Advanced Synthesis of (R)-p-Hydroxyphenylglycine for High-Purity Antibiotic Intermediates
Advanced Synthesis of (R)-p-Hydroxyphenylglycine for High-Purity Antibiotic Intermediates
The pharmaceutical industry continuously demands more efficient and sustainable pathways for producing critical chiral intermediates, particularly those serving as side chains for semi-synthetic beta-lactam antibiotics. Patent CN103524366A introduces a groundbreaking synthesis process for (R)-p-hydroxyphenylglycine, a pivotal building block in the manufacture of broad-spectrum antibiotics. This innovative methodology addresses long-standing challenges in yield optimization and environmental impact by leveraging a sophisticated recycling mechanism for stereoisomers. Unlike conventional routes that discard significant portions of unwanted isomers, this process integrates a dynamic racemization step that converts the less desirable S-enantiomer back into the valuable R-form. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for securing a stable and cost-effective supply chain.
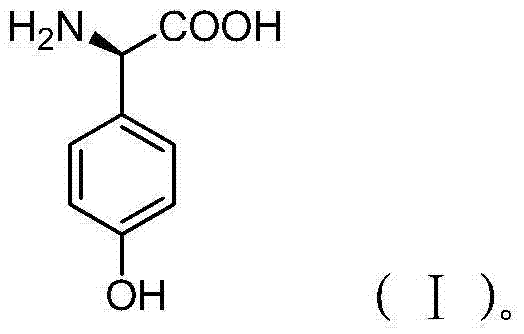
The core innovation lies in the strategic combination of acid-catalyzed condensation followed by a dual-stage chiral resolution. By utilizing phenol and 2-hydroxyglycine as primary feedstocks, the process bypasses the toxic and wasteful reagents associated with older technologies. The resulting molecular architecture, as depicted in the structural analysis, features a critical chiral center that determines the biological activity of the final antibiotic product. Achieving high optical purity (>99% ee) is not merely a regulatory requirement but a fundamental necessity for drug efficacy and safety. This patent provides a robust framework for achieving such purity while maintaining economic viability through intelligent process design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (R)-p-hydroxyphenylglycine has relied heavily on the condensation of phenol, glyoxylic acid, and thioacetamide. While this traditional pathway is technically mature, it suffers from inherent inefficiencies that plague modern manufacturing efforts. The most significant drawback is the relatively low yield, typically hovering between 61% and 65%, which necessitates the processing of larger volumes of raw materials to achieve the same output. Furthermore, the separation of by-products in this route is notoriously difficult, leading to complex purification steps that drive up refining costs and extend production timelines. From an environmental perspective, the generation of substantial amounts of industrial wastewater containing sulfur and nitrogen residues creates immense pressure on waste treatment facilities. These factors combined result in a process that is not only economically burdensome but also increasingly unsustainable in the face of tightening global environmental regulations.
The Novel Approach
In stark contrast, the novel approach detailed in CN103524366A revolutionizes the synthesis landscape by introducing a recycling loop for the S-isomer. Instead of discarding the S-enantiomer obtained during the initial resolution, the process subjects it to a specialized racemization treatment using salicylaldehyde as a catalyst. This allows the S-isomer to revert to a racemic state, from which the desired R-isomer can be extracted once again through a secondary resolution step. This closed-loop system dramatically improves the overall atom economy and significantly boosts the final yield of the target compound. Additionally, the use of 2-hydroxyglycine as a starting material simplifies the reaction profile, reducing the formation of complex impurities and facilitating easier downstream processing. For procurement managers focused on cost reduction in antibiotic manufacturing, this shift represents a move towards a leaner, more resource-efficient production model.
Mechanistic Insights into Acid-Catalyzed Condensation and Dynamic Resolution
The chemical foundation of this synthesis rests on a precise acid-catalyzed condensation reaction between phenol and 2-hydroxyglycine in an organic solvent medium. Sulfuric acid serves as the proton donor, activating the electrophilic centers and facilitating the nucleophilic attack by the phenol ring. The choice of solvent, such as toluene, chlorobenzene, or dichloroethane, plays a critical role in controlling the reaction kinetics and ensuring the formation of the racemic D-pHPG intermediate with high efficiency. Following the condensation, the introduction of a chiral resolving agent, such as (+)-phenylethane sulfonic acid or camphorsulfonic acid, triggers the formation of diastereomeric salts. Due to the differences in solubility between the R-salt and S-salt complexes, the R-isomer preferentially precipitates out of the solution, allowing for physical separation via filtration. This primary resolution step is meticulously controlled by temperature and molar ratios to maximize the recovery of the R-enantiomer while minimizing co-precipitation of the S-form.
The true brilliance of the mechanism, however, is revealed in the treatment of the mother liquor containing the S-isomer. By adjusting the pH and introducing a catalytic amount of salicylaldehyde, the process induces a dynamic racemization of the S-enantiomer. Salicylaldehyde likely forms a Schiff base intermediate with the amino group, labilizing the alpha-proton and allowing for stereochemical inversion. Once racemized, the mixture undergoes a second resolution with additional resolving agent, effectively harvesting the R-isomer that was previously locked in the S-form. This iterative resolution strategy ensures that the theoretical yield limit of 50% inherent in classical resolutions is surpassed, pushing practical yields well above 80%. For technical teams evaluating high-purity chiral intermediates, this mechanism offers a reliable pathway to consistent optical purity without the need for expensive enzymatic or chromatographic separations.
How to Synthesize (R)-p-Hydroxyphenylglycine Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and stoichiometry to replicate the high yields reported in the patent data. The process begins with the condensation of phenol and 2-hydroxyglycine in a selected organic solvent under acidic conditions, followed by a work-up involving water addition and phase separation. The subsequent resolution steps demand precise temperature control, typically between 70°C and 110°C, to ensure optimal crystallization of the diastereomeric salts. The recycling of the S-isomer filtrate is the key differentiator, requiring the addition of alkali to precipitate the free amino acid before subjecting it to the racemization catalyst. Detailed operational parameters, including specific molar ratios of resolving agents and pH adjustments, are critical for success. The standardized synthesis steps outlined below provide a comprehensive guide for scaling this technology from the laboratory to pilot production.
- Condense phenol and 2-hydroxyglycine in an organic solvent with sulfuric acid to form racemic D-pHPG.
- Perform primary chiral resolution using a resolving agent to isolate the R-isomer composite salt.
- Racemize the S-isomer filtrate using salicylaldehyde and perform secondary resolution to recover additional R-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical superiority. The ability to recycle the S-isomer directly translates to a substantial reduction in raw material consumption per kilogram of finished product, effectively lowering the variable cost of goods sold. By eliminating the need for complex byproduct removal and reducing the volume of wastewater generated, the process also mitigates environmental compliance costs and reduces the risk of production stoppages due to waste disposal limits. Furthermore, the reliance on commodity chemicals like phenol and sulfuric acid ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents. This robustness is essential for maintaining commercial scale-up of complex amino acids without encountering bottlenecks associated with specialized feedstock sourcing.
- Cost Reduction in Manufacturing: The implementation of the S-isomer recycling loop fundamentally alters the cost structure of production by maximizing the utility of every mole of starting material. Traditional methods discard nearly half of the synthesized material as the unwanted enantiomer, whereas this process recovers a significant portion of it, thereby drastically reducing the effective cost per unit of the active pharmaceutical ingredient. Additionally, the simplified purification steps reduce the consumption of solvents and energy required for distillation and recrystallization. These cumulative efficiencies result in a leaner manufacturing process that enhances profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing stability is a paramount concern for global pharmaceutical manufacturers, and this process leverages widely available bulk chemicals to mitigate supply risks. Phenol and 2-hydroxyglycine are produced on a massive industrial scale, ensuring that fluctuations in price or availability are minimal compared to niche chiral reagents. The operational simplicity of the process, which relies on standard filtration and neutralization techniques rather than specialized equipment, further ensures that production can be easily transferred between different manufacturing sites. This flexibility allows for reducing lead time for high-purity intermediates by enabling rapid ramp-up of production capacity in response to market demand.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a critical factor in vendor selection. This method generates significantly less hazardous waste compared to thioacetamide-based routes, simplifying the permitting process and reducing the liability associated with effluent treatment. The absence of heavy metals or toxic sulfur byproducts in the final waste stream aligns with green chemistry principles, making it an attractive option for companies aiming to improve their sustainability metrics. The straightforward nature of the unit operations ensures that the process can be scaled from hundreds of kilograms to multi-tonne annual capacities with minimal engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their specific supply chain needs. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for industrial application.
Q: How does this process improve yield compared to traditional glyoxylic acid routes?
A: Traditional methods using glyoxylic acid and thioacetamide often suffer from low yields (around 61-65%) and difficult byproduct separation. This novel process utilizes a recycling strategy where the unwanted S-isomer is racemized and re-resolved, significantly boosting the overall yield of the target R-isomer to over 80%.
Q: What is the specific role of salicylaldehyde in the synthesis?
A: Salicylaldehyde acts as a crucial racemization catalyst in the second stage of the process. It facilitates the conversion of the separated S-isomer back into a racemic mixture, allowing for a second round of chiral resolution that recovers valuable R-isomer material that would otherwise be wasted.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It employs readily available raw materials like phenol and 2-hydroxyglycine, uses common organic solvents, and relies on standard unit operations such as filtration and phase separation, making it highly adaptable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-p-Hydroxyphenylglycine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development and production of life-saving antibiotics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (R)-p-hydroxyphenylglycine adheres to the highest international standards. Our facility is equipped to handle the specific solvent systems and resolution conditions required by this advanced patent, providing our partners with a secure and consistent source of supply.
We invite pharmaceutical manufacturers and procurement teams to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in process optimization, we can help you identify further opportunities for efficiency gains within your supply chain. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Partnering with us means gaining access to a dedicated support system focused on driving your project forward with speed and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
