating it post-generation. This proactive approach to sustainability not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the end-product as being sustainably sourced.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for stakeholders evaluating the transition from legacy methods to this advanced catalytic process. The answers reflect the consensus on how this innovation resolves historical inefficiencies in the production of trimethoxytoluene derivatives.
Q: Why is this synthesis method preferred over traditional dimethyl sulfate routes?
A: This method avoids dimethyl sulfate, eliminating the generation of high-salt wastewater containing sodium methyl sulfate, thereby significantly reducing environmental treatment costs and improving safety.
Q: What purity levels can be achieved with this patented process?
A: The patented process consistently achieves product purity between 98.6% and 99.4%, with yields ranging from 96.0% to 97.7%, making it highly suitable for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes a one-pot approach that reduces equipment investment and uses easily available raw materials, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trimethoxytoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving therapeutics like Coenzyme Q10 depends on the reliability and quality of the underlying chemical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3,4,5-trimethoxytoluene meets the highest industry standards. By leveraging advanced synthesis technologies such as the one described in CN115368217A, we deliver intermediates that empower your R&D and manufacturing teams to focus on innovation rather than supply constraints.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to review specific COA data and route feasibility assessments, we are ready to provide the data-driven insights you need. Partner with us to secure a sustainable, high-quality supply of critical pharmaceutical intermediates that drive your business forward.
