Scalable Synthesis of Polycyclic 2-Amino-Dihydrothiazole Systems for Commercial API Production
Scalable Synthesis of Polycyclic 2-Amino-Dihydrothiazole Systems for Commercial API Production
The pharmaceutical landscape continuously demands innovative scaffolds that offer potent biological activity alongside manufacturability. Patent CN1341107A introduces a sophisticated class of polycyclic 2-amino-dihydrothiazole systems, specifically designed to function as effective anorectic agents and lipid metabolism regulators. These compounds, characterized by their fused ring structures incorporating nitrogen and sulfur heteroatoms, represent a significant advancement over prior art thiazolidine derivatives. The structural versatility allowed by the various substituents (R1 through R4) enables fine-tuning of pharmacokinetic properties, making them ideal candidates for next-generation anti-obesity therapeutics. For R&D directors and procurement specialists, understanding the synthetic accessibility of these complex molecules is paramount for securing a reliable supply chain.
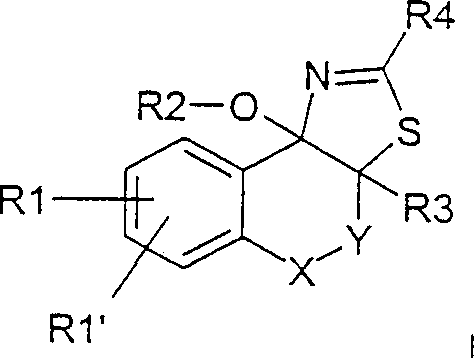
This patent delineates a robust methodology for constructing these polycyclic cores, moving beyond simple substitution to create rigid, stereochemically defined architectures. The core innovation lies in the efficient cyclization strategy that builds the thiazole ring directly onto a pre-functionalized carbocyclic framework. By leveraging activated ketone intermediates, the process avoids harsh conditions that often degrade sensitive functional groups. This approach not only enhances the overall yield but also simplifies the impurity profile, a critical factor for regulatory approval in pharmaceutical manufacturing. The ability to produce these compounds in high purity with scalable chemistry positions them as valuable assets for companies targeting metabolic disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of thiazole-containing pharmaceuticals often relies on multi-step sequences involving unstable intermediates or expensive transition metal catalysts. Conventional routes to similar anorectic agents frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, many legacy processes utilize hazardous reagents or require extreme temperatures that pose safety risks during commercial scale-up. The reliance on chromatographic purification for final products in older methods significantly drives up manufacturing costs and extends lead times, creating bottlenecks in the supply chain. These inefficiencies make it challenging to meet the rigorous quality standards and volume demands of the global pharmaceutical market.
The Novel Approach
The methodology described in CN1341107A offers a streamlined alternative by utilizing a direct cyclization between an activated cyclic ketone and a thiourea derivative. This novel approach eliminates the need for precious metal catalysts, relying instead on fundamental organic transformations that are well-understood and easily controlled. The reaction conditions are mild, typically proceeding between 0°C and 50°C, which minimizes energy consumption and reduces the formation of thermal degradation byproducts. By forming the acid addition salt directly during the reaction, the process facilitates easy isolation through precipitation, bypassing the need for extensive chromatographic purification. This efficiency translates directly into lower production costs and a more sustainable manufacturing footprint.
Mechanistic Insights into Activated Ketone Cyclization
The core of this synthetic strategy involves the transformation of a cyclic ketone (Formula II) into an activated species (Formula III), typically an alpha-halo ketone or an activated ester. This activation step renders the alpha-carbon highly electrophilic, priming it for nucleophilic attack by the sulfur atom of the thiourea (Formula IV). The subsequent intramolecular cyclization closes the thiazole ring, establishing the polycyclic framework with high fidelity. The reaction mechanism is driven by the thermodynamic stability of the resulting aromatic thiazole system, which acts as a driving force for the conversion. Understanding this mechanistic pathway allows chemists to optimize reaction parameters such as solvent polarity and molar ratios to maximize throughput.
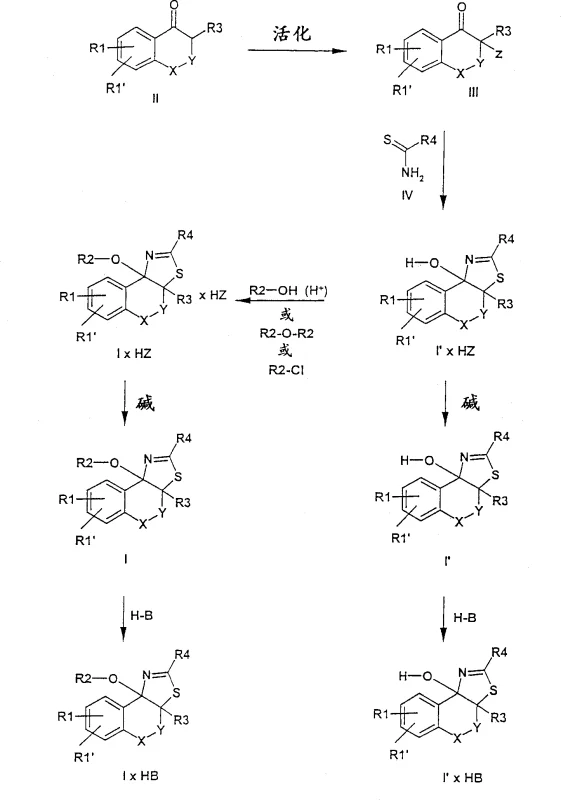
Impurity control is inherently built into this mechanism due to the specificity of the nucleophilic substitution. Since the reaction proceeds in a virtually quantitative manner under optimized conditions, the generation of side products is minimized. The use of inert solvents like acetone, ethyl acetate, or short-chain alcohols ensures that the reactants remain in solution while the product precipitates out as a salt, effectively self-purifying the mixture. Any remaining impurities can be removed through simple washing or recrystallization steps. This high level of chemical selectivity ensures that the final active pharmaceutical ingredient meets stringent purity specifications without requiring resource-intensive downstream processing, thereby enhancing the overall economic viability of the process.
How to Synthesize Polycyclic 2-Amino-Dihydrothiazole Efficiently
The synthesis of these high-value intermediates follows a logical progression from readily available starting materials to the final purified product. The process begins with the selection of the appropriate cyclic ketone precursor, which determines the substitution pattern on the final aromatic ring. Following activation, the coupling with the thiourea component is performed under controlled thermal conditions to ensure complete conversion. The detailed operational parameters, including specific solvent choices and workup procedures, are critical for reproducing the high yields reported in the patent literature. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol below.
- Activate the cyclic ketone precursor (Formula II) using halogenation or esterification to form the activated intermediate (Formula III).
- React the activated intermediate with a substituted thiourea (Formula IV) in a polar organic solvent at controlled temperatures between 0°C and 50°C.
- Isolate the resulting acid addition salt or convert to the free base using organic or inorganic bases, followed by purification via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost stability and operational reliability. The elimination of expensive transition metal catalysts removes a significant variable cost and mitigates the risk associated with heavy metal residue testing. Furthermore, the reagents required, such as cyclic ketones and thioureas, are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. The simplicity of the workup procedure, which often involves filtration of the precipitated salt, reduces the demand for specialized equipment and skilled labor, further driving down operational expenditures.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by avoiding the use of precious metal catalysts and complex purification technologies like preparative HPLC. The reaction proceeds with high atom economy, meaning less raw material is wasted as byproduct, which directly lowers the cost of goods sold. Additionally, the ability to isolate the product as a salt through simple precipitation reduces solvent consumption and energy usage associated with distillation and drying. These factors combine to create a highly economical manufacturing process that remains competitive even at large commercial scales.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and standard organic solvents, this manufacturing route minimizes the risk of supply disruptions. The chemistry does not rely on exotic reagents that might have long lead times or restricted export controls. The robustness of the reaction conditions allows for flexible production scheduling, as the process is tolerant to minor variations in temperature and mixing rates. This flexibility ensures that manufacturers can maintain consistent output levels to meet fluctuating market demands without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been designed to function efficiently from gram to kilogram scales without fundamental changes in the reaction mechanism. The use of common solvents like ethyl acetate and acetone simplifies waste management and solvent recovery, aligning with modern environmental compliance standards. The reduction in hazardous waste generation and the avoidance of toxic heavy metals contribute to a greener manufacturing profile, which is increasingly important for corporate sustainability goals and regulatory approvals in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polycyclic thiazole systems. These insights are derived directly from the patented technology and are intended to clarify the feasibility of integrating this chemistry into existing pharmaceutical pipelines. Understanding these details helps stakeholders make informed decisions regarding licensing, sourcing, and development strategies for new anorectic drug candidates.
Q: What are the primary applications of polycyclic 2-amino-dihydrothiazole compounds?
A: These compounds exhibit significant anorectic effects and beneficial impacts on lipid metabolism, making them highly suitable for the development of anti-obesity medications and treatments for Type II diabetes.
Q: How does this synthesis route improve supply chain reliability?
A: The process utilizes readily available cyclic ketone starting materials and common thiourea reagents, avoiding reliance on scarce precious metal catalysts, which ensures consistent raw material availability and stable pricing.
Q: What purity levels can be achieved with this manufacturing method?
A: The reaction proceeds in a virtually quantitative manner, often yielding analytically pure crude products that require minimal purification, typically achievable through simple recrystallization or precipitation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic 2-Amino-Dihydrothiazole Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the lab to the market. Our technical team is adept at optimizing complex heterocyclic syntheses to meet stringent purity specifications required for clinical and commercial phases. With our rigorous QC labs and state-of-the-art manufacturing facilities, we guarantee the consistency and quality of every batch of polycyclic 2-amino-dihydrothiazole intermediates we supply. We are committed to being a strategic partner who understands the critical nature of your supply chain.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial goals. Let us help you secure a stable, high-quality supply of these vital pharmaceutical intermediates for your next generation of metabolic therapies.
