Advanced Manufacturing of Quinazoline Derivatives for Oncology Drug Supply Chains
Introduction to Next-Generation Quinazoline Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for kinase inhibitors, particularly those targeting the Epidermal Growth Factor Receptor (EGFR). Patent CN101357905B discloses a groundbreaking methodology for preparing 4-[3-chloro-4-(3-fluoro-benzyloxy)-anilino]-6-substituted amino-quinazoline derivatives, which serve as critical intermediates in oncology drug development. This technology addresses significant bottlenecks in traditional manufacturing by reordering the synthetic sequence to prioritize cost-efficiency and yield optimization. By shifting the acylation step to an earlier stage in the pathway, the process mitigates the economic risks associated with late-stage functionalization of high-value precursors. This strategic modification not only enhances the overall throughput but also simplifies the purification landscape, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships aiming for long-term commercial viability.
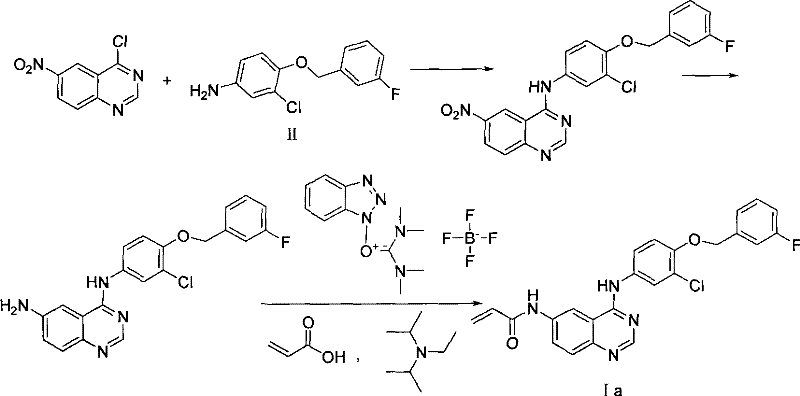
The core innovation lies in the preparation of the quinazoline core (Compound III) prior to its coupling with the expensive aniline component (Compound II). Conventional methods often suffer from cumulative yield losses when complex substituents are introduced at the final stages. In contrast, this patented approach establishes the 6-position substituent, such as an acrylamide or sulfonamide group, on the simpler nitro-amino quinazoline scaffold. This ensures that the costly 3-chloro-4-(3-fluoro-benzyloxy)-aniline is only introduced in the final nucleophilic substitution step, thereby maximizing the return on investment for raw materials. For procurement managers, this translates to a more predictable cost structure and reduced exposure to price volatility of specialized aniline derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes, such as those described in WO2006/071017, typically employ a linear strategy where the 6-position acylation is performed as the terminal step of the synthesis. This approach necessitates the use of expensive condensing agents like O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate to drive the amide bond formation. Such reagents significantly inflate the bill of materials and introduce complex impurity profiles that require rigorous removal. Furthermore, performing this low-yielding acylation reaction on the fully assembled molecule means that any loss directly impacts the expensive aniline moiety that was installed in the preceding step. This inefficiency creates a substantial burden on cost reduction in API manufacturing, as the overall yield is dragged down by the least efficient step occurring at the point of highest value accumulation.
The Novel Approach
The novel methodology presented in CN101357905B fundamentally restructures the synthetic logic by executing the acylation on the 6-amino-quinazolin-4-one intermediate before the final coupling event. By utilizing reactive and inexpensive acylating agents like acryloyl chloride or p-toluenesulfonyl chloride at this earlier stage, the process eliminates the need for costly coupling promoters. This rearrangement allows for the optimization of the acylation conditions independently of the sensitive aniline coupling step. Consequently, the final nucleophilic substitution becomes a high-yielding convergent step, joining two well-characterized fragments. This strategic decoupling of reaction steps enhances the commercial scale-up of complex heterocycles by isolating potential failure points and ensuring that the most expensive raw material is subjected to the fewest number of chemical transformations.
Mechanistic Insights into Early-Stage Acylation Strategy
The mechanistic advantage of this route is rooted in the chemoselectivity achievable on the quinazolinone core. The synthesis begins with the protection of the N3 nitrogen using a tert-butoxycarbonylmethyl group, which prevents unwanted side reactions during subsequent reductions. Following the reduction of the 6-nitro group to an amine, the resulting 6-amino functionality is highly nucleophilic and readily reacts with acid chlorides. This early acylation step is critical because it installs the pharmacophore-defining side chain while the molecule is still relatively small and easy to purify. The use of acryloyl chloride, for instance, proceeds rapidly under mild conditions, avoiding the thermal stress that might degrade more complex downstream intermediates. This precision in functional group manipulation is essential for maintaining high-purity quinazoline derivatives required for clinical-grade applications.
Following the acylation, the protecting group is removed under basic conditions, regenerating the lactam nitrogen and setting the stage for activation. The subsequent halogenation at the 4-position using oxalyl chloride converts the carbonyl oxygen into a good leaving group (chloride), creating the electrophilic center necessary for the final coupling. This sequence ensures that the electron-withdrawing effect of the 6-position substituent is already present during the halogenation, which can modulate the reactivity of the 4-position favorably. The final nucleophilic aromatic substitution with the aniline derivative then proceeds smoothly in protic solvents like isopropanol. This mechanistic flow minimizes the formation of bis-alkylated byproducts or hydrolysis impurities, thereby streamlining the downstream processing and reducing lead time for high-purity intermediates.
How to Synthesize Quinazoline Derivatives Efficiently
The standardized protocol for this synthesis involves a sequential five-step transformation starting from commercially available 6-nitro-3H-quinazolin-4-one. The process is designed to be telescoped where possible, particularly the acylation and deprotection steps, which can be performed in a one-pot fashion to minimize solvent usage and handling time. Operators must maintain strict control over reaction temperatures, particularly during the exothermic addition of sodium hydride and acid chlorides, to ensure safety and reproducibility. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive walkthrough for technical teams looking to implement this route in a GMP environment.
- Perform alkylation of 6-nitro-3H-quinazolin-4-one with tert-butyl chloroacetate in DMF to protect the nitrogen.
- Reduce the nitro group to an amine using iron powder and hydrochloric acid in an ethanol-water mixture.
- Execute acylation with acryloyl chloride followed by base-mediated deprotection to form the acrylamido-quinazolinone core.
- Activate the 4-position via halogenation using oxalyl chloride to generate the reactive chloro-quinazoline intermediate.
- Complete the synthesis via nucleophilic substitution with 3-chloro-4-(3-fluoro-benzyloxy)-aniline in isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this manufacturing route offers distinct advantages by decoupling the production of the quinazoline core from the sourcing of the specialized aniline. This modularity allows for the stocking of key intermediates, thereby enhancing supply chain reliability against fluctuations in the availability of fluorinated benzyl halides or chloro-anilines. The elimination of expensive peptide coupling reagents drastically simplifies the raw material portfolio, reducing the administrative burden of managing hazardous or high-cost chemicals. Moreover, the robust nature of the reactions, which utilize common industrial solvents like DMF, ethanol, and isopropanol, ensures that the process can be transferred easily between different manufacturing sites without requiring specialized equipment modifications.
- Cost Reduction in Manufacturing: The primary economic driver is the substitution of expensive condensing agents with commodity acid chlorides, which significantly lowers the direct material costs. By moving the acylation to an earlier step, the process avoids the financial penalty of losing expensive aniline starting materials in low-yield reactions. The ability to perform one-pot reactions for acylation and deprotection further reduces labor costs and solvent consumption, contributing to substantial cost savings in the overall production budget without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on widely available reagents such as iron powder for reduction and oxalyl chloride for halogenation mitigates the risk of supply disruptions often associated with specialized catalysts. The synthetic route is resilient to minor variations in raw material quality due to the inclusion of recrystallization steps that purge impurities effectively. This robustness ensures consistent delivery schedules, allowing pharmaceutical partners to plan their API production campaigns with greater confidence and reduced buffer stock requirements.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on multi-gram scales in the patent examples. The use of aqueous workups and standard organic solvents facilitates waste stream management, aligning with modern environmental compliance standards. The avoidance of heavy metal catalysts simplifies the residual metal testing and clearance, accelerating the regulatory filing process for new drug applications and ensuring a smoother path to market commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinazoline synthesis platform. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for technology transfer discussions. Understanding these nuances is vital for R&D teams evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: How does this synthesis method reduce production costs compared to prior art?
A: The method shifts the acylation step to an earlier stage using inexpensive acryloyl chloride instead of expensive coupling agents, preventing the loss of costly aniline raw materials in low-yield final steps.
Q: What is the purity profile of the quinazoline intermediates produced?
A: The process utilizes recrystallization steps (e.g., from ethyl acetate or ethanol) after key transformations, ensuring high chemical purity suitable for downstream API manufacturing without extensive chromatography.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the protocol avoids sensitive catalysts and uses robust solvents like DMF and isopropanol, with specific examples demonstrating successful scaling and one-pot reaction capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we leverage deep expertise in heterocyclic chemistry to bring complex patented routes like CN101357905B to commercial reality. Our facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, ensuring seamless technology transfer from lab to plant. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every batch for critical impurities, ensuring that the intermediates supplied meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for EGFR inhibitor programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your drug development goals efficiently and economically.
