Advanced Catalytic Route for 5,10-Diaryl Pyrido Naphthyridine Derivatives: Scalable Pharma Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those exhibiting potent biological activities such as antibacterial properties. Patent CN111393440A introduces a groundbreaking methodology for the preparation of 5,10-diaryl pyrido [4,3-b][1,6] naphthyridine derivatives, a class of compounds critical for developing next-generation quinolone antibiotics. This innovation leverages a novel acidic ionic liquid catalytic system that fundamentally alters the reaction landscape, moving away from traditional stoichiometric acid catalysts towards a more sustainable, recyclable, and efficient protocol. By integrating an ethanol-dimethylformamide-water mixed solvent system, the process achieves exceptional control over reaction kinetics and product crystallization. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier options, this technology represents a significant leap forward in process chemistry, offering a viable solution for the cost reduction in pharmaceutical intermediates manufacturing while adhering to stringent environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of naphthyridine derivatives has relied heavily on protocols utilizing imidazole-based ionic liquids like [Bmim]Br in conjunction with strong organic acids such as p-toluenesulfonic acid (TsOH). While these methods demonstrated feasibility on a laboratory scale, they present severe bottlenecks for industrial application. The primary drawback lies in the non-recyclable nature of the p-toluenesulfonic acid catalyst, which generates substantial acidic waste streams requiring neutralization and disposal, thereby inflating operational expenditures and environmental compliance costs. Furthermore, the imidazole-based solvents are not only expensive to procure but also pose challenges regarding microbial degradation and long-term ecological impact. From a supply chain perspective, the reliance on these specific, high-cost reagents creates vulnerability in raw material sourcing. Additionally, conventional processes often suffer from prolonged reaction times and complex purification workflows involving multiple extraction and chromatography steps, which drastically reduce overall throughput and increase the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a specialized acidic ionic liquid catalyst that functions effectively within a greener, ternary solvent system. This approach eliminates the need for expensive imidazolium salts, replacing them with a more economically viable and environmentally benign medium. The core innovation lies in the synergistic effect of the acidic ionic liquid and the ethanol-dimethylformamide-water mixture, which not only accelerates the condensation reaction but also facilitates product isolation through simple precipitation. As illustrated in the reaction scheme below, the transformation proceeds efficiently under reflux conditions, yielding the target 5,10-diaryl pyrido [4,3-b][1,6] naphthyridine skeleton with remarkable selectivity.
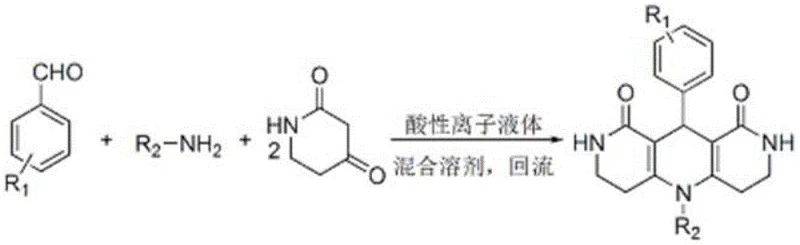
This streamlined workflow allows the catalytic system to remain in the mother liquor after filtration, enabling direct reuse for subsequent batches without significant loss of activity. This closed-loop capability is a game-changer for commercial scale-up of complex heterocyclic intermediates, as it decouples production volume from linear increases in catalyst consumption and waste generation.
Mechanistic Insights into Acidic Ionic Liquid Catalysis
The efficacy of this synthetic route is rooted in the unique physicochemical properties of the acidic ionic liquid catalyst, specifically the triethylammonium hydrogen sulfate-based structure shown below. This catalyst acts as a dual-function promoter, providing both Brønsted acidity to activate the carbonyl groups of the aldehyde and piperidinedione, and a structured ionic environment that stabilizes transition states. The protons donated by the hydrogen sulfate anions facilitate the initial Knoevenagel condensation and subsequent Michael addition steps, which are critical for constructing the fused ring system. Unlike homogeneous mineral acids that can lead to uncontrolled polymerization or tar formation, the ionic liquid offers a buffered acidic environment that moderates reaction vigor, thereby suppressing the formation of oligomeric byproducts and ensuring high chemoselectivity.
![Chemical structure of the acidic ionic liquid catalyst [(Et3NH)2SO][HSO4]2 used in the synthesis](/insights/img/5-10-diaryl-pyrido-naphthyridine-synthesis-pharma-supplier-20260315133429-01.png)
Furthermore, the impurity control mechanism is intrinsically linked to the solvent system's polarity profile. The specific ratio of ethanol, dimethylformamide, and water creates a solubility gradient where the reactants are fully soluble at reflux temperatures, ensuring homogeneous reaction kinetics. However, upon cooling to room temperature, the solubility of the highly polar naphthyridine product drops precipitously, causing it to crash out of the solution as a high-purity solid. This "reactive crystallization" phenomenon effectively excludes soluble impurities and unreacted starting materials, which remain in the supernatant. Consequently, the need for energy-intensive recrystallization or column chromatography is obviated, resulting in a process that consistently delivers high-purity 5,10-diaryl pyrido naphthyridine derivative suitable for downstream pharmaceutical applications without extensive post-processing.
How to Synthesize 5,10-Diaryl Pyrido Naphthyridine Derivative Efficiently
The operational simplicity of this protocol makes it highly attractive for technology transfer teams looking to implement robust manufacturing processes. The synthesis involves a straightforward one-pot procedure where aromatic aldehydes, amines, and 2,4-piperidinedione are combined in the optimized solvent mixture with a catalytic loading of the acidic ionic liquid. The reaction is heated to reflux, typically completing within 75 to 96 minutes, as monitored by thin-layer chromatography. Upon completion, the mixture is simply cooled, allowing the product to precipitate spontaneously. The detailed standardized synthesis steps, including precise molar ratios and specific workup parameters derived from the patent examples, are outlined in the guide below to assist process engineers in replicating these results.
- Charge aromatic aldehyde, amine, and 2,4-piperidinedione into a reactor with an ethanol-dimethylformamide-water mixed solvent and acidic ionic liquid catalyst.
- Heat the mixture to reflux conditions (approximately 75-96 minutes) under atmospheric pressure until TLC indicates complete consumption of starting materials.
- Cool the reaction mixture to room temperature to induce precipitation, then filter, wash the solid with water, and vacuum dry at 80°C to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic advantages beyond mere technical performance. The shift from stoichiometric acids and expensive imidazolium solvents to a recyclable acidic ionic liquid system fundamentally reshapes the cost structure of production. By enabling the direct reuse of the catalytic filtrate, the process dramatically reduces the consumption of fresh catalyst and solvent per kilogram of product. This reduction in material intensity leads to substantial cost savings in pharmaceutical intermediates manufacturing, insulating the supply chain from volatility in raw material pricing. Moreover, the simplified workup procedure, which relies on filtration and water washing rather than complex organic extractions, significantly lowers utility consumption and waste disposal fees, contributing to a leaner and more competitive cost base.
- Cost Reduction in Manufacturing: The elimination of costly imidazole-based ionic liquids and the ability to recycle the acidic catalyst system directly impacts the bottom line. Traditional methods often require fresh catalyst for every batch, generating significant chemical waste that incurs disposal costs. In this novel process, the catalyst remains active in the filtrate, allowing for multiple cycles of reuse without regeneration. This circular economy approach minimizes raw material expenditure and reduces the burden on waste treatment facilities, driving down the overall cost of goods sold (COGS) while maintaining high margins.
- Enhanced Supply Chain Reliability: The reliance on commodity solvents like ethanol and water, rather than specialized ionic liquids that may have limited suppliers, enhances supply security. Ethanol and water are globally available and inexpensive, reducing the risk of supply disruptions. Additionally, the short reaction time of approximately 75 to 96 minutes increases reactor turnover rates, allowing manufacturers to respond more agilely to market demand fluctuations. This efficiency ensures a steady flow of high-quality intermediates, reducing lead times and improving inventory management for downstream API production.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at atmospheric pressure and the absence of hazardous reagents. The use of water as a co-solvent and for washing simplifies effluent treatment, as the wastewater is less contaminated with persistent organic pollutants compared to processes using chlorinated solvents or non-degradable ionic liquids. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, making it a sustainable choice for long-term production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into existing production lines. The following questions address common inquiries regarding catalyst stability, product quality, and operational parameters based on the data provided in the patent documentation. These insights are intended to clarify the practical implications of adopting this acidic ionic liquid catalyzed route for the production of naphthyridine derivatives.
Q: How does the acidic ionic liquid catalyst improve the recycling capability compared to traditional methods?
A: Unlike traditional p-toluenesulfonic acid which generates waste, the acidic ionic liquid catalyst described in patent CN111393440A remains in the filtrate after product precipitation. This allows the catalytic system and solvent to be directly reused for subsequent batches without complex regeneration, significantly reducing waste generation and raw material costs.
Q: What are the purity levels achievable with this specific solvent system?
A: The optimized ethanol-dimethylformamide-water mixed solvent system facilitates effective crystallization upon cooling. Experimental data from the patent indicates that products can achieve purity levels exceeding 98% (e.g., 98.5% in Example 1) directly after filtration and washing, eliminating the need for additional recrystallization steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It operates at atmospheric pressure with short reaction times (75-96 minutes) and utilizes a simple workup procedure involving filtration and water washing. The elimination of expensive imidazolium-based ionic liquids further enhances its economic viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,10-Diaryl Pyrido Naphthyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the potential of the acidic ionic liquid catalysis described in patent CN111393440A and is well-positioned to translate this laboratory innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5,10-diaryl pyrido naphthyridine derivative meets the exacting standards required for global pharmaceutical applications.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in green catalysis and process intensification, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and quality goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
