Advanced One-Pot Synthesis of Alpha-Allyl Substituted Alpha-Beta Unsaturated Ketones for Commercial Scale-Up
Advanced One-Pot Synthesis of Alpha-Allyl Substituted Alpha-Beta Unsaturated Ketones for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is detailed in patent CN108727179B, which introduces a robust method for synthesizing alpha-allyl substituted alpha,beta-unsaturated ketones, esters, and nitriles. These structural motifs are critical building blocks in the construction of complex pharmaceutical active ingredients and agrochemical intermediates. The disclosed technology leverages a sophisticated one-pot allylation-Wittig reaction sequence, utilizing readily available allylic alcohols as alkylating agents instead of the traditionally expensive and hazardous allyl carbonates or acetates. By employing a dual catalytic system comprising an allyl palladium chloride dimer and a bisphosphine ligand, this process achieves high efficiency under remarkably mild conditions, typically ranging from 20 to 100 degrees Celsius. For R&D directors and process chemists seeking reliable pathways to functionalized olefins, this methodology represents a paradigm shift towards greener and more economical synthesis.
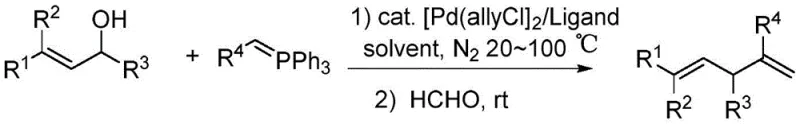
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha-allyl substituted alpha,beta-unsaturated carbonyl systems has been fraught with significant operational and economic challenges. Traditional literature methods often rely on the Morita-Baylis-Hillman (MBH) reaction or variations thereof, which typically necessitate the use of allyl acetates as electrophiles. These conventional protocols are notoriously inefficient, frequently requiring excessive amounts of palladium catalysts, often upwards of 10 mol%, alongside stoichiometric quantities of phosphorus reagents and acetic acid additives to drive the reaction to completion. Furthermore, alternative allylation strategies have depended on allyl carbonates or allyl amine derivatives as alkylating agents. These starting materials are not only difficult to source commercially on a large scale but also introduce substantial safety hazards due to their instability and potential toxicity. Additionally, these older methods often demand harsh reaction conditions, such as temperatures exceeding 100 degrees Celsius, and require the use of highly toxic, volatile organic solvents that complicate waste disposal and increase the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy techniques, the innovative approach outlined in the patent data utilizes simple, inexpensive, and stable allylic alcohols as the primary alkylating reagents. This strategic substitution fundamentally alters the economic and safety profile of the synthesis. The reaction proceeds smoothly in benign solvents such as methanol or even water, eliminating the reliance on hazardous volatile organic compounds. As demonstrated in specific embodiments, reacting cinnamyl alcohol with benzoylphosphine ylide in the presence of a catalytic amount of [Pd(allyl)Cl]2 and dppf ligand yields the desired alpha-allyl substituted product with exceptional efficiency. For instance, performing the reaction in water at 60 degrees Celsius for 12 hours resulted in an isolated yield of 96 percent, showcasing the robustness of the method. This one-pot protocol seamlessly integrates the allylation step with a subsequent Wittig reaction using formaldehyde, thereby streamlining the production workflow and minimizing intermediate isolation steps, which is a crucial factor for cost reduction in API manufacturing.
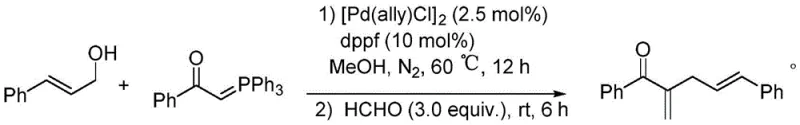
Mechanistic Insights into Pd-Catalyzed Allylation-Wittig Cascade
The success of this transformation hinges on the synergistic interplay between the palladium catalyst and the phosphorus ylide within a carefully tuned coordination environment. The mechanism initiates with the oxidative addition of the allylic alcohol to the low-valent palladium species generated in situ from the [Pd(allyl)Cl]2 dimer and the bidentate dppf ligand. This step forms a reactive pi-allyl palladium complex, which is the key electrophilic intermediate. The stabilized phosphorus ylide then acts as a nucleophile, attacking the pi-allyl complex to effect the C-C bond formation at the alpha-position. Crucially, the choice of the dppf ligand is paramount; its wide bite angle and electronic properties stabilize the palladium center, preventing premature decomposition and ensuring high turnover numbers. Following the allylation, the reaction mixture is treated with formaldehyde. This triggers the Wittig olefination, where the remaining phosphorus species reacts with the aldehyde to generate the exocyclic methylene group, finalizing the alpha,beta-unsaturated structure. This cascade design avoids the accumulation of unstable intermediates and drives the equilibrium forward effectively.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over stepwise syntheses. By conducting the reaction in a one-pot fashion, the exposure of sensitive intermediates to air or moisture is minimized, significantly reducing the formation of hydrolysis byproducts or oxidation impurities. The use of water or alcohol as a solvent further aids in suppressing side reactions that might occur in more polar aprotic solvents. Moreover, the mild reaction temperature of 60 degrees Celsius prevents thermal degradation of the product or the starting materials, which is a common issue in high-temperature allylations. The high selectivity observed, particularly the exclusive formation of the E-isomer in many cases as indicated by NMR data, suggests a highly ordered transition state controlled by the steric bulk of the ligand and the substrate. This level of stereocontrol is vital for pharmaceutical applications where isomeric purity directly impacts biological activity and regulatory approval.
How to Synthesize Alpha-Allyl Substituted Alpha-Beta Unsaturated Ketones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent to ensure reproducibility and high yield. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions. The key lies in the precise stoichiometry of the catalyst system and the sequential addition of reagents. Operators should first establish an inert atmosphere to protect the sensitive palladium catalyst from oxidation before introducing the allylic alcohol and the phosphorus ylide. The reaction progress can be conveniently monitored via TLC, allowing for real-time decision-making regarding the addition of the formaldehyde solution. For a comprehensive understanding of the exact procedural nuances, including specific stirring rates and workup protocols, please refer to the detailed guide below.
- Combine allylic alcohol, phosphorus ylide, [Pd(allyl)Cl]2 catalyst, and diphosphine ligand in a solvent like methanol or water.
- Stir the mixture under nitrogen atmosphere at 60°C for 12 hours to complete the allylation step.
- Add aqueous formaldehyde solution at room temperature and stir for 6 hours to finalize the Wittig reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from expensive, hard-to-source allyl carbonates to commodity-grade allylic alcohols represents a fundamental restructuring of the raw material cost base. Allylic alcohols are produced on a massive industrial scale for various applications, ensuring a stable and continuous supply chain that is less susceptible to market volatility. Furthermore, the ability to utilize water as a reaction solvent drastically reduces the costs associated with solvent purchase, recovery, and disposal. In traditional organic synthesis, the volume of organic solvent waste often exceeds the mass of the product itself; replacing this with water significantly lowers the environmental compliance burden and the associated fees for hazardous waste treatment. This green chemistry approach aligns perfectly with the increasing regulatory pressures faced by multinational corporations to reduce their carbon footprint and improve sustainability metrics.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of costly reagents and the reduction of catalyst loading. Traditional methods often require stoichiometric amounts of activating agents or high loadings of precious metal catalysts, whereas this novel route operates efficiently with as little as 0.5 to 2.5 mol percent of the palladium dimer. This drastic reduction in precious metal usage directly lowers the bill of materials. Additionally, the one-pot nature of the reaction removes the need for intermediate isolation and purification steps, which are typically labor-intensive and result in material loss. By consolidating two distinct chemical transformations into a single reactor vessel, manufacturers can achieve substantial savings in both time and operational expenses, leading to a significantly improved overall profit margin for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Unlike specialized allylating agents that may have limited suppliers and long lead times, allylic alcohols and phosphorus ylides are standard catalog items available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions—tolerating temperatures up to 100 degrees Celsius and functioning well in aqueous media—means that the process is less sensitive to minor fluctuations in utility supplies or environmental controls. This reliability ensures consistent batch-to-batch quality and on-time delivery performance, which are critical KPIs for maintaining trust with downstream pharmaceutical clients who operate on tight Just-In-Time manufacturing schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often reveals hidden bottlenecks, particularly regarding heat transfer and safety. This methodology is inherently scalable because it avoids exothermic hazards associated with unstable reagents and operates at moderate temperatures. The use of water or methanol as solvents simplifies the engineering requirements for the reactor setup, as these solvents have well-understood physical properties and safety profiles. From an environmental compliance standpoint, the reduction in volatile organic compound (VOC) emissions is a major advantage. Regulatory bodies worldwide are tightening restrictions on VOC releases, and processes that minimize organic solvent usage are increasingly favored. By adopting this water-compatible protocol, manufacturers can future-proof their operations against stricter environmental regulations, avoiding costly retrofits or fines while enhancing their corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using allylic alcohols over allyl carbonates in this synthesis?
A: Allylic alcohols are significantly cheaper, more stable, and less toxic than allyl carbonates or amines. Furthermore, this method eliminates the need for additional acidic or basic additives often required by older protocols, simplifying the workup process.
Q: Can this reaction be performed in green solvents like water?
A: Yes, the patent explicitly demonstrates that water can be used as an effective reaction solvent, achieving yields comparable to or even higher than organic solvents like methanol, which greatly enhances the environmental profile of the manufacturing process.
Q: What is the typical catalyst loading required for this transformation?
A: The process is highly efficient, operating effectively with low catalyst loadings. Optimal results are often achieved with just 2.5 mol% of the palladium dimer and 10 mol% of the dppf ligand, reducing heavy metal contamination risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Allyl Substituted Alpha-Beta Unsaturated Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and robust infrastructure. Our team of seasoned process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN108727179B can be reliably replicated on an industrial scale. We understand that high-purity pharmaceutical intermediates demand stringent purity specifications and rigorous QC labs to verify every batch against exacting standards. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of palladium-mediated reactions, including advanced metal scavenging technologies to ensure residual metal levels meet the strictest regulatory limits for drug substances.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Whether you require custom synthesis of complex intermediates or full-scale contract manufacturing, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and detailed route feasibility assessments to demonstrate how we can optimize your supply chain and reduce your overall manufacturing costs while maintaining the highest standards of quality and safety.
