Advanced Palladium-Catalyzed Synthesis of Heteroaryl Alkynes for Pharmaceutical Intermediates
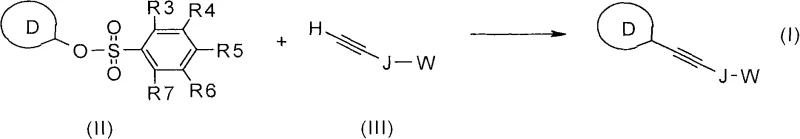
The pharmaceutical and agrochemical industries are constantly seeking more efficient and cost-effective routes to synthesize complex heteroaryl scaffolds, which serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN101687796A introduces a groundbreaking methodology for the regioselective synthesis of substituted heteroaryl-1-alkynes, utilizing a palladium-catalyzed coupling reaction between heteroaryl tosylates and terminal alkynes. This innovation addresses long-standing challenges in cross-coupling chemistry by replacing traditional, moisture-sensitive, and expensive leaving groups with robust tosylate derivatives. By leveraging this technology, manufacturers can achieve superior process stability and economic efficiency, positioning this method as a cornerstone for the reliable production of high-purity pharmaceutical intermediates. The versatility of this approach allows for the incorporation of diverse functional groups, making it an indispensable tool for modern medicinal chemistry and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of heteroaryl alkynes has relied heavily on Sonogashira coupling reactions employing heteroaryl halides or triflates as electrophilic partners. While effective, heteroaryl iodides and bromides often present significant supply chain bottlenecks due to their higher cost and the generation of stoichiometric amounts of heavy metal waste during the reaction. Furthermore, heteroaryl triflates, although highly reactive, suffer from severe stability issues; they are extremely sensitive to moisture and require the use of hazardous and expensive reagents like triflic anhydride (Tf2O) for their preparation. This sensitivity necessitates stringent anhydrous conditions and specialized handling equipment, which drastically increases operational complexity and capital expenditure. Additionally, the purification of triflate intermediates can be cumbersome, often requiring extensive chromatographic separation to remove acidic byproducts, thereby reducing overall process throughput and yield.
The Novel Approach
The methodology described in CN101687796A revolutionizes this landscape by utilizing heteroaryl tosylates and benzenesulfonates as the electrophilic coupling partners. Unlike triflates, tosylates are chemically robust, exhibiting excellent stability against hydrolysis even in the presence of protic solvents, which simplifies reaction conditions and work-up procedures. They are typically obtained as highly crystalline solids, facilitating easy purification through recrystallization rather than complex chromatography. From a procurement perspective, the precursors for tosylates, such as tosyl chloride (TosCl) or tosic anhydride (Tos2O), are commodity chemicals that are significantly cheaper and safer to handle on a large scale than triflating agents. This shift not only enhances the safety profile of the manufacturing process but also delivers substantial cost savings by eliminating the need for exotic reagents and reducing waste disposal costs associated with unstable intermediates.
Mechanistic Insights into Palladium-Catalyzed Tosylate Activation
The success of this transformation hinges on the ability of the palladium catalyst to activate the carbon-oxygen bond of the tosylate group, which is inherently stronger and less reactive than the carbon-halogen bonds found in traditional substrates. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the C-O bond of the heteroaryl tosylate. This step is kinetically challenging and requires the use of specialized, bulky, and electron-rich phosphine ligands, such as 1-[2-(dicyclohexylphosphino)ferrocenyl]ethyldi-tert-butylphosphine (Cy-PF-t-Bu). These ligands create a sterically congested environment around the metal center that promotes the dissociation of the tosylate leaving group and stabilizes the resulting organopalladium intermediate. Following oxidative addition, the terminal alkyne undergoes transmetallation, a process facilitated by the presence of a base like potassium phosphate or carbonate, which deprotonates the alkyne to form a reactive copper or palladium acetylide species in situ.
Impurity control is a critical aspect of this mechanism, particularly regarding the suppression of homocoupling side reactions (Glaser coupling) of the terminal alkyne. The choice of a protic solvent, specifically tert-butanol as highlighted in the patent examples, plays a dual role: it solubilizes the inorganic base and the organic substrates while simultaneously suppressing the formation of diyne byproducts by moderating the reactivity of the alkyne species. Furthermore, the use of heterogeneous or soluble palladium sources that do not aggregate into inactive palladium black ensures a consistent catalytic turnover number. The final reductive elimination step releases the desired heteroaryl alkyne product and regenerates the active palladium(0) catalyst, completing the cycle. This mechanistic understanding allows process chemists to fine-tune ligand-to-metal ratios and base equivalents to maximize yield and minimize the formation of desulfonylated byproducts.
How to Synthesize Heteroaryl Alkynes Efficiently
Implementing this synthesis route requires careful attention to the selection of catalyst systems and reaction parameters to ensure optimal conversion. The general procedure involves charging a reaction vessel with the heteroaryl tosylate substrate, a palladium source such as palladium(II) trifluoroacetate, and the specialized ferrocenyl phosphine ligand under an inert atmosphere. A mild inorganic base, typically potassium phosphate, is added along with a protic solvent like tert-butanol to create a homogeneous reaction mixture. Upon the addition of the terminal alkyne, the system is heated to temperatures ranging from 60°C to 150°C, with 85°C being a preferred setpoint for balancing reaction rate and selectivity.
- Preparation of the reaction mixture by combining heteroaryl tosylate, palladium catalyst (e.g., Pd(TFA)2), bulky phosphine ligand, and inorganic base in a protic solvent.
- Addition of the terminal alkyne substrate under an inert atmosphere and heating the mixture to temperatures between 60°C and 150°C.
- Work-up involving filtration through diatomaceous earth, solvent evaporation, and purification via silica gel chromatography to isolate the target heteroaryl alkyne.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tosylate-based coupling technology offers transformative benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material supply chain; tosyl chlorides and phenols are globally available commodity chemicals with stable pricing, unlike halide precursors which can be subject to market volatility and supply shortages. This stability ensures a continuous flow of materials for long-term production campaigns, mitigating the risk of line stoppages due to missing key starting materials. Moreover, the enhanced stability of the tosylate intermediates allows for extended storage periods without degradation, enabling manufacturers to build strategic inventory buffers without the fear of material spoilage that plagues moisture-sensitive triflates.
- Cost Reduction in Manufacturing: The economic impact of switching to tosylates is profound, driven primarily by the elimination of expensive triflating reagents and the reduction in waste treatment costs. Since tosylates are prepared from cheap precursors and do not require the rigorous anhydrous conditions needed for triflates, the utility consumption for drying solvents and maintaining inert atmospheres is significantly lowered. Additionally, the crystalline nature of many tosylate intermediates allows for purification via simple recrystallization rather than resource-intensive column chromatography, leading to substantial savings in silica gel and solvent usage. This streamlined downstream processing directly translates to a lower cost of goods sold (COGS) and improved profit margins for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the tosylate functionality enhances the overall resilience of the supply chain by reducing the complexity of logistics and storage. Unlike reactive halides or triflates that may require cold chain shipping or specialized containment, tosylates can often be transported and stored under ambient conditions. This flexibility reduces freight costs and simplifies warehouse management. Furthermore, the compatibility of this method with a wide range of functional groups means that a single standardized protocol can be applied to synthesize a diverse library of intermediates, reducing the need for multiple specialized production lines and allowing for greater agility in responding to changing customer demands for different API precursors.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with green chemistry principles. The use of protic solvents like tert-butanol is preferable to toxic aprotic solvents often used in traditional couplings, simplifying solvent recovery and recycling. The reaction generates benign sulfonate salts as byproducts, which are easier to treat in wastewater facilities compared to heavy metal halide wastes. The method has been demonstrated to work efficiently on various scales, indicating strong potential for seamless scale-up from laboratory benchtop to multi-ton commercial production without the need for significant process re-engineering, ensuring a reliable supply of high-purity products for global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed coupling technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the long-term viability of the supply chain.
Q: Why are heteroaryl tosylates preferred over triflates in this synthesis?
A: Heteroaryl tosylates are significantly more stable against hydrolysis compared to triflates, are easier to purify as they often form crystalline solids, and are derived from cheaper reagents like TosCl or Tos2O, reducing overall raw material costs.
Q: What catalyst system is recommended for activating the tosylate bond?
A: The patent suggests using palladium sources like palladium(II) trifluoroacetate combined with bulky, electron-rich phosphine ligands such as 1-[2-(dicyclohexylphosphino)ferrocenyl]ethyldi-tert-butylphosphine to facilitate the oxidative addition step.
Q: Can this method be scaled for industrial production?
A: Yes, the use of stable solid tosylates and standard protic solvents like tert-butanol simplifies handling and safety protocols, making the process highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heteroaryl Tosylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in the development of next-generation therapeutics and agrochemicals. Our team of expert process chemists has extensively validated the tosylate coupling strategy outlined in CN101687796A, ensuring that we can deliver these complex intermediates with the highest levels of purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities and rigorous QC labs to guarantee that every batch meets stringent purity specifications. Our commitment to quality assurance means that we can support your projects from early-stage clinical supply through to full-scale commercial manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced technology. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how switching to tosylate-based routes can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis requirements. Let us be your partner in driving innovation and efficiency in your chemical supply chain.
