Advanced Enzymatic Synthesis of Moxifloxacin Intermediates for Commercial Scale-Up
Advanced Enzymatic Synthesis of Moxifloxacin Intermediates for Commercial Scale-Up
The global demand for third-generation quinolone antibiotics continues to drive innovation in the synthesis of key chiral intermediates. Moxifloxacin, a broad-spectrum antibacterial agent widely used for respiratory tract infections, relies heavily on the availability of high-purity chiral building blocks. Specifically, the synthesis of (4aS,7aS)-octahydro-1H-pyrrolo[3,4-b]pyridine represents a critical bottleneck in the supply chain due to the complexity of establishing two chiral centers simultaneously. 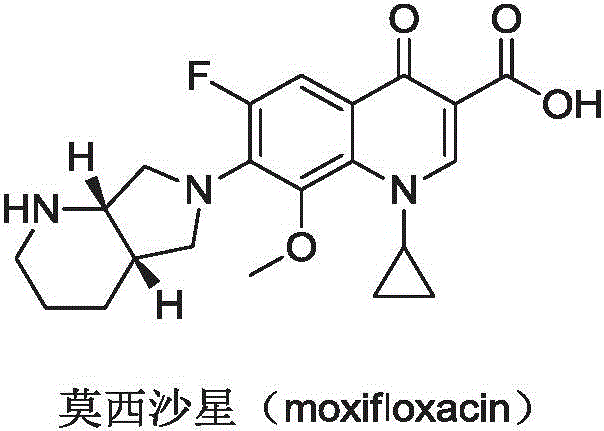 Recent advancements documented in Chinese Patent CN107686852B introduce a groundbreaking biocatalytic approach that addresses these challenges. This patent discloses a preparation method utilizing omega-transaminases to convert ketone precursors directly into the desired chiral amine intermediates. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology offers a compelling alternative to traditional chemical synthesis, promising enhanced stereocontrol and operational simplicity.
Recent advancements documented in Chinese Patent CN107686852B introduce a groundbreaking biocatalytic approach that addresses these challenges. This patent discloses a preparation method utilizing omega-transaminases to convert ketone precursors directly into the desired chiral amine intermediates. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology offers a compelling alternative to traditional chemical synthesis, promising enhanced stereocontrol and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of the moxifloxacin intermediate (4aS,7aS)-octahydro-1H-pyrrolo[3,4-b]pyridine has been dominated by two primary synthetic strategies, both of which present significant logistical and economic hurdles. The first strategy, often referred to as the piperidine route, typically initiates from 3,4-pyridinedicarboxylic acid. This pathway necessitates high-pressure hydrogenation of the pyridine ring, a process that demands specialized, expensive reactor equipment capable of withstanding extreme conditions. Furthermore, the reduction of carboxylic acid carbonyls requires costly reducing agents, and the establishment of chirality often relies on resolution methods. These resolution processes generate substantial amounts of unwanted isomers, creating waste disposal issues and requiring complex recycling protocols to recover the resolving agents. 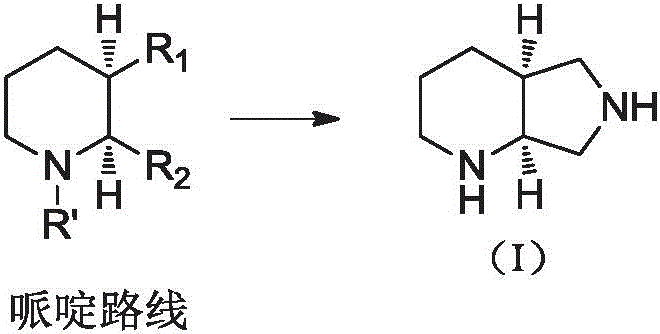 The second strategy, the pyrrolidine route, frequently employs Sharpless asymmetric epoxidation to construct the chiral skeleton. While chemically elegant, this multi-step sequence involves over eleven distinct reaction stages, including desymmetrization, ring-opening, and ring-closing steps. The cumulative yield loss across such a long sequence, combined with the high cost of chiral reagents and the need for rigorous purification at each stage, renders this approach economically unviable for large-scale commercial manufacturing.
The second strategy, the pyrrolidine route, frequently employs Sharpless asymmetric epoxidation to construct the chiral skeleton. While chemically elegant, this multi-step sequence involves over eleven distinct reaction stages, including desymmetrization, ring-opening, and ring-closing steps. The cumulative yield loss across such a long sequence, combined with the high cost of chiral reagents and the need for rigorous purification at each stage, renders this approach economically unviable for large-scale commercial manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in Patent CN107686852B leverages the power of biocatalysis to streamline the synthesis into a concise, efficient workflow. The core innovation lies in the direct transamination of a pyrrolidinone derivative, specifically compound (III), to form the protected chiral amine compound (II). 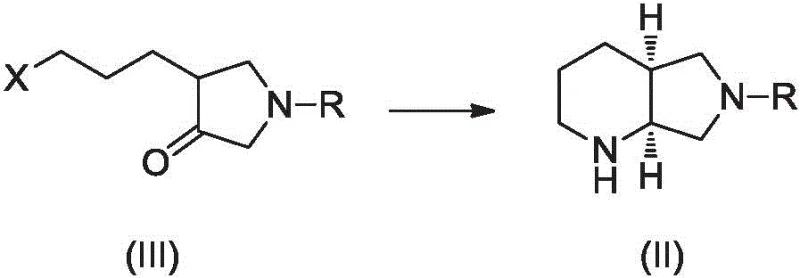 This single enzymatic step effectively establishes the requisite stereochemistry with exceptional precision, bypassing the need for external chiral auxiliaries or resolution steps. The process operates under mild conditions, typically in aqueous or mixed solvent systems at temperatures ranging from 20°C to 55°C, which drastically reduces energy consumption compared to high-temperature or high-pressure chemical alternatives. By condensing what was previously a multi-step chemical sequence into a single biocatalytic transformation followed by a straightforward deprotection, this method significantly shortens the overall process timeline. For supply chain managers, this reduction in unit operations translates directly to reduced lead times and a lower risk of batch failure, ensuring a more robust supply of high-purity pharmaceutical intermediates.
This single enzymatic step effectively establishes the requisite stereochemistry with exceptional precision, bypassing the need for external chiral auxiliaries or resolution steps. The process operates under mild conditions, typically in aqueous or mixed solvent systems at temperatures ranging from 20°C to 55°C, which drastically reduces energy consumption compared to high-temperature or high-pressure chemical alternatives. By condensing what was previously a multi-step chemical sequence into a single biocatalytic transformation followed by a straightforward deprotection, this method significantly shortens the overall process timeline. For supply chain managers, this reduction in unit operations translates directly to reduced lead times and a lower risk of batch failure, ensuring a more robust supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Omega-Transaminase Catalyzed Transamination
The success of this synthetic route hinges on the specific activity and stereoselectivity of the omega-transaminase employed. The patent specifies the use of enzymes with amino acid sequences corresponding to SEQ ID NO.1, SEQ ID NO.2, or SEQ ID NO.3. These biocatalysts facilitate the transfer of an amino group from an ammonia donor, such as isopropylamine or D-alanine, to the ketone substrate (III). The reaction mechanism involves the formation of a Schiff base intermediate between the enzyme's pyridoxal phosphate (PLP) cofactor and the substrate. This transient species allows for the precise delivery of the amino group to the pro-chiral carbon center, enforcing the formation of the (4aS,7aS) configuration. The enzyme's active site is sterically constrained to reject the formation of the undesired enantiomer, which is why the process consistently achieves ee values exceeding 99%. This level of stereocontrol is difficult to replicate with small-molecule catalysts without extensive optimization.
Furthermore, the reaction environment is carefully tuned to maximize enzyme stability and turnover. The process is conducted at a controlled pH of 8.0 to 10.0, maintained through the addition of buffers or bases like ammonia water. The choice of solvent is also critical; while water is the primary medium, co-solvents such as DMSO or isobutyl acetate are utilized to enhance the solubility of the organic substrate (III) without denaturing the protein structure. The patent highlights the option of using immobilized forms of the omega-transaminase. Immobilization not only protects the enzyme from thermal degradation but also facilitates its recovery from the reaction mixture via simple filtration. This capability allows the biocatalyst to be recycled for multiple batches, a feature that is instrumental in driving down the cost of goods sold (COGS) for the final intermediate. The combination of high specificity, mild reaction conditions, and catalyst reusability creates a robust platform for the sustainable manufacturing of complex chiral amines.
How to Synthesize (4aS,7aS)-octahydro-1H-pyrrolo[3,4-b]pyridine Efficiently
The implementation of this biocatalytic route requires careful attention to reaction parameters to ensure optimal yield and purity. The process begins with the preparation of the reaction mixture, where the ketone precursor (III) is dissolved in a suitable solvent system, often a mixture of water and a water-miscible organic solvent to balance solubility and enzyme activity. An ammonia donor is added in stoichiometric excess to drive the equilibrium towards product formation, as transamination reactions are reversible. The pH is adjusted to the alkaline range (8.0-10.0) prior to the introduction of the enzyme and the PLP cofactor. Once the biocatalyst is introduced, the reaction proceeds at moderate temperatures (e.g., 25-50°C) for a duration of 12 to 24 hours. Monitoring is typically performed via HPLC or TLC to confirm the complete consumption of the starting material. Following the reaction, a workup procedure involving pH swings and solvent extraction isolates the protected intermediate (II), which is then subjected to deprotection to yield the final target molecule (I).
- Perform transamination of compound (III) using omega-transaminase (SEQ ID NO.1/2/3) and an ammonia donor in a solvent system at pH 8.0-10.0.
- Isolate the protected intermediate (II) through pH adjustment, extraction, and purification techniques such as column chromatography.
- Execute deprotection of compound (II) via acid hydrolysis or hydrogenolysis to yield the final free base intermediate (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional chemical synthesis to this enzymatic methodology offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing infrastructure. By eliminating the need for high-pressure hydrogenation reactors and cryogenic conditions required for some asymmetric chemical reactions, facilities can utilize standard stainless steel vessels, thereby lowering capital expenditure (CAPEX) barriers for production. This flexibility allows for a more diversified supplier base, as more contract manufacturing organizations (CMOs) possess the capability to run ambient pressure biocatalytic processes compared to those equipped for high-risk hydrogenation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in raw material costs and waste generation. Traditional routes often require expensive chiral resolving agents or precious metal catalysts which are lost or difficult to recover. In this enzymatic route, the use of immobilized enzymes allows for catalyst recycling, effectively amortizing the cost of the biocatalyst over multiple production runs. Additionally, the high stereoselectivity (>99% ee) eliminates the yield losses associated with discarding unwanted isomers during resolution steps. This atom economy ensures that a higher percentage of the starting material ends up in the final product, directly lowering the cost per kilogram of the active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the enzymatic process. Chemical routes involving sensitive reagents like organometallics or high-pressure gases are prone to disruptions due to safety incidents or regulatory inspections. Biocatalytic processes operate under safer, ambient conditions, reducing the risk of unplanned shutdowns. Furthermore, the shorter synthetic sequence reduces the number of intermediate hand-offs and quality control checkpoints. Fewer steps mean fewer opportunities for deviation, resulting in more predictable lead times and a steadier flow of materials to downstream API manufacturers. This reliability is crucial for maintaining continuous production schedules for essential antibiotics like moxifloxacin.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles. The avoidance of heavy metal catalysts removes the burden of extensive metal scavenging and validation steps required to meet strict residual metal limits in pharmaceuticals. The use of aqueous solvent systems reduces the volume of volatile organic compounds (VOCs) emitted during production. Waste streams are predominantly biological or saline in nature, which are easier and cheaper to treat compared to the toxic heavy metal waste generated by traditional chemical reductions. This environmental profile not only reduces waste disposal costs but also future-proofs the supply chain against tightening environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the stereochemical purity achieved in this enzymatic route?
A: The patented process utilizing specific omega-transaminases achieves an enantiomeric excess (ee) value of greater than 99%, ensuring high optical purity critical for API synthesis.
Q: How does this method improve supply chain reliability compared to traditional routes?
A: By eliminating high-pressure hydrogenation and complex chiral resolution steps, the process reduces equipment dependency and operational complexity, leading to more consistent production cycles.
Q: Can the enzyme be reused to lower manufacturing costs?
A: Yes, the patent describes the use of immobilized omega-transaminase forms, which enhance stability and allow for separation and repeated use, significantly driving down catalyst costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moxifloxacin Intermediate Supplier
The technological breakthroughs described in Patent CN107686852B represent a significant leap forward in the manufacturing of chiral quinolone intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methods into robust, industrial-scale processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle both traditional chemical synthesis and advanced biocatalytic transformations, ensuring that we can adapt to the specific needs of your project. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this efficient enzymatic route for your moxifloxacin supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this technology can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of this critical pharmaceutical intermediate.
