Advanced Catalytic Oxidation for Commercial 2,5-Diformylfuran Production and Supply
The global shift towards sustainable chemical manufacturing has placed biomass-derived platform compounds at the forefront of industrial research and development. Among these, the selective oxidation of 5-hydroxymethylfurfural (5-HMF) to 2,5-diformylfuran (DFF) represents a critical transformation for producing high-value pharmaceutical intermediates and polymer monomers. Patent CN112851605B introduces a groundbreaking methodology that addresses the longstanding challenges of catalyst recovery and selectivity in this oxidation process. By utilizing a novel four-component molybdenum-based compound, Co9Fe3BiMo12O51, this technology enables the efficient conversion of biomass feedstocks under relatively mild conditions using molecular oxygen. For R&D directors and procurement specialists seeking a reliable 2,5-diformylfuran supplier, understanding the mechanistic advantages of this patent is essential for evaluating long-term supply chain stability and cost reduction in fine chemical manufacturing. This report analyzes the technical merits of this catalytic system and its implications for commercial scale-up of complex biomass intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran has been plagued by significant technical and environmental hurdles that hinder industrial adoption. Traditional methods often rely on stoichiometric amounts of harsh chemical oxidants, which generate substantial quantities of toxic waste streams and require complex downstream purification processes to remove residual oxidizing agents. Furthermore, many existing catalytic systems are homogeneous in nature, meaning the catalyst exists in the same phase as the reactants, which creates a severe bottleneck in catalyst recovery and reuse. This inability to efficiently separate the catalyst not only drives up the cost of goods sold due to catalyst loss but also introduces risks of metal contamination in the final high-purity 2,5-diformylfuran product, which is unacceptable for pharmaceutical applications. Additionally, conventional heterogeneous systems have frequently suffered from low raw material conversion rates and poor product selectivity, leading to the formation of unwanted over-oxidation byproducts like 2,5-furandicarboxylic acid, thereby complicating the isolation of the target dialdehyde.
The Novel Approach
The methodology disclosed in patent CN112851605B offers a transformative solution by employing a heterogeneous Co9Fe3BiMo12O51 catalyst that operates effectively with molecular oxygen as the terminal oxidant. This approach fundamentally alters the economic and environmental profile of the synthesis by eliminating the need for stoichiometric oxidants and facilitating straightforward catalyst recovery through simple centrifugation. The use of oxygen, a clean and abundant oxidant, ensures that the only byproduct is water, drastically simplifying waste management and aligning with green chemistry principles required by modern regulatory frameworks. Moreover, the specific composition of the four-component molybdenum-based compound provides exceptional stability and activity, allowing the reaction to proceed at atmospheric pressure and moderate temperatures between 120°C and 140°C. This robustness translates directly into reducing lead time for high-purity furan derivatives, as the process avoids the high-pressure equipment and extensive safety protocols associated with more aggressive oxidation methods.
Mechanistic Insights into Co9Fe3BiMo12O51-Catalyzed Selective Oxidation
The core innovation of this technology lies in the unique electronic and structural properties of the Co9Fe3BiMo12O51 catalyst, which facilitates the selective activation of the hydroxymethyl group on the furan ring while preserving the aldehyde functionality. The synergistic interaction between cobalt, iron, bismuth, and molybdenum creates active sites that efficiently adsorb oxygen and transfer it to the substrate, promoting the dehydrogenation of the alcohol moiety to the aldehyde without triggering further oxidation to the carboxylic acid. This high selectivity is critical for maintaining the integrity of the 2,5-diformylfuran structure, which serves as a versatile building block for macrocyclic ligands and antifungal agents. The heterogeneous nature of the catalyst ensures that the active sites remain accessible throughout the reaction cycle, while the solid support prevents the leaching of metal ions into the reaction medium, a common failure mode in less stable catalytic systems.
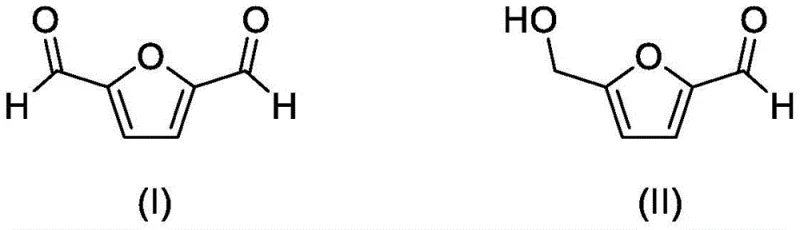
Impurity control is another paramount aspect of this mechanistic design, particularly for clients requiring stringent purity specifications for pharmaceutical intermediates. The catalyst's ability to operate in dimethyl sulfoxide (DMSO) provides a stable solvent environment that solubilizes the biomass-derived 5-HMF effectively while maintaining the catalyst's structural integrity. By minimizing side reactions such as polymerization or ring-opening of the furan moiety, the process ensures a clean reaction profile that simplifies downstream crystallization or distillation steps. The patent data indicates that conversion rates can reach up to 99% with product yields approaching 98%, demonstrating that the mechanistic pathway is highly efficient and reproducible. For supply chain heads, this level of consistency is vital for ensuring supply continuity, as it reduces the variability often associated with biocatalytic or less robust chemical processes.
How to Synthesize 2,5-Diformylfuran Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the efficiency of the Co9Fe3BiMo12O51 catalyst. The process begins with the preparation of the reaction mixture, where the mass ratio of 5-hydroxymethylfurfural to the catalyst is optimized between 1:0.25 and 1:0.8 to ensure sufficient active sites are available without excessive catalyst loading. The reaction is conducted in DMSO, with the solvent volume ranging from 10 to 50 times the mass of the substrate, providing adequate fluidity for mass transfer under heating conditions. Once the mixture is prepared, the system is purged with oxygen to establish an oxidative atmosphere, and the temperature is raised to the 120-140°C range for a duration of 8 to 12 hours. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 5-hydroxymethylfurfural and the Co9Fe3BiMo12O51 catalyst in dimethyl sulfoxide (DMSO) solvent.
- Replace the atmosphere with oxygen and heat the mixture to 120-140°C under normal pressure for 8-12 hours.
- Recover the heterogeneous catalyst via centrifugation, wash with water and ethanol, and dry for reuse while isolating the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic oxidation technology offers substantial strategic benefits for procurement managers and supply chain leaders focused on cost optimization and risk mitigation. The primary advantage lies in the significant cost savings achieved through the elimination of expensive stoichiometric oxidants and the ability to recycle the heterogeneous catalyst multiple times without regeneration. This circular usage of the catalyst drastically reduces the raw material cost per kilogram of product, making the process economically competitive against traditional petrochemical routes. Furthermore, the use of oxygen as the oxidant removes the logistical burden and safety costs associated with transporting and storing hazardous chemical oxidants, thereby streamlining the manufacturing workflow and reducing insurance and compliance overheads.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the reusability of the Co9Fe3BiMo12O51 catalyst, which can be recovered via centrifugation, washed, and dried for subsequent batches. This capability eliminates the recurring cost of purchasing fresh catalyst for every production run, leading to a drastic simplification of the cost structure. Additionally, the high selectivity of the reaction minimizes the loss of valuable 5-HMF feedstock to byproducts, ensuring that the maximum amount of raw material is converted into saleable 2,5-diformylfuran. By avoiding the need for complex purification steps to remove metal contaminants or over-oxidized acids, the downstream processing costs are also significantly reduced, contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions, which operate at atmospheric pressure and moderate temperatures, reducing the risk of equipment failure or safety incidents that could halt production. The catalyst's stability over multiple cycles ensures that production schedules are not disrupted by the need for frequent catalyst synthesis or procurement, providing a predictable and steady output of material. Moreover, the reliance on oxygen and DMSO, which are commodity chemicals with stable global supply chains, mitigates the risk of raw material shortages that often plague specialty chemical manufacturing. This reliability makes the technology an ideal candidate for long-term supply agreements with pharmaceutical and polymer companies.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the heterogeneous nature of the catalyst, which is compatible with standard industrial reactors and filtration equipment. The environmental profile of the process is exceptionally clean, as the use of oxygen generates water as the only byproduct, eliminating the need for costly waste treatment facilities to handle toxic oxidation residues. This alignment with green chemistry standards not only reduces environmental compliance costs but also enhances the brand value of the final product for eco-conscious downstream customers. The ability to scale from 100 kgs to 100 MT annual commercial production without fundamental changes to the chemistry ensures that the technology can grow with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology, based on the specific data and claims found in the patent literature. These answers are designed to provide clarity on the operational parameters and the strategic benefits of adopting this catalytic system for industrial production. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using the Co9Fe3BiMo12O51 catalyst over homogeneous systems?
A: The heterogeneous Co9Fe3BiMo12O51 catalyst allows for easy separation via centrifugation and can be reused multiple times without significant loss of activity, solving the recovery issues common in homogeneous catalytic systems.
Q: What are the optimal reaction conditions for high yield conversion?
A: The patent specifies reacting 5-hydroxymethylfurfural with the catalyst in DMSO at temperatures between 120°C and 140°C under an oxygen atmosphere for 8 to 12 hours to achieve conversion rates up to 99%.
Q: Is this oxidation method environmentally friendly compared to traditional methods?
A: Yes, by utilizing molecular oxygen as the oxidant instead of stoichiometric chemical oxidants, the process significantly reduces chemical waste and avoids the environmental hazards associated with traditional oxidation byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diformylfuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Co9Fe3BiMo12O51 catalytic system for producing high-value biomass intermediates like 2,5-diformylfuran. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory technologies are successfully translated into robust industrial processes. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2,5-diformylfuran meets the exacting standards required for pharmaceutical and specialty polymer applications. We are committed to leveraging this patent technology to deliver consistent quality and supply stability to our global partners.
We invite you to collaborate with us to optimize your supply chain for biomass-derived chemicals. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your R&D and commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
