Revolutionizing Liquid Crystal Intermediates: Scalable Synthesis of Phenyl Triflate Imine Compounds
The chemical landscape for advanced functional materials is constantly evolving, driven by the need for more efficient synthetic routes that minimize waste and maximize purity. Patent CN115850175A introduces a significant breakthrough in the synthesis of phenyl trifluoromethanesulfonate imine compounds, a class of molecules that bridge the gap between complex pharmaceutical intermediates and high-performance liquid crystal materials. This technology leverages a unique acid-catalyzed pathway to install the trifluoromethanesulfonate (-OTf) group directly onto a phenyl azide scaffold, bypassing the traditional and often cumbersome requirement for phenolic hydroxyl precursors. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more atom-economical and operationally simple manufacturing processes. By utilizing cheap and easily available trifluoromethanesulfonic acid as a catalyst, the method achieves high reaction yields while eliminating the generation of difficult-to-remove phosphine oxide byproducts common in older methodologies. This report analyzes the technical merits and commercial viability of this innovation, positioning it as a critical asset for supply chains demanding reliability and cost-efficiency in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of phenyl trifluoromethanesulfonate derivatives has been plagued by synthetic inefficiencies and purification bottlenecks that drive up costs and extend lead times. The conventional wisdom dictates that one must start with a phenol derivative, reacting it with aggressive reagents like trifluoromethanesulfonic anhydride or N-phenylbis(trifluoromethanesulfonyl)imide to install the triflate group. This prerequisite means that if a target molecule lacks a phenolic hydroxyl group, additional synthetic steps are mandatory to introduce this functionality, thereby increasing the step count and reducing overall throughput. Furthermore, when synthesizing the imine (Schiff base) moiety often required for liquid crystal applications, the industry standard has long been the Aza-Wittig reaction. While effective in forming the carbon-nitrogen double bond, this reaction relies heavily on triphenylphosphine as a reductant. The stoichiometric byproduct, triphenylphosphine oxide, possesses a high molecular weight and similar polarity to many organic products, making its removal via standard chromatography or crystallization exceptionally difficult and costly. These cumulative inefficiencies create a substantial burden on manufacturing scalability and purity specifications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN115850175A offers a streamlined, direct route that fundamentally alters the synthetic logic. Instead of relying on phenolic precursors, this novel approach utilizes a phenyl azide compound, specifically 1-azido-2-(iodomethyl)benzene, reacting it with 2,3,3a,4,5,6a-hexahydropentadiene-1,6-dione to form a key intermediate. This intermediate is then subjected to trifluoromethanesulfonic acid catalysis, which simultaneously facilitates cyclization and the installation of the triflate group. 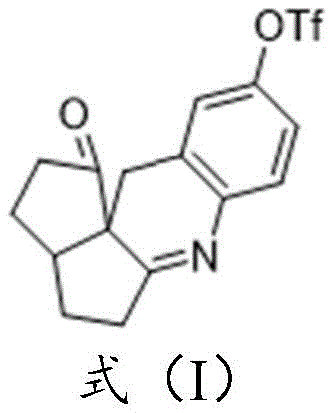 This eliminates the need for pre-functionalized phenols entirely, shaving off multiple synthetic steps and reducing the consumption of expensive starting materials. Moreover, by avoiding the Aza-Wittig reaction, the process generates zero triphenylphosphine oxide, resulting in a much cleaner crude reaction mixture. This simplicity translates directly into operational excellence, allowing for easier scale-up and significantly reduced processing time, which is a critical advantage for a reliable pharmaceutical intermediate supplier aiming to meet tight delivery schedules.
This eliminates the need for pre-functionalized phenols entirely, shaving off multiple synthetic steps and reducing the consumption of expensive starting materials. Moreover, by avoiding the Aza-Wittig reaction, the process generates zero triphenylphosphine oxide, resulting in a much cleaner crude reaction mixture. This simplicity translates directly into operational excellence, allowing for easier scale-up and significantly reduced processing time, which is a critical advantage for a reliable pharmaceutical intermediate supplier aiming to meet tight delivery schedules.
Mechanistic Insights into Acid-Catalyzed Triflation and Cyclization
The core of this innovation lies in the sophisticated yet elegant mechanism driven by trifluoromethanesulfonic acid (TfOH). In the second stage of the synthesis, the intermediate species, which contains both azide and ketone functionalities within a rigid bicyclic framework, undergoes a transformative reaction upon exposure to the strong acid. The TfOH acts not merely as a proton source but as a potent activator that likely promotes the decomposition of the azide group to a reactive nitrene or imine species in situ, which then attacks the adjacent carbonyl or facilitates the elimination required to form the stable imine bond. Concurrently, the highly nucleophilic triflate anion or the acid itself participates in the substitution at the aromatic ring, effectively installing the -OTf leaving group at the para-position relative to the newly formed imine system. This tandem process is highly efficient because it combines bond formation and functional group installation into a single operational step. The reaction conditions are meticulously controlled, typically proceeding at low temperatures such as -20°C in dichloromethane, which helps to suppress side reactions and maintain the integrity of the sensitive imine linkage. This level of control is essential for achieving the high purity required for electronic and optical applications.
From an impurity control perspective, the mechanistic pathway offers distinct advantages over phosphine-mediated routes. In traditional Aza-Wittig chemistry, the reduction of the azide to the amine/imine generates triphenylphosphine oxide (TPPO) in a 1:1 molar ratio with the product. TPPO is notorious for co-eluting with products and forming stable complexes, often requiring specialized scavengers or repeated recrystallizations to remove. In the mechanism described in this patent, the nitrogen extrusion from the azide is managed through acid catalysis and intramolecular cyclization, producing nitrogen gas as the primary volatile byproduct rather than a heavy solid waste.  The absence of phosphorus-containing byproducts simplifies the downstream workup significantly; a simple aqueous wash and extraction protocol is often sufficient to isolate the product with high purity. This mechanistic cleanliness ensures that the final material meets stringent specifications for trace metal and organic impurities, a key concern for R&D directors developing next-generation liquid crystal displays or potent API intermediates where trace contaminants can ruin batch consistency.
The absence of phosphorus-containing byproducts simplifies the downstream workup significantly; a simple aqueous wash and extraction protocol is often sufficient to isolate the product with high purity. This mechanistic cleanliness ensures that the final material meets stringent specifications for trace metal and organic impurities, a key concern for R&D directors developing next-generation liquid crystal displays or potent API intermediates where trace contaminants can ruin batch consistency.
How to Synthesize Phenyl Triflate Imine Efficiently
The practical execution of this synthesis is designed for robustness and reproducibility in a pilot or commercial plant setting. The process begins with the preparation of the critical Intermediate A, which serves as the scaffold for the final transformation. This involves reacting 1-azido-2-(iodomethyl)benzene with the diketone precursor in the presence of a mild base like potassium carbonate in tetrahydrofuran. Once this intermediate is isolated and purified, the final triflation step is performed under inert atmosphere to prevent moisture interference, which could hydrolyze the sensitive triflate group. The reaction is quenched carefully with saturated sodium bicarbonate to neutralize the strong acid catalyst, followed by standard organic extraction techniques. This standardized workflow minimizes operator error and ensures consistent batch-to-batch quality, making it an ideal candidate for technology transfer from lab to production scale.
- Synthesize Intermediate A by reacting 1-azido-2-(iodomethyl)benzene with 2,3,3a,4,5,6a-hexahydropentadiene-1,6-dione using potassium carbonate in tetrahydrofuran.
- Purify Intermediate A via ethyl acetate extraction and column chromatography to ensure high purity before the final step.
- React Intermediate A with trifluoromethanesulfonic acid in dichloromethane at low temperatures (-20°C) to yield the final phenyl triflate imine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that go beyond mere chemical curiosity. The primary driver for cost reduction here is the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized phenolic precursors and expensive phosphine reagents, the bill of materials is significantly streamlined. The use of trifluoromethanesulfonic acid, a commodity chemical, replaces costly coupling agents, leading to substantial cost savings in reagent procurement. Furthermore, the removal of the purification bottleneck associated with triphenylphosphine oxide means that manufacturing cycles are shorter. Less time spent on chromatography or recrystallization translates to higher equipment turnover and lower labor costs per kilogram of product. This efficiency allows for more competitive pricing structures without sacrificing margin, a crucial factor in the highly price-sensitive fine chemical market.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of high-cost reagents and waste disposal fees. Traditional methods generate significant solid waste in the form of phosphine oxides, which require specific disposal protocols and add to the environmental compliance burden. By switching to this acid-catalyzed method, the waste profile is shifted towards simpler aqueous salts and volatile gases, which are cheaper and easier to treat. Additionally, the higher reaction yield reported in the patent implies that less starting material is wasted, directly improving the cost of goods sold (COGS). The avoidance of multi-step sequences to install phenolic groups further reduces solvent consumption and energy usage, contributing to a leaner, more cost-effective manufacturing model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, commodity-grade starting materials. 1-azido-2-(iodomethyl)benzene and the diketone precursor are accessible from multiple global suppliers, reducing the risk of single-source dependency that often plagues specialized pharma intermediates. The robustness of the reaction conditions—using common solvents like dichloromethane and tetrahydrofuran—means that production is not vulnerable to shortages of exotic reagents. This stability ensures that lead times for high-purity pharmaceutical intermediates can be kept short and predictable. For clients managing just-in-time inventory systems, this reliability is invaluable, as it mitigates the risk of production stoppages due to raw material delays, ensuring a continuous flow of critical materials for downstream applications.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is inherently scalable due to its operational simplicity. The reaction does not require extreme pressures or temperatures, and the exotherm is manageable with standard cooling systems, making it safe for large-scale reactors. From an environmental standpoint, the process aligns well with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The absence of heavy metal catalysts or persistent organic pollutants like phosphine oxides simplifies wastewater treatment and regulatory reporting. This ease of compliance accelerates the approval process for new manufacturing sites and reduces the administrative overhead associated with environmental health and safety (EHS) audits, facilitating faster commercial scale-up of complex organic intermediates.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the synthesis and handling of these compounds. The following insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is critical for process chemists who need to integrate this intermediate into larger synthetic schemes or formulate it into final products. These clarifications aim to provide a clear picture of the operational parameters and potential integration points for this advanced chemical technology.
Q: How does this method improve purification compared to Aza-Wittig reactions?
A: Traditional Aza-Wittig reactions generate stoichiometric amounts of triphenylphosphine oxide, a high molecular weight byproduct that is notoriously difficult to remove. This novel acid-catalyzed route completely bypasses the use of triphenylphosphine, significantly simplifying downstream purification and improving overall yield.
Q: What are the primary applications of the phenyl triflate imine compound?
A: The compound possesses dual functionality. The triflate (-OTf) group serves as an excellent leaving group for cross-coupling reactions like Suzuki and Heck couplings, making it valuable for pharmaceutical synthesis. Simultaneously, the imine (-RC=N-) structure provides liquid crystal properties, suitable for display material applications.
Q: Is the trifluoromethanesulfonic acid catalyst recoverable?
A: While the patent highlights the use of cheap and readily available trifluoromethanesulfonic acid to drive the reaction efficiently, standard workup procedures involve quenching with saturated sodium bicarbonate. This neutralizes the acid, ensuring the final product is free from acidic impurities, though the acid itself is consumed or neutralized in the process rather than recycled in a closed loop.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl Triflate Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust infrastructure. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields seen in the lab are replicated faithfully on an industrial scale. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of phenyl trifluoromethanesulfonate imine meets the exacting standards required for liquid crystal and pharmaceutical applications. We understand the critical nature of supply continuity and have optimized our logistics to support global demand efficiently.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
