Advanced Nickel-Catalyzed Cyanation for Scalable Aryl Nitrile Manufacturing
Advanced Nickel-Catalyzed Cyanation for Scalable Aryl Nitrile Manufacturing
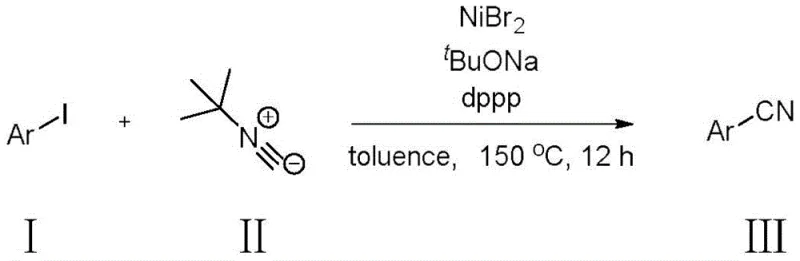
The landscape of organic synthesis is constantly evolving towards safer, more sustainable, and cost-effective methodologies, particularly in the production of high-value intermediates. A pivotal advancement in this domain is detailed in patent CN114213204A, which discloses a novel method for synthesizing aryl nitrile compounds utilizing tert-butyl isonitrile as a cyanating agent under nickel catalysis. This technology represents a significant departure from traditional reliance on hazardous cyanide sources, offering a pathway that aligns with modern green chemistry principles while maintaining high synthetic efficiency. For R&D directors and procurement strategists alike, this innovation addresses critical pain points regarding operator safety, raw material costs, and supply chain stability. By leveraging earth-abundant nickel catalysts and low-toxicity reagents, this process not only enhances the safety profile of nitrile synthesis but also opens new avenues for the economical production of complex pharmaceutical and agrochemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon-cyanide bond has been fraught with significant challenges, primarily stemming from the extreme toxicity and volatility of traditional cyanating reagents. Conventional industrial processes often rely on hydrogen cyanide (HCN), trimethylsilyl cyanide (TMSCN), or soluble metal cyanides such as potassium cyanide (KCN) and sodium cyanide (NaCN). These substances pose severe occupational health risks, requiring specialized containment infrastructure and rigorous safety protocols that drastically inflate operational expenditures. Furthermore, alternative non-metallic sources like K4[Fe(CN)6], while less toxic, suffer from poor solubility in low-polarity organic solvents, necessitating the use of high-boiling, high-polarity solvents that complicate downstream purification and energy-intensive solvent recovery. Additionally, many established methods depend on precious metal catalysts like palladium or rhodium, the prices of which are subject to volatile market fluctuations and geopolitical supply constraints, creating uncertainty for long-term production planning.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN114213204A introduces a paradigm shift by employing tert-butyl isonitrile as a stable, low-toxicity cyanogen source coupled with an inexpensive nickel catalyst system. This approach effectively circumvents the acute toxicity hazards associated with free cyanide ions, thereby reducing the regulatory burden and safety infrastructure costs required for manufacturing. The use of nickel bromide (NiBr2) as the catalyst precursor offers a compelling economic advantage over palladium-based systems, significantly lowering the cost of goods sold (COGS) for the final aryl nitrile product. Moreover, the reaction conditions are remarkably robust, utilizing common organic solvents like toluene and standard phosphine ligands, which simplifies the logistical complexity of raw material sourcing. This novel route not only ensures a safer working environment for production personnel but also delivers a streamlined process that is inherently more amenable to scale-up in a commercial GMP setting.
Mechanistic Insights into Ni-Catalyzed Cyanation
The core of this technological breakthrough lies in the efficient nickel-catalyzed cross-coupling mechanism that facilitates the formation of the C-CN bond. The catalytic cycle typically initiates with the oxidative addition of the aryl iodide substrate to the active nickel(0) species, generated in situ from the nickel(II) precatalyst and the phosphine ligand. This step is critical as it activates the relatively inert carbon-iodine bond, setting the stage for subsequent transmetallation or ligand exchange. Unlike palladium cycles which can be sluggish with certain sterically hindered substrates, the nickel center exhibits unique electronic properties that allow for rapid turnover even with diverse aryl iodides. The tert-butyl isonitrile then coordinates to the metal center, serving as the cyanide donor. Through a series of migratory insertion and reductive elimination steps, the aryl group and the cyano moiety are coupled to release the desired aryl nitrile product, regenerating the active nickel catalyst to continue the cycle. This mechanistic pathway is highly efficient, minimizing the formation of homocoupling byproducts that often plague transition metal-catalyzed reactions.
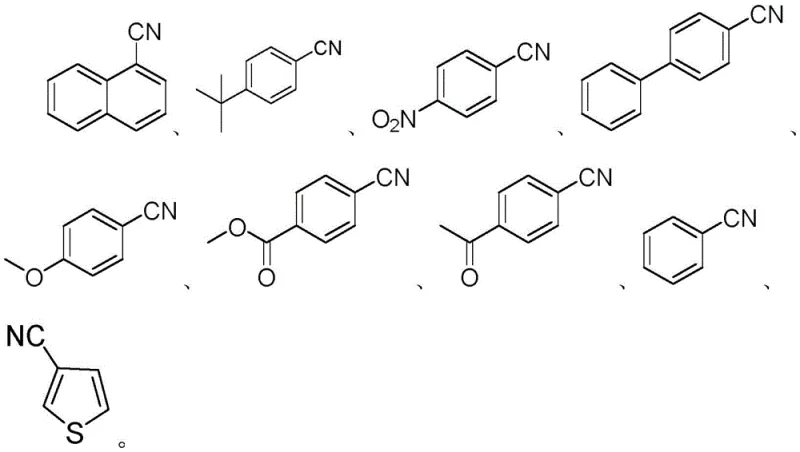
Furthermore, the choice of ligands and bases plays a pivotal role in controlling the impurity profile and ensuring high product purity, a key concern for pharmaceutical applications. The patent specifies the use of bidentate phosphine ligands such as 1,3-bis(diphenylphosphino)propane (dppp), which stabilize the nickel center and prevent catalyst decomposition at the elevated reaction temperatures of 150°C. The presence of a strong base like sodium tert-butoxide is essential for facilitating the activation of the isonitrile and neutralizing the hydrogen iodide byproduct formed during the reaction. This careful balancing of the catalytic system ensures that side reactions, such as the hydrolysis of the nitrile group to an amide or carboxylic acid, are suppressed. Consequently, the resulting crude reaction mixture is cleaner, reducing the load on purification columns and improving the overall yield of the isolated high-purity aryl nitrile, as evidenced by the successful synthesis of various substituted derivatives shown in the substrate scope.
How to Synthesize Aryl Nitriles Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and reproducibility, making it an ideal candidate for technology transfer from the laboratory to the pilot plant. The general procedure involves charging a reactor with the aryl iodide starting material, the nickel catalyst, the phosphine ligand, and the base under an inert nitrogen atmosphere to exclude oxygen and moisture, which could deactivate the catalyst. Tert-butyl isonitrile is then introduced, and the mixture is heated to reflux in toluene, typically around 150°C, for a duration of approximately 12 hours to ensure complete conversion. One of the distinct advantages of this method is the straightforward workup protocol; the reaction mixture can be directly treated with silica gel to adsorb polar impurities and catalyst residues before solvent removal. This eliminates the need for complex aqueous extractions that often generate large volumes of cyanide-contaminated wastewater. For a detailed, step-by-step breakdown of the standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Under a nitrogen protective atmosphere, combine the aryl iodide substrate, nickel bromide catalyst (NiBr2), sodium tert-butoxide base, and dppp ligand in an organic solvent such as toluene.
- Add tert-butyl isonitrile as the cyanation source to the reaction mixture and stir vigorously while heating the system to approximately 150°C for 12 hours.
- Upon completion, cool the reaction, dilute with ethyl acetate, treat with silica gel to remove impurities, and purify via column chromatography to isolate the high-purity aryl nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed cyanation technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary driver for adoption is the substantial reduction in raw material costs achieved by replacing expensive precious metal catalysts with abundant nickel salts. This shift not only lowers the direct material cost but also insulates the supply chain from the volatility of the platinum group metals market. Additionally, the use of tert-butyl isonitrile significantly reduces the safety compliance costs associated with handling剧毒 (highly toxic) cyanide salts. Facilities do not require the same level of specialized containment or emergency response infrastructure, leading to lower overheads and insurance premiums. The simplified workup procedure, which avoids extensive aqueous washing, further contributes to cost reduction by minimizing solvent consumption and wastewater treatment expenses, thereby enhancing the overall economic viability of the manufacturing process.
- Cost Reduction in Manufacturing: The transition from palladium to nickel catalysts represents a massive opportunity for cost optimization in fine chemical manufacturing. Nickel is orders of magnitude cheaper than palladium, and its use in this protocol does not compromise catalytic activity or product quality. By eliminating the need for costly metal scavengers often required to reduce residual palladium levels in pharmaceutical intermediates, the downstream purification costs are also significantly curtailed. Furthermore, the high atom economy of using tert-butyl isonitrile ensures that reagent costs are kept in check, providing a leaner cost structure that improves margin potential for high-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and commercially stable reagents. Unlike specialized cyanating agents that may have limited suppliers or long lead times, nickel bromide, dppp ligands, and tert-butyl isonitrile are commodity chemicals with robust global supply networks. This availability reduces the risk of production stoppages due to raw material shortages. Moreover, the reaction's tolerance to various functional groups means that a single standardized protocol can be applied to a wide range of aryl iodide substrates, simplifying inventory management and allowing for flexible production scheduling to meet fluctuating market demands without the need for process re-validation.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently greener and easier to scale. The avoidance of water-soluble cyanide salts drastically reduces the toxicity of the effluent stream, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations. The use of toluene, a common industrial solvent, allows for easy recovery and recycling via distillation, supporting circular economy initiatives within the plant. The robustness of the reaction conditions, specifically the ability to run at elevated temperatures without catalyst degradation, ensures that the process performs consistently when scaled from kilogram to multi-ton batches, guaranteeing reliable delivery timelines for downstream customers.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries based on the patent data. These questions address critical aspects such as catalyst selection, substrate compatibility, and purification strategies. Understanding these nuances is essential for R&D directors planning to integrate this methodology into their existing synthetic routes. The following insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development discussions.
Q: What are the safety advantages of using tert-butyl isonitrile over traditional cyanide sources?
A: Unlike highly toxic and volatile reagents like HCN or soluble metal cyanides (KCN, NaCN), tert-butyl isonitrile exhibits significantly lower toxicity, reducing occupational hazards and simplifying waste disposal protocols in industrial settings.
Q: Can this nickel-catalyzed method tolerate sensitive functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating esters, ketones, nitro groups, and ethers without significant side reactions or degradation of the sensitive moieties.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The use of inexpensive nickel catalysts instead of precious metals like palladium, combined with a straightforward workup procedure involving silica gel treatment, makes this route highly scalable and economically viable for ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Nitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the nickel-mediated cyanation described in CN114213204A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aryl nitrile intermediate meets the highest industry standards. Whether you require custom synthesis of complex nitrile derivatives or large-scale toll manufacturing, our facility is designed to support your growth from clinical trials to market launch.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce your manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to implement this greener, more economical nickel-catalyzed route. We encourage you to contact our technical procurement team today to request specific COA data for our existing nitrile portfolio or to discuss route feasibility assessments for your proprietary compounds. Let us help you secure a reliable, cost-effective, and safe supply of high-quality aryl nitriles for your next generation of pharmaceutical or agrochemical products.
