Advanced Copper-Catalyzed Synthesis of N-Methyl-3-Benzyl Mercapto-4-Amino Maleimide Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign routes to synthesize complex heterocyclic scaffolds, particularly maleimide derivatives which serve as critical linkers in antibody-drug conjugates (ADCs) and kinase inhibitors. Patent CN111548299A discloses a groundbreaking methodology for the preparation of N-methyl-3-benzyl mercapto-4-amino maleimide compounds, addressing long-standing issues regarding reagent stability and odor. This innovation utilizes a transition metal copper-catalyzed tandem reaction involving benzyl Bunte salts, secondary amines, and N-methylmaleimide under an oxygen atmosphere. By shifting away from traditional thiol-based reagents, this technology offers a robust platform for generating high-purity intermediates essential for modern drug discovery pipelines. The strategic implementation of this synthetic route allows manufacturers to bypass the logistical nightmares associated with volatile sulfur compounds while maintaining exceptional yields and structural diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-disubstituted maleimides has relied heavily on nucleophilic substitution reactions using activated maleimide precursors. As illustrated in earlier literature such as the work by Dubinina et al. in 2002, the standard approach involved reacting 3,4-dichloromaleimide with amines and thiophenols under basic conditions. 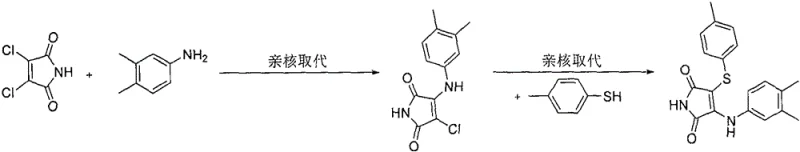 This conventional pathway presents severe drawbacks for industrial application, primarily due to the high cost and limited availability of 3,4-dichloromaleimide starting materials. Furthermore, the reliance on free thiophenols introduces significant safety and environmental hazards; these compounds are notoriously malodorous, toxic, and prone to oxidation, complicating waste management and worker safety protocols. The requirement for harsh basic conditions and the generation of stoichiometric halide waste further diminish the atom economy and green chemistry profile of these legacy methods, making them increasingly obsolete for large-scale pharmaceutical manufacturing.
This conventional pathway presents severe drawbacks for industrial application, primarily due to the high cost and limited availability of 3,4-dichloromaleimide starting materials. Furthermore, the reliance on free thiophenols introduces significant safety and environmental hazards; these compounds are notoriously malodorous, toxic, and prone to oxidation, complicating waste management and worker safety protocols. The requirement for harsh basic conditions and the generation of stoichiometric halide waste further diminish the atom economy and green chemistry profile of these legacy methods, making them increasingly obsolete for large-scale pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel methodology described in CN111548299A leverages a copper-catalyzed C(sp2)-H thioamination strategy that fundamentally alters the reactant landscape. Instead of pre-functionalized dichloromaleimides, this process utilizes readily available N-methylmaleimide, secondary amines, and benzyl Bunte salts as the sulfur source.  This three-component coupling reaction proceeds efficiently under an oxygen atmosphere, utilizing molecular oxygen as the terminal oxidant, which is both cost-effective and environmentally friendly. The use of Bunte salts (S-sulfonates) is a game-changer; these reagents are odorless, stable solids that are easy to handle and store, eliminating the volatility issues associated with thiols. The reaction tolerates a wide range of secondary amines, including cyclic amines like morpholine and piperidine, as well as acyclic amines, demonstrating remarkable substrate scope. This shift not only simplifies the supply chain by removing the need for specialized chlorinated precursors but also significantly enhances the operational safety profile of the synthesis facility.
This three-component coupling reaction proceeds efficiently under an oxygen atmosphere, utilizing molecular oxygen as the terminal oxidant, which is both cost-effective and environmentally friendly. The use of Bunte salts (S-sulfonates) is a game-changer; these reagents are odorless, stable solids that are easy to handle and store, eliminating the volatility issues associated with thiols. The reaction tolerates a wide range of secondary amines, including cyclic amines like morpholine and piperidine, as well as acyclic amines, demonstrating remarkable substrate scope. This shift not only simplifies the supply chain by removing the need for specialized chlorinated precursors but also significantly enhances the operational safety profile of the synthesis facility.
Mechanistic Insights into Copper-Catalyzed C-H Thioamination
The core of this technological advancement lies in the intricate catalytic cycle mediated by cuprous iodide (CuI). Mechanistic studies suggest that the reaction initiates with the activation of the Bunte salt by the copper catalyst, potentially generating a sulfur-centered radical species or a copper-sulfur intermediate. Under the oxidative conditions provided by molecular oxygen, the copper center facilitates the cleavage of the S-SO3 bond in the Bunte salt, releasing the active thiolating species in situ. This species then attacks the electron-deficient double bond of the N-methylmaleimide, likely following a radical addition pathway or a concerted metal-mediated insertion. Simultaneously, the secondary amine participates in the functionalization of the adjacent position, leading to the dual substitution pattern observed in the final product. The synergy between the copper catalyst and the oxygen atmosphere is crucial; control experiments detailed in the patent reveal that other copper salts such as copper acetate, copper chloride, or copper bromide fail to drive the reaction, highlighting the unique electronic properties of the iodide ligand in stabilizing the active catalytic species.
Impurity control is inherently built into this mechanism through the high chemoselectivity of the copper catalyst. Unlike traditional nucleophilic substitutions which often suffer from over-alkylation or hydrolysis of the imide ring, this oxidative coupling is highly specific for the C-H bonds at the 3 and 4 positions of the maleimide ring. The use of 1,2-dichloroethane (DCE) as the solvent plays a pivotal role in solubilizing the organic substrates while maintaining the stability of the copper catalyst; screening data indicates that polar aprotic solvents like DMF or non-polar solvents like toluene result in no reaction, suggesting a delicate balance of polarity and coordination ability is required. This specificity ensures that the resulting N-methyl-3-benzyl mercapto-4-amino maleimide compounds are obtained with high purity, minimizing the formation of regioisomers or side products that would otherwise require costly and yield-reducing purification steps downstream.
How to Synthesize N-Methyl-3-Benzyl Mercapto-4-Amino Maleimide Efficiently
Implementing this synthesis requires precise adherence to the optimized reaction parameters established in the patent examples to ensure reproducibility and maximum yield. The process involves a straightforward one-pot procedure where the molar ratio of benzyl Bunte salt, secondary amine, and N-methylmaleimide is maintained at approximately 3:2:1. The reaction is typically conducted at temperatures between 100°C and 120°C, with 100°C being the preferred setpoint to balance reaction kinetics and energy consumption. A critical operational step is the establishment of an oxygen-rich environment; the reaction vessel must be purged with oxygen gas at least three times before heating to ensure sufficient oxidant is available for the catalytic cycle. Following the reaction period of 18 to 24 hours, the workup involves cooling the mixture, dilution with ethyl acetate, and filtration to remove inorganic salts, followed by standard column chromatography purification.
- Combine benzyl Bunte salt (3 equiv), secondary amine (2 equiv), N-methylmaleimide (1 equiv), and CuI (10 mol%) in 1,2-dichloroethane.
- Purge the reaction vessel with oxygen gas three times to ensure an oxidative atmosphere.
- Stir the mixture at 100°C for 24 hours, then cool, dilute with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Bunte salt-based methodology translates into tangible strategic advantages beyond mere chemical elegance. The primary benefit is the drastic simplification of raw material sourcing; Bunte salts are commercially available, stable solids that do not require the specialized cold-chain logistics or hazardous material handling protocols mandated by volatile thiophenols. This stability significantly reduces inventory shrinkage and waste disposal costs associated with degraded reagents. Furthermore, the elimination of expensive 3,4-dichloromaleimide precursors lowers the direct material cost per kilogram of the final intermediate. The robustness of the reaction conditions, specifically the tolerance for ambient pressure oxygen rather than high-pressure reactors or inert gas manifolds, simplifies the equipment requirements for scale-up, allowing for utilization of standard glass-lined steel reactors found in most multipurpose chemical plants.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost, hazardous reagents with commodity chemicals. By utilizing benzyl Bunte salts instead of thiophenols, facilities avoid the substantial expenses related to scrubbing systems and personal protective equipment required for malodorous sulfur compounds. Additionally, the high atom economy of the three-component coupling minimizes waste generation, leading to lower effluent treatment costs. The use of CuI as a catalyst at low loading (10 mol%) ensures that precious metal recovery costs are negligible compared to processes requiring palladium or rhodium. These factors collectively contribute to a significantly reduced cost of goods sold (COGS) for the final maleimide intermediates, enhancing margin potential for downstream drug products.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, non-perishable starting materials. Unlike thiols which can oxidize upon storage leading to batch-to-batch variability, Bunte salts maintain their integrity over extended periods, ensuring consistent reaction performance. The solvent system, favoring 1,2-dichloroethane, relies on a widely available industrial solvent with a stable global supply chain, reducing the risk of production stoppages due to solvent shortages. Moreover, the simplicity of the workup procedure, which avoids complex aqueous quenches or pH adjustments, shortens the overall batch cycle time. This efficiency allows manufacturers to respond more rapidly to fluctuating demand from pharmaceutical clients, effectively reducing lead time for high-purity API precursors without compromising on quality standards.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The replacement of foul-smelling thiols with odorless salts drastically improves the working environment, reducing the risk of community complaints and regulatory scrutiny. The use of molecular oxygen as the oxidant generates water as the only byproduct of the oxidation step, minimizing the load of heavy metal waste or toxic organic byproducts in the wastewater stream. The reaction demonstrates excellent scalability, as evidenced by the consistent yields across various amine substrates in the patent examples, indicating that heat and mass transfer limitations are minimal. This makes the commercial scale-up of complex heterocyclic compounds feasible even in facilities with moderate infrastructure, ensuring a continuous and compliant supply of critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this copper-catalyzed thioamination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on catalyst selection, solvent effects, and substrate compatibility. Understanding these nuances is essential for process chemists aiming to transfer this laboratory-scale protocol into a GMP-compliant manufacturing environment.
Q: Why are Bunte salts preferred over thiophenols in this synthesis?
A: Bunte salts are odorless and stable solids, whereas thiophenols are malodorous, unstable liquids that pose significant environmental and handling challenges in industrial settings.
Q: What is the critical catalyst for this transformation?
A: Cuprous iodide (CuI) is the essential catalyst. Other copper salts like copper acetate, chloride, or bromide fail to promote the reaction effectively.
Q: Can this reaction be scaled for commercial production?
A: Yes, the use of stable solid reagents, simple oxygen atmosphere, and robust solvent systems like 1,2-dichloroethane makes this process highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-3-Benzyl Mercapto-4-Amino Maleimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality maleimide derivatives play in the development of next-generation therapeutics. Our technical team has extensively evaluated the synthetic route disclosed in CN111548299A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-methyl-3-benzyl mercapto-4-amino maleimide meets the exacting standards required for clinical and commercial applications. We are committed to delivering reliable pharmaceutical intermediates that empower your R&D efforts.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project requirements. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how this odorless, copper-catalyzed route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your critical maleimide building blocks.
