Advanced Triazoloquinazoline Synthesis And Commercial Scale-Up For Respiratory Drug Development
The pharmaceutical landscape for respiratory diseases continues to evolve, driven by the urgent need for safer and more effective treatments for conditions like Chronic Obstructive Pulmonary Disease (COPD) and asthma. Patent CN1352644A introduces a novel class of 1-amino triazolo[4,3-a]quinazolin-5-ones and/or 5-thiones that function as potent and selective inhibitors of phosphodiesterase 4 (PDE4). Unlike earlier generations of PDE inhibitors which suffered from low therapeutic indices and significant side effects such as nausea and vomiting, these new chemical entities offer a refined pharmacological profile. The core innovation lies in the specific substitution patterns on the triazoloquinazoline scaffold, which allow for precise modulation of enzyme affinity. As a reliable pharmaceutical intermediate supplier, understanding the depth of this intellectual property is crucial for developing next-generation anti-inflammatory agents that can overcome the limitations of current therapies like Ariflo.
![General Formula I and II of Triazolo[4,3-a]quinazolin-5-ones showing variable substituents R, X1, X2, and NR4R5](/insights/img/triazoloquinazoline-pde4-inhibitor-pharma-supplier-20260309101106-04.webp)
The development of selective PDE4 inhibitors has historically been challenged by the lack of specificity among the seven major families of cyclic nucleotide phosphodiesterases. Conventional methods often relied on methylxanthines like theophylline, which inhibit multiple PDE families indiscriminately, leading to systemic toxicity and narrow therapeutic windows. These non-specific compounds interfere with various cellular functions beyond the target tissue, causing adverse cardiovascular and gastrointestinal events. Furthermore, earlier attempts at selective inhibition often failed to distinguish sufficiently between PDE4 subtypes or cross-reacted with PDE3 and PDE5, limiting their clinical utility in chronic inflammatory conditions. The novel approach detailed in the patent addresses these deficiencies by engineering a rigid heterocyclic core that fits specifically into the catalytic site of PDE4, thereby minimizing off-target interactions and enhancing the safety profile for long-term administration in patients with severe respiratory compromise.
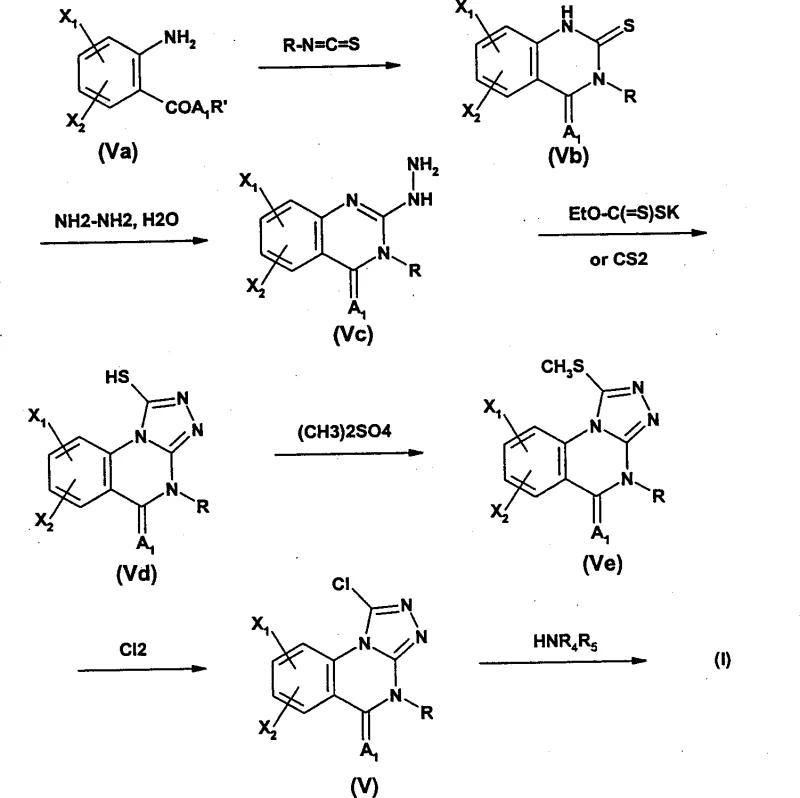
The novel synthetic strategy employs a convergent pathway that builds the triazolo[4,3-a]quinazoline skeleton through a series of robust and scalable reactions. The process typically begins with the cyclization of appropriately substituted anthranilic acid esters with isothiocyanates to form 2-thioquinazolin-4-ones, which are then converted to hydrazino intermediates. A critical step involves the ring closure to form the triazole moiety using reagents like potassium xanthate, followed by functionalization at the nitrogen atoms. This modular approach allows for the independent variation of substituents at the 1, 4, and 7 positions, enabling rapid structure-activity relationship (SAR) studies. By decoupling the synthesis of the core from the introduction of diverse side chains, manufacturers can optimize yields and purity at each stage, ensuring a consistent supply of high-quality intermediates suitable for further drug development.
Mechanistically, the inhibitory activity of these compounds is governed by three distinct interaction points within the PDE4 catalytic site. The first point involves the aromatic ring containing substituents X1 and X2, where the presence of a halogen or methyl group at the 7-position significantly enhances binding affinity and selectivity compared to unsubstituted or 8-substituted analogs. The second interaction point is the substituent R at the 4-position, which occupies a large hydrophobic cavity within the enzyme; variations here, such as cinnamyl or pyridylalkyl groups, allow for fine-tuning of lipophilicity and bioavailability without compromising potency. The third point is the amino group NR4R5 at the 1-position, where cyclic amines like pyrrolidine or azepane provide optimal steric fit. This multi-point attachment model explains why specific combinations of substituents result in nanomolar IC50 values, offering a clear roadmap for medicinal chemists to design derivatives with improved pharmacokinetic properties and reduced emetic potential.
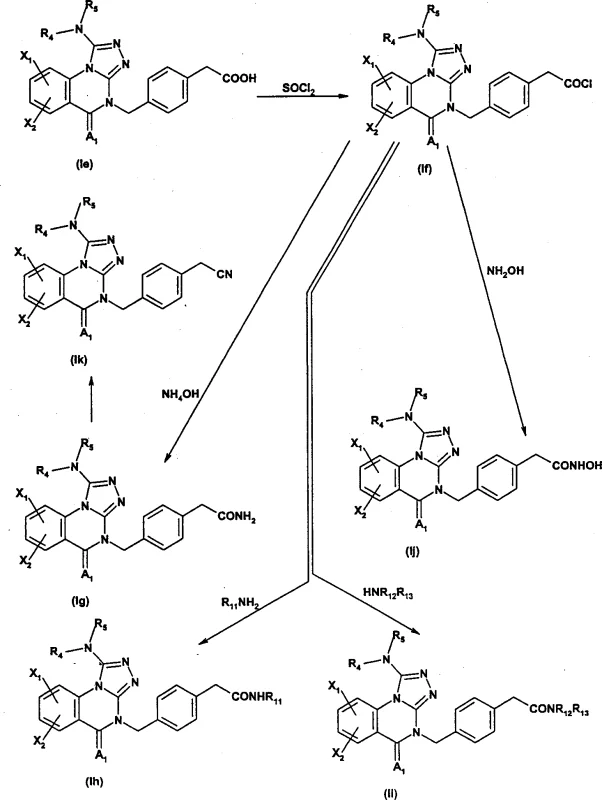
Impurity control is paramount in the synthesis of complex heterocycles intended for pharmaceutical use, and the described routes incorporate several purification checkpoints to ensure high purity specifications. The use of crystallization steps, particularly after the formation of key intermediates like the 1-bromo or 1-methylthio derivatives, effectively removes regioisomers and unreacted starting materials. Additionally, the final alkylation or amination steps are conducted under controlled basic conditions to minimize the formation of N-alkylation byproducts at unintended sites. The patent emphasizes the separation of N3 and N4 regioisomers via chromatography when necessary, although optimized conditions often favor the desired isomer. This rigorous attention to impurity profiles ensures that the resulting active pharmaceutical ingredients (APIs) meet stringent regulatory standards, reducing the risk of toxicological issues during preclinical and clinical evaluation phases.
How to Synthesize Triazoloquinazoline PDE4 Inhibitors Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and minimize waste. The patent outlines multiple pathways, with Synthetic Route 2 being particularly advantageous for large-scale production due to its use of commodity chemicals and mild reaction conditions. The process involves the initial formation of a quinazolinone core, followed by hydrazine displacement and triazole ring closure. Subsequent functionalization allows for the introduction of diverse pharmacophores. Detailed operational procedures, including solvent choices like dimethylformamide or 1,2-dimethoxyethane and specific temperature ranges for reflux, are critical for reproducibility. For teams looking to implement this chemistry, adhering to the specified stoichiometry and workup protocols is essential to achieve the reported purity levels.
- Cyclize substituted anthranilic acid esters with alkyl isothiocyanates to form 2-thioquinazolin-4-ones.
- Treat the thioquinazolinone with hydrazine hydrate to generate the 2-hydrazino intermediate.
- Cyclize the hydrazino derivative using potassium xanthate or carbon disulfide to form the triazolo ring system, followed by alkylation or amination.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic routes described in patent CN1352644A offer significant advantages in terms of raw material availability and process robustness. The starting materials, such as substituted anthranilic acids and various amines, are widely available from global chemical suppliers, reducing the risk of supply chain bottlenecks. The avoidance of precious metal catalysts or exotic reagents means that the cost of goods sold (COGS) can be kept competitive, making these intermediates attractive for generic drug development or new chemical entity (NCE) programs. Furthermore, the convergent nature of the synthesis allows for parallel processing of different fragments, which can drastically reduce overall lead time for high-purity pharmaceutical intermediates. This flexibility enables manufacturers to respond quickly to changing demand without compromising on quality or delivery schedules.
- Cost Reduction in Manufacturing: The synthetic methodology eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions, relying instead on classical organic transformations that are well-understood and cost-effective. By utilizing standard reagents like hydrazine hydrate and alkyl halides, the process avoids the high costs associated with specialized catalytic systems and their subsequent removal. This simplification of the chemical process translates directly into substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more budget-efficient allocation of resources towards downstream formulation and clinical trials. Additionally, the high atom economy of the cyclization steps minimizes waste generation, further contributing to economic efficiency.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals ensures a stable and resilient supply chain, as these materials are produced by multiple vendors worldwide, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, means that production can be easily transferred between different manufacturing sites without significant re-validation efforts. This geographical flexibility is crucial for maintaining continuity of supply in the face of global logistical disruptions. Moreover, the stability of the intermediates allows for stockpiling if necessary, providing an additional buffer against market volatility and ensuring that project timelines remain on track regardless of external pressures.
- Scalability and Environmental Compliance: The processes described are inherently scalable, having been demonstrated from gram to multi-gram scales in the patent examples, with clear pathways to tonnage production. The use of common solvents like ethanol, methanol, and dichloromethane facilitates easy recovery and recycling, aligning with modern green chemistry principles and environmental regulations. The absence of heavy metals simplifies waste treatment and disposal, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards not only avoids potential regulatory fines but also enhances the corporate social responsibility profile of the supply chain, making it more appealing to ethically conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazoloquinazoline derivatives. Understanding these aspects is vital for stakeholders involved in the sourcing and development of PDE4 inhibitors. The answers are derived directly from the technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What is the primary therapeutic application of these triazoloquinazoline derivatives?
A: These compounds act as selective inhibitors of phosphodiesterase 4 (PDE4), making them highly relevant for treating inflammatory conditions such as Chronic Obstructive Pulmonary Disease (COPD), asthma, and allergic rhinitis by increasing intracellular cAMP levels.
Q: How does the synthetic route ensure scalability for commercial production?
A: The patent describes convergent synthetic routes using readily available starting materials like anthranilic acid esters and isothiocyanates. The processes utilize standard reagents and avoid exotic catalysts, facilitating straightforward scale-up from laboratory to multi-ton manufacturing.
Q: What structural features contribute to PDE4 selectivity in these molecules?
A: Selectivity is driven by specific substituents at the 7-position (such as halogens or methyl groups) and the nature of the amine group at the 1-position. The patent highlights that 7-substituted compounds show higher selectivity for PDE4 over other isoforms like PDE3 and PDE5.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazoloquinazoline Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at navigating the complexities of heterocyclic chemistry, ensuring that every batch of triazoloquinazoline intermediates meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of respiratory drug development and are committed to providing a seamless supply chain solution that supports your R&D milestones. Our facility is equipped to handle the specific reaction conditions required for these syntheses, guaranteeing consistency and quality that aligns with global regulatory expectations.
We invite you to engage with our technical procurement team to discuss your specific requirements for Customized Cost-Saving Analysis. By collaborating with us, you can access specific COA data and route feasibility assessments tailored to your project needs. Whether you require small quantities for preclinical studies or large volumes for commercial launch, our infrastructure is designed to support your growth. Contact us today to explore how our expertise in PDE4 inhibitor intermediates can accelerate your drug development pipeline and bring innovative respiratory therapies to patients faster.
