Advanced Manufacturing of 2-Methyl-1-[4-(methylthio)phenyl]-2-(4-morpholinyl)-1-propanone for UV Curing Applications
Introduction to Advanced Photoinitiator Synthesis
The global demand for high-efficiency ultraviolet (UV) curing agents continues to surge, driven by the expanding printing ink, coating, and electronic materials sectors. At the forefront of this innovation is the synthesis of 2-methyl-1-[4-(methylthio)phenyl]-2-(4-morpholinyl)-1-propanone, a critical photoinitiator known for its superior UV absorption and compatibility with acrylic ester prepolymers. Patent CN101659644B introduces a groundbreaking methodology that fundamentally redefines the production landscape for this specialty chemical. By shifting away from hazardous halogenation and volatile gas handling, this technology offers a robust, environmentally friendly pathway that aligns perfectly with modern green chemistry principles. For R&D directors and procurement strategists, understanding this shift is vital, as it represents not just a chemical improvement, but a strategic supply chain advantage.
This report analyzes the technical depth of patent CN101659644B, highlighting how the implementation of phase-transfer catalysis (PTC) resolves long-standing bottlenecks in yield and purity. The transition from legacy bromine-based or gas-phase thioetherification routes to this aqueous-organic biphasic system marks a significant maturation in fine chemical manufacturing. As a reliable photoinitiator supplier, recognizing the nuances of this synthetic route allows stakeholders to better evaluate cost structures and quality assurances. The following sections provide a comprehensive dissection of the mechanistic advantages and commercial implications of this proprietary technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this photoinitiator has been plagued by four distinct problematic pathways, each carrying severe economic and technical liabilities. The earliest methods, such as those described in EP0088050 and CN1354175A, relied heavily on the use of liquid bromine for the alpha-bromination of ketones. Liquid bromine is not only increasingly expensive due to resource scarcity but also presents formidable handling hazards, requiring specialized corrosion-resistant equipment and rigorous safety protocols. Furthermore, these routes often necessitated complex cyclization steps that resulted in lower overall recovery rates, driving up the cost of goods sold (COGS) and creating significant waste disposal challenges for manufacturers.
Subsequent attempts to optimize the process, such as the route disclosed in WO2006034966A, attempted to bypass bromine by utilizing chlorination on methylthio-intermediates. However, this approach introduced a critical chemical flaw: the susceptibility of the sulfur atom to electrophilic attack. During the chlorination step, substantial side reactions occur where the methylthio group is inadvertently chlorinated, leading to the formation of structurally similar byproducts. 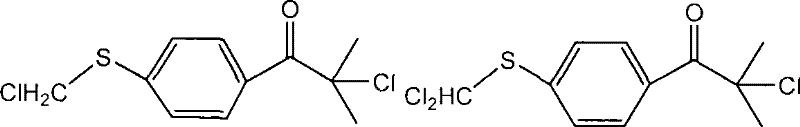 As illustrated in the structural analysis, these impurities account for approximately 10% of the crude product, severely compromising purity and necessitating energy-intensive recrystallization processes that further erode yield. Additionally, other legacy methods utilizing methanethiol gas suffered from volatility issues, where the reagent would escape the reaction matrix before conversion, creating malodorous emissions and inconsistent stoichiometry.
As illustrated in the structural analysis, these impurities account for approximately 10% of the crude product, severely compromising purity and necessitating energy-intensive recrystallization processes that further erode yield. Additionally, other legacy methods utilizing methanethiol gas suffered from volatility issues, where the reagent would escape the reaction matrix before conversion, creating malodorous emissions and inconsistent stoichiometry.
The Novel Approach
In stark contrast to these legacy deficiencies, the methodology outlined in CN101659644B employs a sophisticated nucleophilic substitution strategy that elegantly circumvents previous pitfalls. The core innovation lies in the use of 2-methyl-2-(4-morpholinyl)-1-(4-chlorophenyl)-1-acetone as a stable starting material, which undergoes thioetherification with sodium methyl mercaptide in the presence of a quaternary ammonium salt phase-transfer catalyst. This reaction occurs within an organic-water two-phase system, leveraging the unique properties of PTC to shuttle reactive anions across the interface. 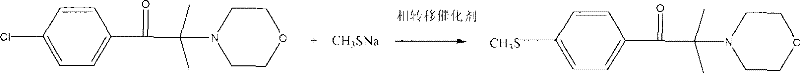 This approach completely eliminates the need for elemental bromine and avoids the problematic chlorination of sulfur-containing intermediates. By conducting the reaction in a controlled biphasic environment, the process achieves conversion rates exceeding 99%, with isolated yields consistently reaching between 95% and 98%. This represents a paradigm shift in cost reduction in specialty chemical manufacturing, offering a cleaner, safer, and significantly more economical route to market.
This approach completely eliminates the need for elemental bromine and avoids the problematic chlorination of sulfur-containing intermediates. By conducting the reaction in a controlled biphasic environment, the process achieves conversion rates exceeding 99%, with isolated yields consistently reaching between 95% and 98%. This represents a paradigm shift in cost reduction in specialty chemical manufacturing, offering a cleaner, safer, and significantly more economical route to market.
Mechanistic Insights into Phase-Transfer Catalyzed Thioetherification
The success of this novel synthesis hinges on the precise orchestration of phase-transfer catalysis (PTC), a mechanism that dramatically enhances reaction kinetics in heterogeneous systems. In this specific transformation, the quaternary ammonium salt acts as a molecular ferry, transporting the nucleophilic methyl mercaptide anion (CH3S-) from the aqueous phase into the organic phase where the chloro-precursor resides. Once in the organic phase, the lipophilic ion pair reacts rapidly with the electrophilic carbon center of the ketone precursor via an SN2 mechanism. This interfacial catalysis ensures that the concentration of the active nucleophile remains high at the reaction site, driving the equilibrium towards completion without the need for extreme temperatures or pressures. The result is a highly efficient process that minimizes energy consumption while maximizing throughput, a key consideration for large-scale industrial operations.
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic substitution routes. Because the reaction relies on nucleophilic attack on a pre-halogenated aromatic ring rather than electrophilic halogenation of a sulfur-containing ring, the chemoselectivity is inherently superior. The sulfur atom in the sodium methyl mercaptide reagent is already in its reduced, nucleophilic state and is not subjected to oxidative conditions that could lead to sulfoxide or sulfone formation, nor is it exposed to chlorinating agents that cause the side reactions seen in prior art. This intrinsic selectivity means that the crude product profile is exceptionally clean, reducing the burden on downstream purification units. For quality assurance teams, this translates to a more consistent impurity profile and easier compliance with stringent specifications required for high-end UV curing applications in food packaging or electronics.
How to Synthesize 2-Methyl-1-[4-(methylthio)phenyl]-2-(4-morpholinyl)-1-propanone Efficiently
Implementing this synthesis route requires careful attention to the biphasic reaction parameters to ensure optimal mass transfer and conversion. The process begins by charging the reactor with the chloro-precursor, an organic solvent such as toluene or chlorobenzene, and an aqueous solution of sodium methyl mercaptide. A catalytic amount of a quaternary ammonium salt, such as tetrabutylammonium bromide, is then introduced to initiate the phase transfer cycle. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below.
- Combine 2-methyl-2-(4-morpholinyl)-1-(4-chlorophenyl)-1-acetone, aqueous sodium methyl mercaptide, and a quaternary ammonium salt phase-transfer catalyst in a reaction vessel.
- Stir and heat the biphasic mixture for 6 to 20 hours until the residual chloro-precursor content drops below 0.5%.
- Separate the organic phase, wash with water to neutrality, concentrate, and recrystallize the residue to obtain the high-purity target photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend far beyond simple chemical yield. The elimination of liquid bromine removes a volatile cost factor from the bill of materials, stabilizing pricing models against fluctuations in the halogen market. Furthermore, the avoidance of toxic methanethiol gas simplifies facility requirements, removing the need for specialized gas scrubbing infrastructure and reducing insurance premiums associated with hazardous material storage. These factors combine to create a more resilient and cost-effective supply chain, ensuring continuity of supply even during periods of raw material constraint.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of expensive and hazardous reagents with commodity chemicals. By utilizing sodium methyl mercaptide and avoiding the multi-step bromination or problematic chlorination sequences, the overall number of unit operations is reduced. This simplification leads to substantial cost savings in terms of labor, energy, and solvent consumption. Additionally, the high yield of 95-98% minimizes raw material waste, ensuring that nearly every kilogram of input is converted into saleable product, thereby optimizing the return on investment for production assets.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically the chloro-precursor and sodium methyl mercaptide, are widely available commodities with stable global supply chains. Unlike niche reagents that may be subject to geopolitical supply shocks or single-source dependencies, these inputs can be sourced from multiple qualified vendors. This diversification reduces the risk of production stoppages and allows for more flexible inventory management. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further enhancing supply reliability.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior to legacy methods. The biphasic system generates significantly less hazardous waste, and the absence of bromine and sulfur gases simplifies effluent treatment. This alignment with green chemistry principles facilitates easier regulatory approval and permits for capacity expansion. The process is inherently scalable, having been demonstrated to work efficiently from laboratory bench scales up to multi-ton commercial batches, making it an ideal candidate for meeting growing market demand without proportional increases in environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this photoinitiator. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational safety.
Q: How does this new method improve product purity compared to traditional chlorination routes?
A: Traditional methods involving the chlorination of methylthio-intermediates often suffer from severe side reactions where the sulfur atom is also chlorinated, generating difficult-to-remove byproducts. This novel phase-transfer catalysis method utilizes a pre-formed chloro-precursor and performs a nucleophilic substitution, completely avoiding electrophilic attack on the sulfur group and ensuring significantly higher purity without complex purification steps.
Q: What are the safety advantages of using sodium methyl mercaptide over methanethiol gas?
A: Previous processes utilizing methanethiol gas faced challenges with volatility and malodorous emissions, posing significant safety and environmental risks. By employing an aqueous solution of sodium methyl mercaptide in a closed biphasic system, this method eliminates the handling of toxic gases, drastically improving operational safety and reducing the burden on waste gas treatment systems.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process is highly scalable. It operates under mild conditions using common solvents like toluene or chlorobenzene and avoids hazardous reagents like liquid bromine. The use of a robust phase-transfer catalyst system allows for efficient mixing and reaction kinetics in large-scale reactors, making it ideal for commercial manufacturing from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN101659644B requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of photoinitiator meets the exacting standards required for high-performance UV curing systems.
We invite you to collaborate with us to leverage these technological advancements for your product lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized synthesis can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the global specialty chemicals market.
