Advanced Synthesis of Lithium 2-fluoro-malonate Difluoroborate for Next-Gen Silicon Anode Batteries
Advanced Synthesis of Lithium 2-fluoro-malonate Difluoroborate for Next-Gen Silicon Anode Batteries
The relentless pursuit of higher energy density in energy storage systems has driven the industry toward silicon-based anode materials, yet their commercial viability has long been hindered by structural instability during cycling. Addressing this critical bottleneck, patent CN111138462B discloses a robust preparation method for lithium 2-fluoro-malonate difluoroborate (LiFMDFB), a novel functional additive designed to stabilize the Solid Electrolyte Interphase (SEI) on silicon surfaces. This technical breakthrough offers a viable pathway for manufacturers seeking to enhance battery longevity without compromising capacity. As a leading entity in fine chemical synthesis, we recognize the immense potential of this organoboron compound to revolutionize the electrolyte landscape. The following analysis dissects the synthetic innovation, mechanistic advantages, and commercial implications of this technology for global supply chains.
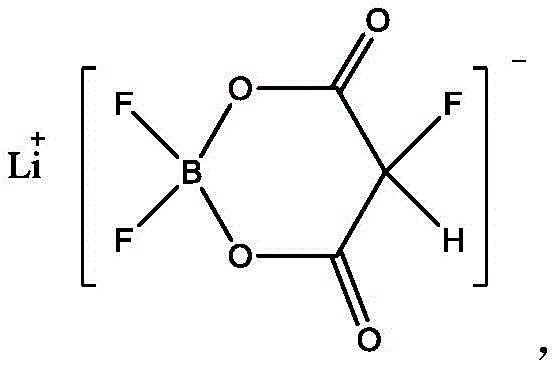
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specialized organoboron electrolyte additives has been plagued by complex multi-step sequences that often rely on hazardous fluorinating agents or moisture-sensitive intermediates. Traditional routes frequently suffer from poor atom economy and generate significant quantities of inorganic salt waste, which complicates downstream purification and drives up disposal costs. Furthermore, achieving the ultra-low metal impurity levels required for high-performance battery electrolytes—typically in the parts-per-billion range—is exceptionally difficult using crude precipitation methods. These inefficiencies result in batch-to-batch variability that poses severe risks to cell consistency, forcing procurement teams to manage high safety stocks and accept inflated pricing structures due to low overall process yields.
The Novel Approach
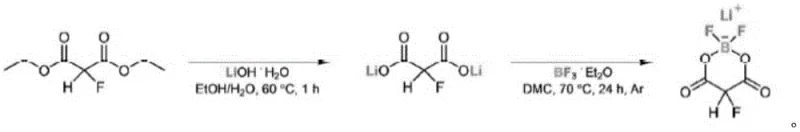
In stark contrast, the methodology outlined in the patent introduces a streamlined two-stage process that begins with the mild alkaline hydrolysis of 2-fluoro-malonic diesters. This approach avoids the use of aggressive reagents, instead utilizing lithium hydroxide in an aqueous medium to cleanly generate the lithium salt precursor. The subsequent complexation with boron trifluoride complexes in organic carbonates allows for precise control over the reaction kinetics, facilitating the formation of the stable five-membered boron chelate ring. By integrating a targeted recrystallization step using non-polar anti-solvents, the process effectively strips away trace metal contaminants and unreacted starting materials. This refined workflow not only simplifies the operational complexity but also ensures a consistent supply of high-purity material suitable for sensitive electrochemical applications.
Mechanistic Insights into Organoboron Chelate Formation
The efficacy of lithium 2-fluoro-malonate difluoroborate lies in its unique molecular architecture, where the boron atom is tetrahedrally coordinated with two fluorine atoms and two oxygen atoms from the malonate backbone. During the synthesis, the nucleophilic attack of the carboxylate oxygens on the electron-deficient boron center creates a thermodynamically stable cyclic structure. This rigidity is crucial for the additive's performance in the battery cell, as it prevents premature decomposition at the anode interface. The presence of the fluorine substituent on the alpha-carbon further enhances the electron-withdrawing character of the molecule, which modulates the reduction potential to ensure that the additive decomposes preferentially before the bulk solvent. This mechanism leads to the formation of a flexible yet robust SEI layer rich in boron and fluorine species, which can accommodate the massive volume expansion of silicon particles without fracturing.
From a purity perspective, the reaction mechanism inherently favors the exclusion of transition metal impurities which are detrimental to battery cycle life. The coordination chemistry involved in the final crystallization step selectively incorporates the lithium-boron complex into the crystal lattice while excluding ions such as iron, chromium, and nickel, which remain in the mother liquor. Experimental data from the patent indicates that this selective crystallization can reduce metal impurities to levels as low as 0.01 ppm for iron and nickel. For R&D directors, this implies that the material can be integrated into high-voltage or high-nickel cathode systems without triggering accelerated degradation pathways. The precise control over stoichiometry during the complexation phase ensures that no free Lewis acidic boron species remain, which could otherwise catalyze unwanted side reactions with the carbonate solvent matrix.
How to Synthesize Lithium 2-fluoro-malonate Difluoroborate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control during the hydrolysis and drying phases to prevent decarboxylation or thermal degradation of the fluorinated backbone. The process begins with the preparation of a concentrated lithium hydroxide solution, followed by the controlled addition of the fluoro-diester under agitation to ensure homogeneous mixing and complete conversion. Once the lithium salt is isolated via vacuum drying, it is critical to maintain an inert atmosphere during the subsequent reaction with the boron trifluoride complex to prevent moisture ingress, which could hydrolyze the sensitive B-F bonds. The final purification via reverse crystallization in hexane or similar alkanes is the key determinant of final product quality, requiring precise temperature ramping to maximize crystal growth and impurity rejection. Detailed standardized operating procedures for each unit operation are essential for maintaining reproducibility at scale.
- Perform alkaline hydrolysis on 2-fluoro-malonic diester using a lithium hydroxide aqueous solution at controlled temperatures to generate the lithium salt intermediate.
- Concentrate the resulting aqueous solution and subject it to vacuum drying at elevated temperatures to isolate solid lithium 2-fluoromalonate.
- React the dried lithium salt with a boron trifluoride complex in an organic solvent, followed by filtration and recrystallization to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route presents a compelling value proposition centered on cost efficiency and supply security. By utilizing readily available starting materials such as fluoro-malonic esters and commodity chemicals like lithium hydroxide and dimethyl carbonate, the process mitigates the risk of raw material bottlenecks that often plague specialty chemical supply chains. The elimination of exotic catalysts and the reliance on standard stainless steel reactor equipment significantly lower the barrier to entry for manufacturing, allowing for broader sourcing options and reduced dependency on single-source suppliers. This diversification of the supply base is critical for maintaining continuity in the face of global logistical disruptions, ensuring that battery manufacturers can secure the necessary volumes of high-performance additives without excessive lead times.
- Cost Reduction in Manufacturing: The streamlined nature of the synthesis directly translates to lower operational expenditures by reducing the number of unit operations and minimizing solvent consumption. Unlike traditional methods that may require multiple purification columns or chromatographic separations, this route achieves high purity through simple crystallization, drastically cutting down on energy usage and waste treatment costs. The high yield demonstrated in the patent examples suggests that raw material utilization is optimized, meaning less feedstock is wasted as byproducts. Consequently, the overall cost of goods sold for the final electrolyte additive is significantly reduced, allowing battery producers to achieve better margins or pass savings on to consumers in the competitive electric vehicle market.
- Enhanced Supply Chain Reliability: The robustness of the chemical process ensures consistent output quality, which is vital for long-term supply agreements in the automotive sector. Because the reaction conditions are relatively mild and do not require extreme pressures or cryogenic temperatures, the risk of unplanned plant shutdowns due to equipment failure is minimized. Furthermore, the stability of the intermediates allows for flexible production scheduling, enabling manufacturers to build inventory buffers during periods of low demand. This operational flexibility provides a strategic advantage for supply chain heads who must balance just-in-time delivery requirements with the need for safety stock, ultimately fostering stronger partnerships between chemical suppliers and cell manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop experiments to multi-ton industrial production without fundamental changes to the chemistry. The use of green solvents like dimethyl carbonate and the avoidance of heavy metal catalysts align with increasingly stringent environmental regulations regarding volatile organic compounds and toxic waste discharge. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of fines or production halts due to environmental violations. Additionally, the high atom economy of the reaction means that waste generation is kept to a minimum, supporting corporate sustainability goals and enhancing the brand reputation of companies adopting this greener manufacturing technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of lithium 2-fluoro-malonate difluoroborate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on performance metrics and process capabilities. Understanding these details is essential for stakeholders evaluating the integration of this additive into next-generation battery formulations. We encourage technical teams to review these points closely to assess the fit for their specific R&D and production requirements.
Q: What is the primary advantage of Lithium 2-fluoro-malonate difluoroborate in silicon-based anodes?
A: It stabilizes the Solid Electrolyte Interphase (SEI) film, preventing the continuous consumption of electrolyte and lithium ions caused by the volume expansion of silicon materials during cycling.
Q: How does the patented synthesis method improve product purity compared to conventional routes?
A: The method utilizes specific recrystallization techniques and controlled hydrolysis conditions that effectively remove metal impurities like Fe, Cr, and Ni, achieving ion chromatographic purity levels exceeding 99.5%.
Q: Is this synthesis route scalable for industrial electrolyte additive production?
A: Yes, the process relies on standard unit operations such as hydrolysis, vacuum drying, and recrystallization using common solvents like dimethyl carbonate, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lithium 2-fluoro-malonate Difluoroborate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a reliable commercial reality for the global battery industry. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced ion chromatography and ICP-MS instrumentation to guarantee that every batch meets the exacting standards required for silicon-anode electrolytes. Our commitment to quality assurance means that customers receive material with consistently low metal impurity profiles, safeguarding the performance and safety of their final battery cells.
We invite procurement directors and R&D leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized synthesis can lower your total cost of ownership. Contact us today to discuss how we can support your supply chain with high-purity electrolyte additives that drive the next generation of energy storage innovation.
