Advanced Synthesis of Spiro Indoline Phenanthroline Oxazine for Commercial Photochromic Applications
Advanced Synthesis of Spiro Indoline Phenanthroline Oxazine for Commercial Photochromic Applications
The rapid evolution of optoelectronic materials has driven an intense demand for high-performance photochromic compounds that offer superior durability and reversible switching capabilities. Patent CN1300146C introduces a groundbreaking class of spiro[indoline-phenanthroline oxazine] compounds that address the critical limitations of earlier generations of photochromic dyes. Unlike traditional naphtho-spirooxazines which often suffer from limited fatigue resistance, these novel derivatives utilize a rigid phenanthroline backbone to enhance thermal stability and cycling endurance. This technological leap is particularly significant for industries requiring long-term reliability, such as optical data storage, smart windows, and advanced security inks. By leveraging substituted 1,10-phenanthroline as a key raw material, the disclosed method enables the precise tuning of photochromic properties through structural modification, offering a versatile platform for next-generation functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic photochromic materials has been dominated by indoline spironaphthooxazines and spiroquinooxazines, which, while effective, present inherent challenges in commercial scalability and performance longevity. Conventional synthesis routes for these legacy compounds often involve complex multi-step sequences that generate significant chemical waste and require rigorous purification to remove trace impurities that can act as quenchers for the photochromic effect. Furthermore, the molecular architecture of naphthalene-based spirooxazines can be susceptible to photodegradation after repeated cycling, leading to a phenomenon known as fatigue where the material loses its ability to revert to its original state. This degradation limits their utility in high-value applications like rewritable optical disks or permanent protective coatings where thousands of switching cycles are required without loss of fidelity. Additionally, the solubility profiles of older derivatives often restrict their compatibility with common polymer matrices used in industrial coating formulations.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by replacing the naphthalene core with a substituted 1,10-phenanthroline moiety, fundamentally altering the electronic and steric environment of the spiro center. This structural innovation not only improves the thermal stability of the closed-ring form but also facilitates a cleaner synthetic pathway that relies on the condensation of readily available precursors. ![General synthetic route for spiro[indoline-phenanthroline oxazine] showing condensation of phenanthroline oxime and indole salt](/insights/img/spiro-indoline-phenanthroline-oxazine-supplier-photochromic-20260308113810-02.webp) As illustrated in the reaction scheme, the process bypasses the need for harsh transition metal catalysts often found in cross-coupling reactions, instead utilizing straightforward acid-base chemistry and thermal activation. The resulting compounds demonstrate exceptional fatigue resistance, capable of undergoing repeated quality-color killing circulation without tangible photolysis. This robustness is complemented by a simplified purification protocol involving recrystallization from lower boiling alcohols, which significantly reduces downstream processing costs and enhances the overall economic viability of the manufacturing process for bulk production.
As illustrated in the reaction scheme, the process bypasses the need for harsh transition metal catalysts often found in cross-coupling reactions, instead utilizing straightforward acid-base chemistry and thermal activation. The resulting compounds demonstrate exceptional fatigue resistance, capable of undergoing repeated quality-color killing circulation without tangible photolysis. This robustness is complemented by a simplified purification protocol involving recrystallization from lower boiling alcohols, which significantly reduces downstream processing costs and enhances the overall economic viability of the manufacturing process for bulk production.
Mechanistic Insights into Photochromic Ring-Opening and Closing
The photochromic behavior of these spiro[indoline-phenanthroline oxazine] derivatives is governed by a reversible electrocyclic reaction that alters the conjugation length of the molecule upon irradiation. 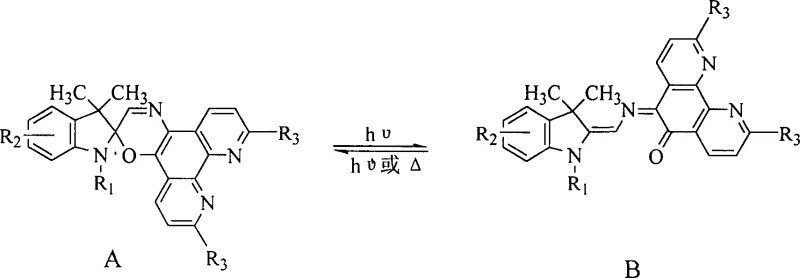 In the ground state, the molecule exists as a colorless spiro compound (Form A), characterized by an orthogonal arrangement of the indoline and oxazine planes which prevents electronic conjugation across the spiro carbon. Upon exposure to UV light of a specific wavelength, the C-O bond in the oxazine ring undergoes heterolytic cleavage, leading to a planar merocyanine structure (Form B) that exhibits strong visible light absorption and distinct coloration. This open form is stabilized by the extended pi-system of the phenanthroline unit, which delocalizes the charge separation effectively. The reversal process, returning the molecule to its colorless state, can be triggered thermally or by irradiation with visible light of a different wavelength, allowing for precise control over the optical density of the material. Understanding this mechanistic equilibrium is crucial for R&D teams aiming to optimize response times and contrast ratios for specific device applications.
In the ground state, the molecule exists as a colorless spiro compound (Form A), characterized by an orthogonal arrangement of the indoline and oxazine planes which prevents electronic conjugation across the spiro carbon. Upon exposure to UV light of a specific wavelength, the C-O bond in the oxazine ring undergoes heterolytic cleavage, leading to a planar merocyanine structure (Form B) that exhibits strong visible light absorption and distinct coloration. This open form is stabilized by the extended pi-system of the phenanthroline unit, which delocalizes the charge separation effectively. The reversal process, returning the molecule to its colorless state, can be triggered thermally or by irradiation with visible light of a different wavelength, allowing for precise control over the optical density of the material. Understanding this mechanistic equilibrium is crucial for R&D teams aiming to optimize response times and contrast ratios for specific device applications.
Impurity control is paramount in maintaining the efficiency of this photochromic cycle, as side products can interfere with the ring-opening kinetics or act as permanent traps for the excited state. The patent emphasizes the importance of using high-purity starting materials, specifically the substituted 1,10-phenanthroline-5,6-diketone monoxime hydrochloride, to minimize the formation of non-photochromic byproducts. The choice of organic base during the condensation step also plays a critical role; bases such as triethylamine or piperidine facilitate the deprotonation necessary for cyclization without promoting unwanted side reactions like polymerization or decomposition of the sensitive oxime intermediate. By strictly controlling the molar ratios—typically maintaining a slight excess of the indole iodide salt (1:1 to 1:1.5)—the process ensures complete consumption of the limiting reagent, thereby simplifying the isolation of the target spiro compound and reducing the burden on final purification steps.
How to Synthesize Spiro[indoline-phenanthroline oxazine] Efficiently
The synthesis of these high-value photochromic intermediates follows a logical progression from commodity chemicals to specialized functional materials, beginning with the oxidation of substituted 1,10-phenanthroline. The initial step involves treating the phenanthroline precursor with sodium bromide and concentrated nitric acid in sulfuric acid to generate the 5,6-diketone intermediate, which is subsequently converted to the monoxime hydrochloride salt through reaction with hydroxylamine hydrochloride. This oxime derivative serves as the electrophilic partner in the final condensation reaction with various substituted indole iodide salts. The versatility of this approach allows for the introduction of diverse functional groups at the R1, R2, and R3 positions, enabling chemists to fine-tune the solubility, melting point, and absorption maxima of the final product. Detailed operational parameters, including specific temperature ramps and solvent choices, are critical for maximizing yield and ensuring batch-to-batch consistency in a commercial setting.
- Prepare substituted 1,10-phenanthroline-5,6-diketone monoxime hydrochloride through oxidation and oximation of substituted 1,10-phenanthroline.
- Condense the phenanthroline oxime derivative with a substituted indole iodide salt in the presence of an organic base such as triethylamine or piperidine.
- Reflux the mixture at 80-100°C for 15-24 hours, then purify the resulting spiro compound via filtration and recrystallization using lower boiling alcohols.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers substantial strategic benefits rooted in process simplicity and raw material accessibility. The elimination of complex catalytic systems and the reliance on standard organic solvents like ethanol significantly lowers the barrier to entry for manufacturing, reducing both capital expenditure on specialized equipment and operational expenditures related to solvent recovery and waste disposal. The robust nature of the reaction conditions, which tolerate reflux temperatures between 80°C and 100°C, ensures that the process can be safely scaled from laboratory benchtop quantities to multi-tonne industrial reactors without encountering the exothermic runaway risks associated with more volatile chemistries. Furthermore, the high yields reported for specific derivatives, such as the 96% yield achieved for the benzo-fused variant (SP7), indicate a highly atom-economical process that minimizes raw material waste and maximizes output per batch.
- Cost Reduction in Manufacturing: The streamlined purification protocol described in the patent eliminates the need for expensive chromatographic separations in many cases, relying instead on simple filtration and recrystallization techniques that are easily automated in large-scale production facilities. By avoiding the use of precious metal catalysts, the process removes the cost volatility associated with commodities like palladium or platinum, leading to a more predictable and stable cost of goods sold. The ability to recover and recycle the alcohol solvents used in the reaction further contributes to long-term cost savings, aligning with green chemistry principles that are increasingly mandated by global regulatory bodies. This economic efficiency makes the material highly competitive for price-sensitive markets such as consumer eyewear and packaging security features.
- Enhanced Supply Chain Reliability: The starting materials, including substituted 1,10-phenanthrolines and indole derivatives, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues specialty chemical supply chains. The synthetic route's tolerance for a variety of substituents means that if a specific raw material faces a shortage, alternative analogues can often be substituted with minimal re-optimization of the process, ensuring continuity of supply for downstream customers. Additionally, the solid-state stability of the intermediate oxime hydrochloride allows for it to be stockpiled as a buffer inventory, decoupling the production of the final spiro compound from the immediate availability of the phenanthroline precursor and smoothing out production scheduling.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous byproducts, primarily consisting of inorganic salts that can be treated through standard wastewater protocols, simplifying environmental compliance and reducing the cost of effluent treatment. The absence of heavy metals in the final product is a significant advantage for applications in consumer goods and electronics, where strict limits on substances like lead, cadmium, and mercury are enforced. The high thermal stability of the final spiro compounds also reduces the risk of degradation during transport and storage, lowering insurance costs and minimizing product returns due to quality issues. This combination of safety, scalability, and environmental friendliness positions the technology as a sustainable choice for long-term industrial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochromic technology, derived directly from the experimental data and background analysis of the patent documentation. These insights are intended to assist technical decision-makers in evaluating the feasibility of integrating these materials into their existing product lines or R&D pipelines. Understanding the specific performance metrics and processing requirements is essential for successful technology transfer and commercialization.
Q: What are the primary advantages of spiro[indoline-phenanthroline oxazine] over traditional naphtho-spirooxazines?
A: According to patent CN1300146C, these novel compounds exhibit significantly improved thermal stability and superior fatigue resistance, allowing for repeated color-change cycles without tangible photolysis, which is critical for long-life optical storage and protective coatings.
Q: What are the typical reaction conditions for synthesizing these photochromic materials?
A: The synthesis involves a condensation reaction between substituted 1,10-phenanthroline-5,6-diketone monoxime hydrochloride and substituted indole iodide salts. The process typically requires refluxing in alcohol solvents at temperatures between 80°C and 100°C for 15 to 24 hours in the presence of organic bases.
Q: Can the photochromic properties be tuned for specific applications?
A: Yes, the patent describes varying substituents (R1, R2, R3) on the indole and phenanthroline rings. For instance, introducing a fused benzene ring (as in compound SP7) can drastically increase yield to near-quantitative levels (96%) while maintaining excellent photochromic reversibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro[indoline-phenanthroline oxazine] Supplier
As the global demand for advanced photochromic materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a critical competitive edge in bringing these innovations to market. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency regardless of volume. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the photochromic performance and chemical integrity of every batch before shipment. Our team of process chemists is adept at optimizing the condensation and purification steps described in the patent to maximize yield and minimize impurity profiles, delivering a product that meets the highest industry standards.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific application requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new derivatives, we are ready to provide the support you need. Contact us today to request samples and discover how our expertise in fine chemical synthesis can accelerate your product development timeline and enhance your market position.
