Advanced Sulfonylamidine Synthesis: Scalable Technology for Global Pharmaceutical Intermediates
Advanced Sulfonylamidine Synthesis: Scalable Technology for Global Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly amidine derivatives which serve as critical scaffolds in drug discovery. A significant technological breakthrough in this domain is documented in patent CN111574413B, which details a novel preparation method for sulfonylamidine using 2-aminomethylpyridine and DMF-DMA as the amine source. This innovation addresses long-standing challenges regarding safety and scalability that have plagued conventional amidination strategies. By shifting away from hazardous reagents towards a catalytic system involving copper salts and stable amine sources, this technology offers a compelling value proposition for R&D directors focused on impurity control and procurement managers seeking cost-effective supply chains. The following analysis dissects the technical merits and commercial implications of this synthesis route for high-value fine chemical intermediates.
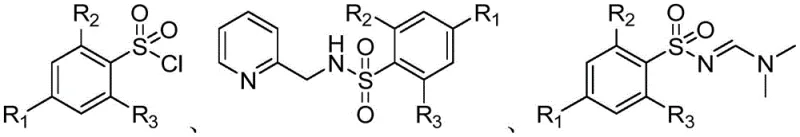
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonylamidine compounds has relied heavily on the use of diazonium salts or organic azides as the primary nitrogen source, presenting severe safety and operational bottlenecks for commercial manufacturing. Diazonium tetrafluoroborate salts, while reactive, are notoriously unstable and possess explosive potential, necessitating specialized containment facilities and rigorous safety protocols that drastically inflate capital expenditure. Furthermore, alternative routes utilizing sulfonyl azides often require copper catalysis in conjunction with carbon tetrachloride, a solvent that is increasingly restricted due to its environmental toxicity and ozone-depleting characteristics. When simple sulfonamides are employed, the process frequently demands harsh dehydrating agents like oxalyl chloride or thionyl chloride, which generate corrosive sulfur dioxide gas and require excessive stoichiometric usage to drive the reaction to completion. These legacy methods not only compromise worker safety but also create complex waste streams that challenge environmental compliance teams, making them less attractive for modern green chemistry initiatives.
The Novel Approach
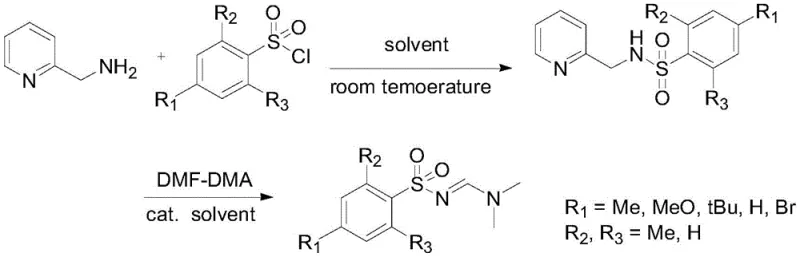
In stark contrast, the methodology outlined in the referenced patent introduces a paradigm shift by employing DMF-DMA (N,N-dimethylformamide dimethyl acetal) as a stable and manageable amine source. This approach eliminates the need for shock-sensitive precursors, thereby fundamentally altering the risk profile of the manufacturing process and allowing for operation in standard chemical reactors without explosive-proof modifications. The reaction proceeds through a distinct intermediate, N-(2-picolyl)sulfonamide, formed by the initial coupling of sulfonyl chloride with 2-aminomethylpyridine under mild basic conditions. This two-step sequence allows for the isolation and purification of the intermediate, providing a critical quality control checkpoint that ensures the final product meets stringent purity specifications before the final cyclization step. The utilization of 2-aminomethylpyridine leverages the specificity of the picolyl group to facilitate the subsequent reaction with DMF-DMA, expanding the synthetic toolbox available for generating diverse sulfonylamidine libraries with high structural fidelity.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this synthetic innovation lies in the copper-catalyzed transformation of the secondary sulfonamide intermediate into the target sulfonylamidine, a process that warrants deep mechanistic scrutiny for process chemists. The reaction utilizes copper salts, such as copper acetate monohydrate, to activate the DMF-DMA reagent, facilitating the transfer of the amidine moiety to the sulfonamide nitrogen atom. The presence of the 2-picolyl group on the nitrogen atom is not merely a protecting group but acts as a directing element that enhances the nucleophilicity and stabilizes the transition state during the cyclization event. Operating at temperatures between 60°C and 100°C in polar solvents like ethylene glycol provides the necessary thermal energy to overcome the activation barrier while maintaining a homogeneous reaction phase that ensures consistent heat transfer. This mechanistic pathway avoids the formation of radical species common in azide chemistry, resulting in a cleaner reaction profile with fewer side products and a simplified impurity spectrum that is easier to characterize and control during regulatory filings.
Controlling the impurity profile is paramount for pharmaceutical intermediates, and this route offers distinct advantages in minimizing hard-to-remove byproducts. The use of stoichiometric organic bases like triethylamine in the first step ensures complete scavenging of the hydrochloric acid byproduct, preventing acid-catalyzed decomposition of the sensitive sulfonamide linkage. In the second step, the choice of ethylene glycol as a solvent not only solubilizes the polar intermediates but also suppresses the hydrolysis of DMF-DMA, which can otherwise lead to dimethylamine impurities that are difficult to separate. The catalytic nature of the copper species means that metal residues can be kept to trace levels, especially when combined with standard aqueous workup procedures involving ethyl acetate extraction. This results in a final product with HPLC purity consistently exceeding 99%, reducing the burden on downstream purification processes such as recrystallization or column chromatography, which are often cost-prohibitive at a commercial scale.
How to Synthesize Sulfonylamidine Efficiently
Implementing this synthesis route requires precise adherence to the optimized reaction parameters to maximize yield and reproducibility across different batch sizes. The process begins with the dissolution of the sulfonyl chloride in a chlorinated solvent, followed by the controlled addition of the amine and base to manage the exotherm effectively. Following the isolation of the intermediate, the second stage involves mixing with DMF-DMA and the copper catalyst in a high-boiling alcohol solvent, where temperature control is critical to prevent solvent degradation. For detailed operational parameters, stoichiometry, and safety handling instructions, please refer to the standardized protocol below which outlines the critical process steps validated by experimental data.
- React sulfonyl chloride with 2-aminomethylpyridine in dichloromethane with triethylamine to form the N-(2-picolyl)sulfonamide intermediate.
- Mix the intermediate with DMF-DMA and copper acetate catalyst in ethylene glycol solvent.
- Heat the mixture to 60-100°C for 6-10 hours, then extract and purify to obtain high-purity sulfonylamidine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing technology offers substantial benefits that directly impact the bottom line and supply chain resilience for global buyers. By replacing hazardous and regulated reagents with stable, commodity-grade chemicals, the process reduces the regulatory burden associated with transportation and storage, leading to lower logistics costs and fewer supply disruptions. The high efficiency of the reaction minimizes raw material consumption per kilogram of output, effectively lowering the variable cost of goods sold without compromising on quality standards. Furthermore, the robustness of the chemistry allows for flexible manufacturing scheduling, as the reactions are not sensitive to minor fluctuations in ambient conditions, ensuring consistent delivery performance even in varying seasonal climates. This reliability is crucial for procurement managers who need to secure long-term contracts for active pharmaceutical ingredients without the risk of batch failures or quality deviations.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazonium salts significantly lowers the raw material cost structure while reducing the need for specialized safety infrastructure. The high yield reported in the patent data implies less waste generation and lower disposal costs, contributing to a more lean and efficient production model. Additionally, the simplified workup procedure reduces solvent consumption and energy usage during the concentration and drying phases, further driving down operational expenditures. These cumulative efficiencies allow for a more competitive pricing strategy in the market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as sulfonyl chlorides and DMF-DMA mitigates the risk of supply shortages that often plague niche reagent markets. The stability of the intermediate allows for potential stockpiling, creating a buffer against demand spikes and ensuring business continuity for downstream customers. Moreover, the process does not rely on single-source catalysts or proprietary enzymes, diversifying the supplier base and strengthening negotiation leverage. This structural resilience ensures that lead times remain predictable, supporting just-in-time manufacturing models for our partners.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, moving seamlessly from laboratory glassware to industrial reactors without the need for high-pressure equipment or cryogenic cooling. The use of ethylene glycol and ethyl acetate aligns with modern solvent selection guides, facilitating easier waste treatment and recycling compared to chlorinated or aromatic solvents used in older methods. This environmental compatibility simplifies the permitting process for new production lines and supports corporate sustainability goals. Consequently, manufacturers can ramp up production capacity rapidly to meet market demand while maintaining a strong environmental, social, and governance (ESG) profile.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled answers based on the specific data points and experimental results found in the patent literature. These responses clarify the safety profile, cost drivers, and scalability potential of the process, providing transparency for stakeholders evaluating this route for their supply chain. Understanding these nuances is essential for making informed decisions about adopting this technology for large-scale production of sulfonylamidine derivatives.
Q: Why is this sulfonylamidine synthesis method safer than traditional routes?
A: Traditional methods often rely on unstable and explosive diazonium salts or azides. This patented route utilizes stable DMF-DMA and mild copper catalysis, significantly reducing safety risks and handling hazards in industrial settings.
Q: What represents the primary cost driver in this manufacturing process?
A: The primary cost drivers are the specialized amine source (2-aminomethylpyridine) and the copper catalyst. However, the high yield and simplified workup reduce overall operational expenses compared to multi-step hazardous alternatives.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (60-100°C) and utilize common solvents like ethylene glycol and dichloromethane, making it highly adaptable for scale-up from kilogram to metric ton quantities without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylamidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of sulfonylamidine intermediate meets the exacting standards required by global regulatory agencies. Our commitment to technical excellence ensures that your projects proceed without interruption due to quality issues or supply constraints.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and enhance your competitive advantage in the marketplace.
