Advanced Synthetic Routes for Brassinosteroid Intermediates and Commercial Scalability
Advanced Synthetic Routes for Brassinosteroid Intermediates and Commercial Scalability
The chemical landscape for synthesizing biologically active steroid derivatives has evolved significantly with the disclosure of patent CN87106588A, which outlines a robust methodology for preparing specific steroid intermediates essential for the production of brassinosteroids. These compounds serve as potent plant-growth regulators, and their efficient manufacture is critical for the agricultural sector. The patent details a streamlined synthetic pathway starting from readily available sterols, such as brassicasterol or stigmasterol derivatives, represented by general formula (1), and converting them through a series of optimized transformations into the highly valued diene-ketone structures of formula (5). 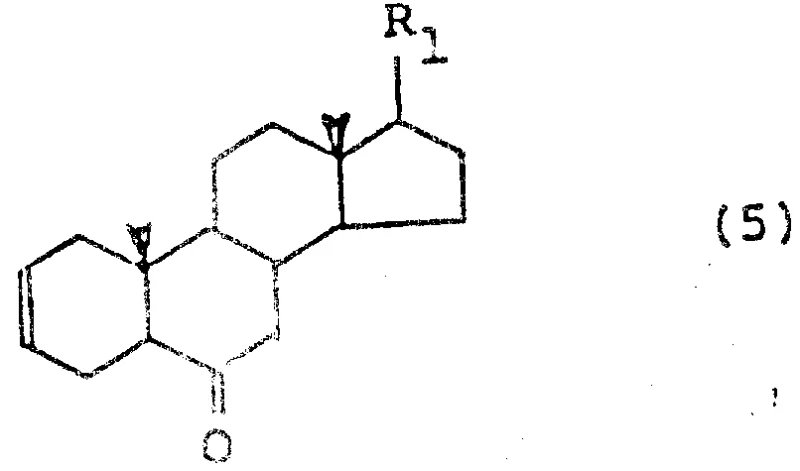 This technological breakthrough addresses long-standing inefficiencies in steroid functionalization, particularly regarding solvent management and reaction continuity, offering a compelling value proposition for R&D directors seeking high-purity agrochemical intermediates. By leveraging specific sulfonic acid amine salts for the final elimination step and optimizing oxidation protocols, this process ensures superior yield consistency and operational simplicity compared to legacy methods described in earlier literature.
This technological breakthrough addresses long-standing inefficiencies in steroid functionalization, particularly regarding solvent management and reaction continuity, offering a compelling value proposition for R&D directors seeking high-purity agrochemical intermediates. By leveraging specific sulfonic acid amine salts for the final elimination step and optimizing oxidation protocols, this process ensures superior yield consistency and operational simplicity compared to legacy methods described in earlier literature.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex steroid intermediates like those described in formula (4) and (5) has been plagued by operational discontinuities that hinder industrial scalability. Prior art, including references from J. Org. Chem. and various Japanese patent publications, typically necessitates a disjointed workflow where each reaction step requires a complete change of solvent environment. For instance, transitioning from a sulfonation reaction to a hydrolysis step often demands exhaustive aqueous washing, drying, and solvent evaporation to remove incompatible reagents before the next transformation can commence. This batch-to-batch fragmentation not only inflates processing time but also leads to significant material loss during isolation and purification stages. Furthermore, conventional oxidation methods frequently rely on harsh conditions, such as strong mineral acids in Jones oxidation, which can compromise acid-sensitive functional groups elsewhere on the steroid nucleus, thereby limiting the scope of substrates that can be effectively utilized without extensive protection-deprotection strategies.
The Novel Approach
In stark contrast, the methodology disclosed in CN87106588A introduces a cohesive strategy that maintains solvent compatibility across multiple reaction stages, specifically utilizing aromatic or aliphatic hydrocarbons like toluene or benzene as a continuous medium. 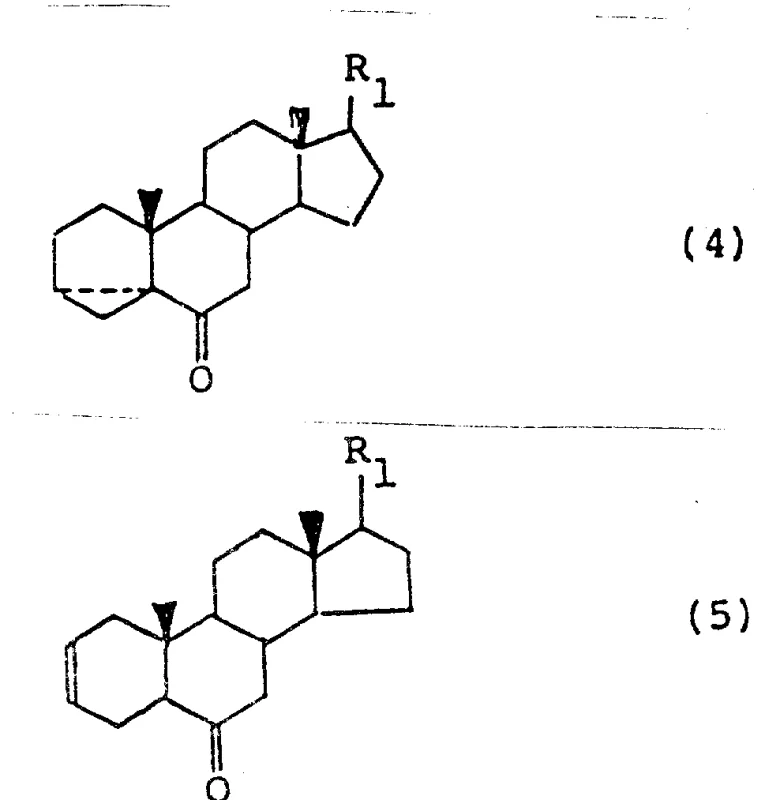 This innovation allows for the direct progression from the initial sulfonate ester formation (Formula 2) to the isomerized alcohol (Formula 3) and subsequently to the ketone (Formula 4) without the need for intermediate solvent stripping. The process cleverly integrates phase-transfer catalysis concepts during the hydrolysis step, enabling efficient reaction kinetics in a biphasic system that simplifies workup procedures. By minimizing unit operations and reducing the reliance on volatile chlorinated solvents in favor of recoverable hydrocarbons, this novel approach drastically simplifies the manufacturing workflow. The ability to perform three or four consecutive reactions in a single vessel or solvent system represents a paradigm shift in cost reduction in agrochemical intermediate manufacturing, directly addressing the pain points of procurement managers focused on operational expenditure.
This innovation allows for the direct progression from the initial sulfonate ester formation (Formula 2) to the isomerized alcohol (Formula 3) and subsequently to the ketone (Formula 4) without the need for intermediate solvent stripping. The process cleverly integrates phase-transfer catalysis concepts during the hydrolysis step, enabling efficient reaction kinetics in a biphasic system that simplifies workup procedures. By minimizing unit operations and reducing the reliance on volatile chlorinated solvents in favor of recoverable hydrocarbons, this novel approach drastically simplifies the manufacturing workflow. The ability to perform three or four consecutive reactions in a single vessel or solvent system represents a paradigm shift in cost reduction in agrochemical intermediate manufacturing, directly addressing the pain points of procurement managers focused on operational expenditure.
Mechanistic Insights into Sulfonate-Mediated Isomerization and Oxidation
The core of this synthetic route lies in the precise manipulation of the steroid A-ring and side chain through a sequence of activation, migration, and oxidation events. The initial reaction involves the conversion of the C3-hydroxyl group of the starting sterol into a sulfonate ester (Formula 2) using reagents like methanesulfonyl chloride or p-toluenesulfonyl chloride in the presence of organic bases such as triethylamine. This activation is crucial as it converts a poor leaving group into an excellent one, setting the stage for the subsequent skeletal rearrangement. Upon treatment with aqueous base, potentially aided by phase-transfer catalysts like tetrabutylammonium bromide, the sulfonate undergoes hydrolysis coupled with a double bond migration. This isomerization shifts the unsaturation from the typical delta-5 position to the delta-6 position (or related isomers depending on the specific substrate), yielding the allylic alcohol of Formula (3). This mechanistic pathway is highly regioselective, ensuring that the stereochemical integrity of the chiral centers at C8, C9, and C14 is preserved while establishing the necessary functionality for the upcoming oxidation.
Following the isomerization, the oxidation of the secondary alcohol to the corresponding ketone (Formula 4) is executed with remarkable flexibility. The patent elucidates two distinct mechanistic pathways: the classical Jones oxidation utilizing chromic acid in acetone, and a milder, metal-free alternative employing N-halo succinimides (such as N-bromosuccinimide or N-iodosuccinimide) in the presence of quaternary ammonium salts. 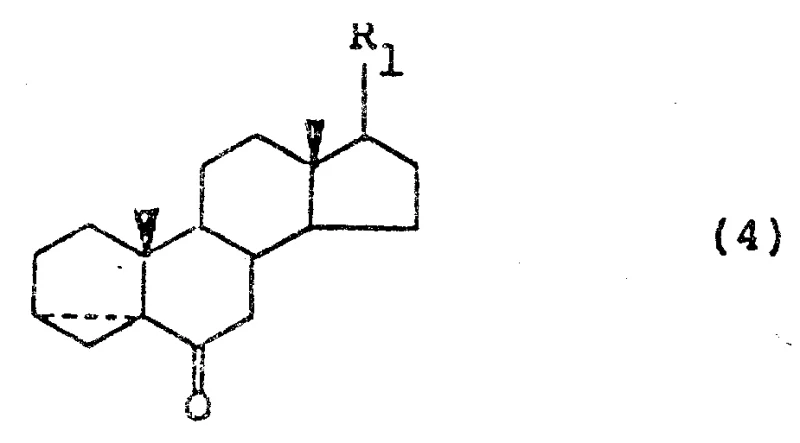 The latter mechanism operates via the formation of an alkoxy-ammonium species or similar activated intermediate that facilitates hydride abstraction without generating toxic chromium waste. This is particularly advantageous for maintaining high purity standards, as it eliminates the need for rigorous heavy metal scavenging steps that often complicate downstream processing. The resulting ketone (Formula 4) serves as the pivotal precursor for the final dehydration, where the introduction of the delta-2 double bond is achieved through an acid-catalyzed elimination mechanism driven by the thermal decomposition of sulfonic acid amine salts, ensuring the formation of the conjugated dienone system essential for biological potency.
The latter mechanism operates via the formation of an alkoxy-ammonium species or similar activated intermediate that facilitates hydride abstraction without generating toxic chromium waste. This is particularly advantageous for maintaining high purity standards, as it eliminates the need for rigorous heavy metal scavenging steps that often complicate downstream processing. The resulting ketone (Formula 4) serves as the pivotal precursor for the final dehydration, where the introduction of the delta-2 double bond is achieved through an acid-catalyzed elimination mechanism driven by the thermal decomposition of sulfonic acid amine salts, ensuring the formation of the conjugated dienone system essential for biological potency.
How to Synthesize Brassinosteroid Intermediates Efficiently
To implement this sophisticated synthetic route effectively, manufacturers must adhere to strict control parameters regarding temperature, stoichiometry, and solvent quality to maximize yield and minimize impurity profiles. The process begins with the careful sulfonation of the starting sterol at controlled low temperatures to prevent side reactions, followed by a carefully managed hydrolysis step where the ratio of water to organic solvent is critical for driving the isomerization equilibrium. Subsequent oxidation requires precise monitoring, whether using the exothermic Jones reagent or the slower N-halo succinimide protocol, to avoid over-oxidation. Finally, the thermal dehydration step demands high-temperature stability and the selection of appropriate high-boiling polar aprotic solvents like dimethylacetamide to facilitate the elimination. For a detailed breakdown of the specific reagent quantities, reaction times, and purification techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- React starting steroid (Formula 1) with sulfonyl halides in aromatic solvents to form sulfonate esters (Formula 2).
- Perform base-catalyzed hydrolysis and isomerization in a biphasic system to yield the isomeric alcohol (Formula 3).
- Oxidize the alcohol to the corresponding ketone (Formula 4) using Jones reagent or N-halo succinimides.
- Execute thermal dehydration using sulfonic acid amine salts to generate the final diene-ketone product (Formula 5).
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of the synthetic methodology described in CN87106588A offers profound benefits that extend beyond mere chemical yield, fundamentally altering the cost structure of producing high-purity steroid derivatives. The primary advantage lies in the drastic simplification of the unit operations; by enabling continuous processing in a single solvent system, the need for intermediate isolation, drying, and solvent exchange is virtually eliminated. This reduction in processing steps translates directly into lower energy consumption for heating and vacuum distillation, as well as a significant decrease in labor hours required for reactor turnover and cleaning. For procurement managers, this means a more predictable production schedule with fewer bottlenecks, allowing for better alignment with just-in-time inventory models and reducing the working capital tied up in work-in-progress materials. The streamlined workflow also minimizes the exposure of sensitive intermediates to atmospheric conditions, thereby enhancing the overall consistency of the final product quality.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous solvent swaps that characterize traditional steroid synthesis. By utilizing common aromatic hydrocarbons like toluene throughout the sequence, manufacturers can leverage bulk purchasing power for a single solvent type rather than maintaining a diverse inventory of specialized solvents. Furthermore, the option to use N-halo succinimides for oxidation avoids the regulatory and disposal costs associated with hexavalent chromium waste, which is a significant hidden cost in traditional Jones oxidations. The qualitative reduction in waste generation and the ability to recycle the primary reaction solvent multiple times without extensive purification creates a leaner manufacturing model that substantially lowers the cost of goods sold (COGS) without compromising on the stringent purity specifications required for agrochemical applications.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as reagents, such as methanesulfonyl chloride, triethylamine, and sodium carbonate, which are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenic cooling for most steps, ensures that production can be maintained even during fluctuations in utility availability. Additionally, the high yields reported in the patent examples for each transformation step imply a higher throughput per batch, allowing manufacturers to meet large-volume orders with fewer reactor runs. This efficiency is critical for reducing lead time for high-purity agrochemical intermediates, ensuring that downstream formulators receive their raw materials consistently and without delay, thus stabilizing the entire value chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exotic catalysts or extreme pressure requirements, making it compatible with standard glass-lined or stainless steel reactor infrastructure found in most fine chemical plants. The shift towards metal-free oxidation pathways aligns with increasingly stringent environmental regulations regarding heavy metal discharge, future-proofing the manufacturing site against tighter compliance standards. The thermal dehydration step, while requiring elevated temperatures, is conducted in closed systems with solvent recovery capabilities, minimizing volatile organic compound (VOC) emissions. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for multinational corporations when selecting reliable agrochemical intermediate suppliers for their long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this steroid synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing production lines. The answers provided reflect the specific conditions and outcomes observed in the patent examples, offering a realistic expectation of performance and potential challenges.
Q: What are the advantages of the continuous solvent system in this patent?
A: The patent describes using consistent aromatic or aliphatic hydrocarbon solvents (like toluene) throughout multiple steps, eliminating the need for intermediate solvent swaps and extensive washing, which significantly reduces waste and processing time.
Q: How is the oxidation step optimized for industrial safety?
A: While traditional Jones oxidation uses chromium, the patent offers an alternative using N-halo succinimides with quaternary ammonium salts, avoiding heavy metal contamination and strong acidic conditions that could damage sensitive protecting groups.
Q: What represents the key structural transformation in the final step?
A: The final step involves a regioselective dehydration of the 6-ketone steroid using specific sulfonic acid amine salts at elevated temperatures (150-200°C) to introduce the critical delta-2 double bond required for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brassinosteroid Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of agrochemical and pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN87106588A can be translated into reliable industrial reality. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for plant-growth regulator synthesis. Our facility is equipped to handle the specific solvent systems and thermal conditions required for this process, guaranteeing a supply continuity that supports your long-term strategic goals.
We invite you to collaborate with us to optimize your supply chain for steroid derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patented technology can drive value for your organization. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-value fine chemical intermediates.
