Advanced Synthesis of Triazole Alcohol Antifungal Intermediates for Commercial Scale-Up
The escalating global burden of fungal infections, particularly deep mycosis in immunocompromised patients, has created an urgent demand for next-generation antifungal therapeutics with broader spectra and lower toxicity profiles. Patent CN1292378A addresses this critical gap by disclosing a sophisticated class of 1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-3-(N-methyl-N-substituted benzylamino)-2-propanol compounds. These molecules represent a significant leap forward in medicinal chemistry, specifically targeting resistant strains such as Aspergillus fumigatus where traditional azoles often fail. For R&D directors and procurement strategists, understanding the synthetic accessibility of these high-value scaffolds is paramount. The patent outlines a robust, multi-step pathway that transforms readily available starting materials like m-difluorobenzene into potent antifungal intermediates through a series of well-defined chemical transformations including Friedel-Crafts acylation and selective epoxidation. This technological breakthrough not only expands the chemical space for drug discovery but also offers a viable roadmap for the reliable pharmaceutical intermediates supplier market to meet the growing clinical needs for effective antifungal agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of antifungal agents has been plagued by narrow antimicrobial spectrums and the rapid emergence of drug resistance, particularly among deep-seated fungal pathogens. Conventional synthesis routes for triazole derivatives often suffer from harsh reaction conditions, poor regioselectivity, and the generation of difficult-to-remove impurities that compromise the safety profile of the final Active Pharmaceutical Ingredient (API). Many existing processes rely on unstable intermediates or require expensive transition metal catalysts that necessitate complex downstream purification to meet regulatory limits for residual metals. Furthermore, traditional methods frequently struggle to introduce the specific N-methyl-N-substituted benzylamino moiety with high efficiency, leading to low overall yields and inconsistent batch-to-batch quality. These inefficiencies translate directly into higher manufacturing costs and extended lead times, creating bottlenecks for the cost reduction in API manufacturing that pharmaceutical companies desperately seek. The inability of older generations of antifungals to effectively treat Aspergillus fumigatus infections highlights the structural limitations of previous chemical architectures, necessitating a fundamental redesign of the molecular scaffold.
The Novel Approach
The methodology presented in CN1292378A circumvents these historical challenges through a cleverly designed convergent synthesis that maximizes atom economy and operational simplicity. By utilizing a Friedel-Crafts acylation strategy followed by a phase-transfer catalyzed triazole substitution, the process establishes the core pharmacophore under mild conditions that preserve the integrity of the sensitive fluorine substituents. A key innovation lies in the generation of the 1-[2-(2,4-difluorophenyl)-2,3-epoxypropyl]-1H-1,2,4-triazole mesylate intermediate, which serves as a highly reactive electrophile for the final coupling step. This epoxide intermediate allows for the precise installation of diverse N-methyl-N-substituted benzylamine groups via a nucleophilic ring-opening reaction, enabling the rapid generation of a library of analogues to optimize biological activity. The use of trimethylsulfoxonium iodide for epoxidation and N-bromosuccinimide (NBS) for benzylic bromination ensures high selectivity and minimizes side reactions. This novel approach not only enhances the antifungal spectrum but also streamlines the production workflow, making it an attractive candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Friedel-Crafts Acylation and Epoxide Ring-Opening
The cornerstone of this synthesis is the initial Friedel-Crafts acylation, where m-difluorobenzene reacts with chloroacetyl chloride in the presence of anhydrous aluminum trichloride (AlCl3). This Lewis acid-catalyzed reaction proceeds through the formation of a highly electrophilic acylium ion, which attacks the electron-rich aromatic ring at the position ortho to one fluorine and para to the other, driven by the directing effects of the halogen substituents. The reaction is carefully controlled at temperatures between 50°C and 55°C to prevent polyacylation and ensure the formation of 2-chloro-2',4'-difluoroacetophenone with a commendable yield of approximately 82%. Following this, the introduction of the triazole ring is achieved via a nucleophilic substitution where the triazole nitrogen displaces the chlorine atom. This step is facilitated by potassium carbonate (K2CO3) and tetraethylammonium chloride (TEBA) acting as a phase transfer catalyst in dichloromethane, allowing the reaction to proceed efficiently at 0°C to 5°C. The subsequent epoxidation using trimethylsulfoxonium iodide converts the ketone functionality into a strained three-membered oxirane ring, a transformation that is critical for the molecule's biological interaction with the fungal lanosterol 14α-demethylase enzyme.
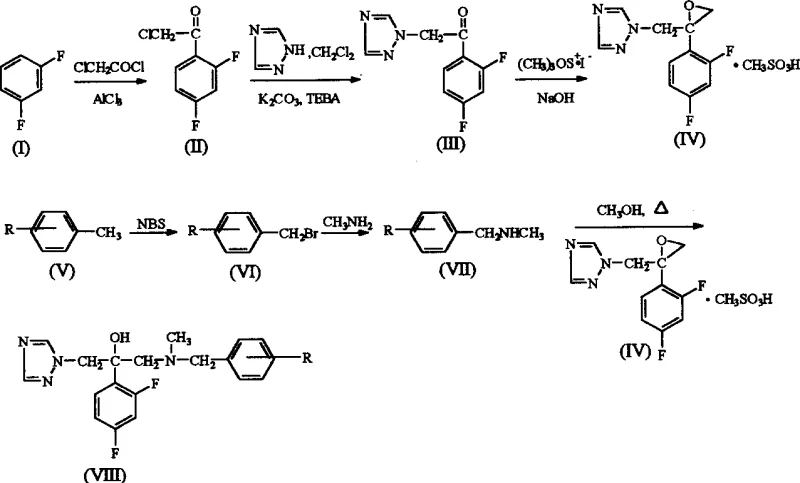
The final stage of the synthesis involves the nucleophilic ring-opening of the epoxy intermediate by the substituted N-methyl-benzylamine. This reaction is conducted in methanol under basic conditions provided by sodium hydroxide, which deprotonates the amine to enhance its nucleophilicity. The amine attacks the less hindered carbon of the epoxide ring, resulting in the formation of the target 2-propanol backbone with high stereochemical control. The patent data indicates that this coupling step proceeds with excellent efficiency, yielding the final product in roughly 82.8% after purification. From an impurity control perspective, the use of recrystallization solvents such as hexane and ethyl acetate mixtures is crucial for removing unreacted starting materials and side products. The rigorous control of reaction parameters, including the stoichiometric ratios of reagents and the precise maintenance of temperature gradients throughout the multi-step sequence, ensures that the final high-purity pharmaceutical intermediates meet the stringent quality standards required for clinical development. This mechanistic understanding allows process chemists to identify critical control points and optimize the workflow for maximum throughput.
How to Synthesize 1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-3-(N-methyl-N-substituted benzylamino)-2-propanol Efficiently
The synthesis of these potent antifungal intermediates requires a disciplined approach to reaction engineering, beginning with the preparation of the key acetophenone precursor. Operators must ensure that the aluminum trichloride is strictly anhydrous to prevent catalyst deactivation during the acylation step, and the addition of chloroacetyl chloride must be dropwise to manage the exotherm effectively. Following the isolation of the chloro-ketone, the triazole substitution demands careful pH control and efficient stirring to maintain the suspension of the inorganic base in the organic phase. The subsequent epoxidation step is particularly sensitive to moisture and temperature, requiring the use of dry toluene and precise heating at 60°C to drive the formation of the ylide intermediate. Finally, the coupling of the epoxy mesylate with the benzylamine derivative should be monitored closely via TLC to prevent over-reaction or degradation of the sensitive epoxide ring. Detailed standardized synthesis steps are provided below to guide the replication of this high-yielding process.
- Perform Friedel-Crafts acylation of m-difluorobenzene with chloroacetyl chloride using anhydrous AlCl3 to generate 2-chloro-2',4'-difluoroacetophenone.
- React the acetophenone derivative with 1H-1,2,4-triazole in the presence of TEBA and K2CO3 to form the triazole ketone intermediate.
- Execute epoxidation using trimethylsulfoxonium iodide followed by mesylation, then couple with N-methyl-substituted benzylamine to yield the final propanol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting the synthesis route described in CN1292378A are profound, primarily due to the reliance on commodity chemicals and the elimination of exotic reagents. The starting materials, such as m-difluorobenzene and chloroacetyl chloride, are widely available in the global chemical market, ensuring a stable supply chain and mitigating the risk of raw material shortages that often plague specialty chemical manufacturing. The process avoids the use of precious metal catalysts like palladium or platinum, which are subject to volatile pricing and supply constraints, thereby significantly reducing the direct material costs associated with production. Furthermore, the high yields reported in the patent examples, ranging from 82% to over 96% in key steps like the benzylic bromination, indicate a highly efficient process that minimizes waste generation and maximizes the output per batch. This efficiency translates directly into substantial cost savings in pharmaceutical intermediates manufacturing, allowing buyers to negotiate more favorable pricing structures while maintaining healthy margins.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize unit operations and simplify purification protocols, which drastically lowers the operational expenditure (OPEX) required for production. By avoiding complex chromatographic separations in favor of crystallization and filtration, the process reduces solvent consumption and energy usage, leading to a greener and more cost-effective manufacturing footprint. The high atom economy of the Friedel-Crafts and epoxidation steps ensures that a maximum proportion of the input mass is converted into the desired product, reducing the cost of goods sold (COGS). Additionally, the ability to synthesize a wide range of analogues from a common epoxy intermediate allows for economies of scale, where a single production line can be adapted to manufacture multiple derivatives without significant retooling costs.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain resilience by reducing the dependency on single-source suppliers for niche reagents. Since the synthesis relies on standard organic transformations that can be performed in general-purpose chemical reactors, it offers flexibility in manufacturing location and capacity. The stability of the intermediates, particularly the epoxy mesylate salt, allows for potential stockpiling or semi-finished goods inventory strategies, which can buffer against demand fluctuations. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on schedule to meet clinical trial timelines or commercial launch dates without disruption.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as evidenced by the successful execution of reactions on gram-to-multigram scales in the patent examples with consistent results. The use of relatively benign solvents like methanol, toluene, and ethyl acetate simplifies waste management and solvent recovery systems, aiding in compliance with increasingly strict environmental regulations. The absence of heavy metal residues eliminates the need for expensive scavenging steps, further streamlining the production process. This environmental compatibility not only reduces disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, making the technology attractive for long-term commercial partnerships and green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triazole alcohol antifungal compounds. The answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of this technology. Understanding these details is essential for stakeholders evaluating the potential integration of this synthetic route into their existing portfolios. The information covers aspects ranging from biological efficacy to process scalability, ensuring a comprehensive overview for decision-makers.
Q: What represents the key innovation in patent CN1292378A regarding antifungal activity?
A: The patent introduces a novel class of 1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-3-(N-methyl-N-substituted benzylamino)-2-propanol compounds that demonstrate superior efficacy against deep mycosis pathogens like Aspergillus fumigatus, overcoming limitations found in existing drugs like fluconazole.
Q: How does the described synthesis route ensure high purity for pharmaceutical applications?
A: The process utilizes controlled crystallization and recrystallization steps, such as using hexane:ethyl acetate systems, alongside specific temperature controls (0°C to 5°C) during critical substitution reactions to minimize impurity formation and ensure stringent purity specifications.
Q: Is this synthetic pathway suitable for large-scale industrial production?
A: Yes, the route employs standard industrial reagents like aluminum trichloride and N-bromosuccinimide (NBS) and achieves consistent yields (e.g., 82% to 96% in key steps), indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Alcohol Antifungal Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry disclosed in CN1292378A for the development of next-generation antifungal therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of anhydrous aluminum trichloride and the precise temperature control needed for the epoxidation steps. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards, minimizing the risk of delays in your drug development pipeline. Our commitment to technical excellence ensures that the complex stereochemistry and functional group tolerance required for these molecules are maintained throughout the manufacturing process.
We invite you to collaborate with us to leverage this advanced synthetic technology for your antifungal drug projects. Our team of expert process chemists is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain and deep technical expertise that can accelerate your time to market. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you bring these vital antifungal medicines to the patients who need them most.
