Advanced Synthesis of 3-Methylflavone-8-Carboxylic Acid: A Breakthrough in Scalable API Intermediate Manufacturing
Advanced Synthesis of 3-Methylflavone-8-Carboxylic Acid: A Breakthrough in Scalable API Intermediate Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates. A pivotal development in this domain is detailed in patent CN111153881A, which outlines a superior preparation method for 3-methylflavone-8-carboxylic acid, a key intermediate in the production of flavoxate hydrochloride. This compound is essential for treating urinary tract disorders, and its demand necessitates a supply chain capable of delivering high-purity materials consistently. The disclosed technology addresses longstanding challenges associated with traditional synthesis routes, specifically targeting issues related to harsh reaction conditions, excessive reagent usage, and difficult post-reaction purification. By shifting towards a solid-supported catalytic system, this innovation offers a compelling value proposition for R&D directors and procurement managers alike, promising not only enhanced chemical quality but also significant operational efficiencies in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-methylflavone-8-carboxylic acid has been plagued by severe process inefficiencies that hinder industrial viability. The conventional route typically involves reacting 2-hydroxy-3-propionyl methyl benzoate with a substantial excess of benzoyl chloride and sodium benzoate at extremely high temperatures reaching 190°C. These harsh thermal conditions create significant safety hazards and place immense stress on reactor equipment, leading to increased maintenance costs and potential downtime. Furthermore, the reliance on excessive reagents results in poor atom economy, generating a large volume of chemical waste that requires costly disposal protocols. Perhaps most critically for quality control, the traditional method inevitably leads to the residue of benzoic acid in the final intermediate. This impurity is notoriously difficult to remove completely and often causes the final flavoxate hydrochloride product to exceed impurity limits, thereby compromising the safety and efficacy profile of the finished drug product.
The Novel Approach
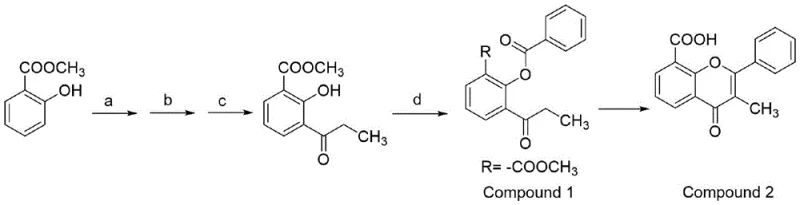
In stark contrast to these legacy methods, the innovative process described in the patent introduces a streamlined, two-step strategy that fundamentally reimagines the cyclization mechanism. The core breakthrough lies in the utilization of dry alkaline alumina as a solid support and catalyst for the cyclization step. Instead of relying on liquid-phase reagents at extreme temperatures, the precursor, 2-benzoyloxy-3-propionyl methyl benzoate, is mixed with the alumina and heated to a significantly milder range of 165-175°C. This reduction in thermal load not only enhances operational safety but also improves the selectivity of the reaction. The process cleverly combines the cyclization and hydrolysis reactions into a cohesive sequence, drastically shortening the overall production timeline. By eliminating the need for excessive benzoyl chloride and sodium benzoate, the new route achieves a much higher atom utilization rate, ensuring that raw materials are converted efficiently into the desired product rather than wasted as byproducts.
Mechanistic Insights into Alumina-Supported Cyclization and Hydrolysis
The mechanistic elegance of this synthesis lies in the multifunctional role played by the dry alkaline alumina. In the critical cyclization step (Step S2), the alumina serves as a heterogeneous base catalyst that facilitates the intramolecular condensation required to form the flavone backbone. The surface basicity of the alumina activates the carbonyl groups within the precursor molecule, promoting the nucleophilic attack necessary for ring closure without the need for aggressive liquid bases that complicate downstream processing. Operating under vacuum during this phase aids in the continuous removal of water generated during the condensation, driving the equilibrium towards product formation according to Le Chatelier's principle. This solid-state or semi-solid state reaction environment minimizes side reactions that are common in solution-phase chemistry at high temperatures, thereby preserving the structural integrity of the sensitive flavone scaffold and ensuring a cleaner reaction profile.
Following the cyclization, the process seamlessly transitions into a hydrolysis step within the same reaction vessel, showcasing excellent process integration. Methanol and a sodium hydroxide solution are introduced to the reaction mixture while it is still warm, typically around 75-80°C. This allows for the efficient saponification of the methyl ester group to yield the free carboxylic acid functionality essential for the final API structure. The alkaline conditions provided by the sodium hydroxide, combined with the residual basicity of the alumina, ensure complete conversion of the ester. Crucially, the heterogeneous nature of the catalyst allows for its physical separation via simple filtration once the reaction is complete. This ease of separation is a major advantage over homogeneous catalysts, which often require complex extraction or chromatography steps to remove trace metal or salt contaminants, thus directly contributing to the high purity specifications achievable with this method.
How to Synthesize 3-Methylflavone-8-Carboxylic Acid Efficiently
Implementing this advanced synthesis route requires precise control over reaction parameters to maximize yield and purity, particularly during the transition from precursor preparation to the final cyclization. The process begins with the meticulous preparation of the intermediate 2-benzoyloxy-3-propionyl methyl benzoate through a sequence of bromination, Friedel-Crafts acylation, hydrogenation, and benzoylation, each step requiring specific temperature controls and stoichiometric precision. Once this precursor is secured, the focus shifts to the solid-supported transformation where the interaction between the organic substrate and the inorganic alumina support dictates the success of the batch. Operators must ensure the alumina is thoroughly dried prior to use to prevent premature hydrolysis or deactivation of the catalytic sites. The heating profile during the cyclization phase must be carefully managed to maintain the optimal 165-175°C window, avoiding thermal degradation while ensuring sufficient energy for ring closure. For a comprehensive understanding of the specific operational parameters and safety protocols required for execution, the detailed standardized synthesis steps are outlined in the guide below.
- Preparation of Precursor: Conduct bromination, Friedel-Crafts acylation with propionyl chloride, hydrogenation, and benzoylation to obtain 2-benzoyloxy-3-propionyl methyl benzoate.
- Solid-Supported Cyclization: Mix the precursor with dry alkaline alumina and heat to 165-175°C under vacuum to effect cyclization.
- Hydrolysis and Isolation: Treat the reaction mixture with methanol and sodium hydroxide at 75-80°C, filter to recover alumina, and acidify to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical yield. The primary advantage lies in the drastic simplification of the post-reaction workup, which directly correlates to reduced processing time and lower labor costs per kilogram of output. By eliminating the need for excessive reagents and the subsequent difficult purification steps associated with removing benzoic acid residues, the manufacturing process becomes significantly more lean and agile. This efficiency gain allows for faster batch turnover rates, enabling suppliers to respond more rapidly to fluctuating market demands for flavoxate intermediates. Furthermore, the ability to recycle the alkaline alumina catalyst represents a substantial reduction in raw material consumption, insulating the supply chain from volatility in reagent pricing and reducing the overall carbon footprint of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and wasteful reagents such as excessive benzoyl chloride and sodium benzoate. In traditional methods, the cost of purchasing, handling, and disposing of these surplus chemicals adds a significant burden to the cost of goods sold. By shifting to a catalytic system with high atom economy, the new route minimizes raw material input while maximizing output. Additionally, the recyclability of the alumina catalyst means that a major consumable cost is amortized over multiple batches, leading to a structurally lower cost base for the intermediate. This cost efficiency can be passed down the supply chain, offering competitive pricing advantages for downstream API manufacturers without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex purification bottlenecks and the availability of specialized reagents. This novel method mitigates those risks by simplifying the purification process to basic filtration and crystallization, removing the dependency on complex chromatographic separations or extensive solvent extractions. The milder reaction conditions also reduce the risk of equipment failure or unplanned shutdowns due to thermal stress, ensuring more consistent production schedules. Moreover, the use of common, commercially available reagents like propionyl chloride and benzoyl chloride in stoichiometric amounts ensures that raw material sourcing remains stable and predictable, safeguarding against supply disruptions that could delay API production timelines.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial tonnage often exposes hidden inefficiencies, particularly regarding waste management. This synthesis route is inherently designed for scalability, as the solid-supported reaction mechanism translates well to larger reactors without the mixing and heat transfer issues often seen in viscous or heterogeneous liquid systems. The significant reduction in chemical waste, specifically the avoidance of large quantities of benzoic acid byproducts, simplifies wastewater treatment and solid waste disposal, ensuring compliance with increasingly stringent environmental regulations. This 'green chemistry' profile not only reduces regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, making it a preferred choice for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived directly from the comparative data and process descriptions found in the patent literature, providing clarity on how this method outperforms legacy techniques. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this intermediate into their existing production lines. The answers highlight the specific operational improvements in purity, yield, and safety that define the value of this innovation.
Q: How does the alumina-catalyzed method improve purity compared to traditional synthesis?
A: The novel method avoids the use of excessive benzoyl chloride and sodium benzoate required in traditional high-temperature routes. By utilizing dry alkaline alumina as a solid support, the process eliminates residual benzoic acid contamination, achieving product purity levels up to 99.5% without complex purification steps.
Q: What are the temperature advantages of this new preparation method?
A: Traditional methods require harsh reaction temperatures around 190°C, posing significant safety and equipment risks. The patented process lowers the cyclization temperature to a milder range of 165-175°C, reducing energy consumption and facilitating easier industrial scale-up while maintaining high reaction efficiency.
Q: Is the catalyst used in this synthesis recyclable?
A: Yes, a key feature of this green chemistry approach is the use of dry alkaline alumina, which acts as both a catalyst and a solid support. After the reaction, the alumina can be filtered, dried, and recycled for subsequent batches, significantly reducing solid waste generation and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylflavone-8-Carboxylic Acid Supplier
As the global demand for high-quality urological medications continues to rise, securing a dependable source of critical intermediates like 3-methylflavone-8-carboxylic acid is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies such as the alumina-catalyzed route to deliver superior products. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We adhere to stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the 99.5% purity benchmark essential for downstream API synthesis, thereby minimizing your risk of impurity-related failures.
We invite you to collaborate with us to optimize your supply chain for flavoxate hydrochloride production. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient manufacturing process can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to validate the quality and compatibility of our intermediates with your current manufacturing processes.
