Advanced Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
Advanced Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer enhanced biological profiles and streamlined synthetic accessibility. Patent CN114163436A introduces a significant breakthrough in this domain by disclosing a new class of diarylmethane derivatives containing an indolizine core. These compounds are not merely structural variations but represent a strategic evolution in medicinal chemistry, designed to overcome the limitations of prior art molecules which often suffered from narrow spectrum activity. As a reliable pharmaceutical intermediate supplier, understanding the depth of this innovation is crucial for R&D teams aiming to develop next-generation therapeutics. The disclosed compounds exhibit a unique dual functionality, possessing both potent antibacterial and anticancer activities, which addresses a critical gap in current drug discovery pipelines where multi-target agents are increasingly valued.
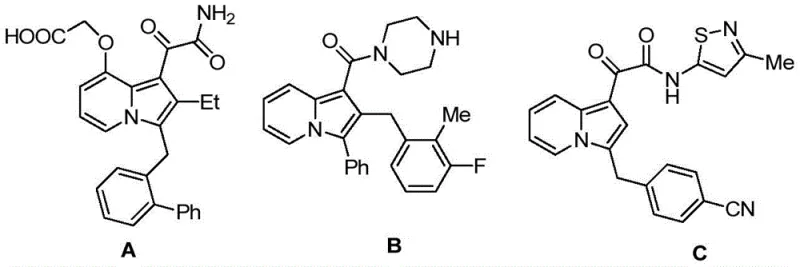
The structural versatility of these derivatives is defined by their ability to incorporate diverse substituents, allowing for fine-tuning of pharmacokinetic properties. The general formula encompasses variants where the indolizine ring is fused with either benzofuran or indole moieties, creating a rigid, planar architecture that is often favorable for intercalating into DNA or binding to specific enzyme pockets. This structural complexity, however, traditionally poses significant challenges in synthesis. The patent provides a robust solution by detailing a preparation method that transforms simple, commercially available precursors into these high-value intermediates. For procurement managers and supply chain heads, this represents a tangible opportunity for cost reduction in API manufacturing, as the route avoids exotic reagents and relies on established chemical transformations that can be easily sourced and managed within a global supply network.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diarylmethane structures containing indolizine has been fraught with inefficiencies. Conventional routes often require multi-step sequences involving harsh reaction conditions, toxic reagents, and tedious purification protocols that drive up both the cost and the environmental footprint of production. Many existing methods struggle with regioselectivity, leading to complex mixtures of isomers that are difficult to separate, thereby reducing the overall yield and purity of the final active pharmaceutical ingredient. Furthermore, prior art compounds, such as those referenced in the background of the patent, frequently exhibit only a single type of biological activity, limiting their therapeutic potential and necessitating the synthesis of entirely new scaffolds to address different disease pathways. This fragmentation in drug design leads to increased R&D expenditure and prolonged timelines for bringing new medicines to market, a pain point that this new technology aims to resolve through molecular efficiency.
The Novel Approach
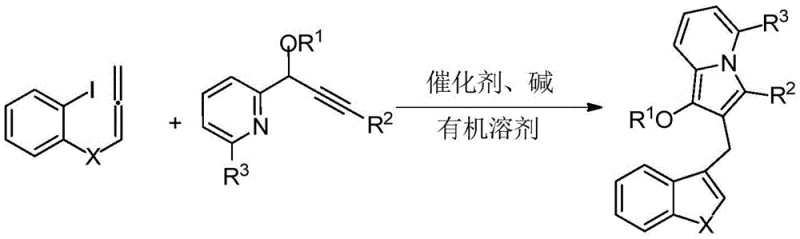
The innovative methodology described in the patent utilizes a palladium-catalyzed tandem cyclization reaction that constructs the complex indolizine-diarylmethane skeleton in a single operational step. This approach leverages the reactivity of 2-iodophenyl allene compounds and propargyl pyridine derivatives, which undergo a sophisticated cascade of bond-forming events to generate the target structure with high precision. By employing a palladium catalyst, such as tetrakis(triphenylphosphine)palladium, the reaction proceeds under relatively mild thermal conditions, typically between 60°C and 90°C, which preserves sensitive functional groups and minimizes energy consumption. This one-pot strategy drastically simplifies the process flow, eliminating the need for isolating unstable intermediates and reducing the total number of unit operations required. The result is a streamlined synthesis that offers superior atom economy and operational simplicity, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the intricate mechanism of the palladium-catalyzed cascade. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive organopalladium intermediate. This species then undergoes a migratory insertion into the alkyne moiety of the propargyl pyridine component, forming a new carbon-carbon bond and setting the stage for the subsequent cyclization. The presence of the base, such as potassium carbonate or cesium carbonate, plays a critical role in facilitating the deprotonation steps necessary for the final aromatization or ring closure, ensuring the formation of the stable indolizine core. This mechanistic pathway is highly efficient because it couples two distinct bond-forming events into a single catalytic cycle, thereby maximizing the utility of the catalyst and minimizing side reactions that could lead to impurity formation.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise approaches. Because the cyclization occurs intramolecularly or in a tightly coupled intermolecular fashion immediately following the initial activation, the opportunity for intermolecular polymerization or oligomerization of the reactive allene or alkyne species is significantly reduced. The use of specific ligands on the palladium center further enhances regioselectivity, ensuring that the cyclization occurs at the desired position to yield the specific indolizine isomer required for biological activity. This high level of control translates directly to a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final product meets the rigorous quality standards demanded by regulatory bodies for clinical-grade materials. Understanding these mechanistic nuances allows process chemists to optimize reaction parameters such as solvent polarity and catalyst loading to further enhance performance.
How to Synthesize Indolizine Diarylmethane Derivatives Efficiently
The practical execution of this synthesis involves a straightforward protocol that balances reaction efficiency with ease of handling. The process begins by combining the key starting materials—the 2-iodophenyl allene derivative and the propargyl pyridine compound—in a suitable organic solvent such as N,N-dimethylformamide or toluene. A palladium catalyst is introduced along with an inorganic base to initiate the transformation. The reaction mixture is then heated under an inert atmosphere, typically nitrogen, to maintain catalyst stability and prevent oxidation of sensitive intermediates. Following the completion of the reaction, which is monitored to ensure full conversion, the workup procedure involves standard aqueous extraction to remove inorganic salts and polar byproducts. The organic layer is dried and concentrated, and the resulting crude material is purified using column chromatography to isolate the high-purity indolizine derivative. Detailed standardized synthesis steps are provided in the guide below.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst, alkali, and organic solvent in a reaction vessel under inert atmosphere.
- Stir and react the mixture at a temperature between 60-90°C for a duration of 4 to 12 hours to facilitate the tandem cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying, filtration, solvent removal, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary advantage lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By consolidating multiple synthetic steps into a single tandem reaction, the requirement for intermediate isolation, drying, and quality control testing is eliminated, leading to a drastic reduction in labor costs and facility occupancy time. Furthermore, the raw materials utilized in this process, specifically the iodo-allene and propargyl pyridine precursors, are derived from commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of supply chain disruptions associated with bespoke or proprietary starting materials.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final product is not the only cost saver; the process itself is designed for efficiency. By avoiding the use of expensive protecting group strategies often required in traditional heterocycle synthesis, the overall material cost is substantially lowered. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, which lowers the capital expenditure barrier for production. Additionally, the high selectivity of the palladium-catalyzed system minimizes the formation of difficult-to-remove impurities, reducing the volume of solvents and silica gel required for purification, which represents a significant variable cost saving in large-scale manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent output quality, which is vital for maintaining a reliable supply of critical intermediates. Since the reaction tolerates a wide range of substituents on the aromatic rings, manufacturers can produce a diverse library of analogues using the same core platform technology, allowing for rapid response to changing market demands or clinical trial requirements. The use of common solvents like DMF and ethyl acetate further simplifies logistics, as these materials are universally stocked and do not require special hazardous material handling permits beyond standard regulations. This flexibility ensures that production schedules can be maintained even when specific niche reagents face temporary shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards typically associated with aggressive reagents. The workup procedure relies on liquid-liquid extraction and crystallization or chromatography, techniques that are well-understood and easily adapted for continuous processing or large-batch operations. From an environmental standpoint, the atom economy of the tandem cyclization is superior to stepwise alternatives, resulting in less chemical waste generation per kilogram of product. This aligns with modern green chemistry principles and helps pharmaceutical companies meet their sustainability goals by reducing the E-factor of their manufacturing processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine-containing diarylmethane derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing drug development or manufacturing workflows.
Q: What are the primary biological activities of these new indolizine derivatives?
A: Unlike previous compounds that often exhibited single biological activity, these novel derivatives simultaneously possess both antibacterial and anticancer activities, making them highly valuable for dual-target drug development.
Q: Is the palladium catalyst recoverable in this synthesis process?
A: While the patent describes a homogeneous catalytic system using palladium salts like tetrakis(triphenylphosphine)palladium, standard industrial workup procedures such as filtration and chromatography are employed to ensure the final product meets stringent purity specifications regarding heavy metal residues.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the method utilizes readily available raw materials and mild reaction conditions (60-90°C) in common organic solvents, which significantly simplifies the engineering requirements for scaling up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Diarylmethane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN114163436A for the development of advanced antibacterial and anticancer agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indolizine intermediate meets the exacting standards required for clinical and commercial applications. We are committed to delivering high-purity indolizine derivatives that empower your research and development teams to push the boundaries of medicinal chemistry.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in turning complex chemical innovations into commercial realities.
