Industrial Scale-Up of High-Purity Pest Control Agent Intermediates via Novel Acylation
Industrial Scale-Up of High-Purity Pest Control Agent Intermediates via Novel Acylation
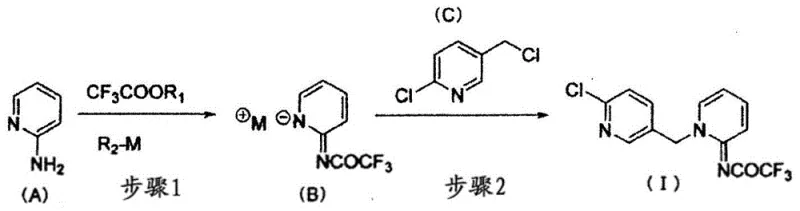
The global demand for effective pest control agents necessitates robust, scalable, and economically viable synthetic routes for key intermediates. Patent CN109715606B discloses a groundbreaking methodology for the production of N-[1-((6-chloropyridin-3-yl)methyl)pyridin-2(1H)-ylidene]-2,2,2-trifluoroacetamide, a compound of significant utility in the agrochemical sector. This technical insight report analyzes the proprietary process which utilizes a trifluoroacetate and a metal alkali compound to transform 2-aminopyridine into the target 2-acyliminopyridine structure. By shifting away from traditional acylation limitations, this invention offers a pathway to high-purity agrochemical intermediates with exceptional yield efficiency. For procurement managers and R&D directors seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this route is critical for securing long-term supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art documents, specifically international publications WO2012/029672 and WO2016/005276, describe methods for synthesizing 2-acyliminopyridine derivatives that suffer from significant industrial drawbacks. Traditional acylation reactions often require excessive amounts of trifluoroacetate reagents, leading to inflated raw material costs and complex downstream purification processes. Furthermore, these legacy methods are characterized by low yields and prolonged reaction times; for instance, some prior processes require up to 18 hours for the alkylation step alone, severely bottlenecking production throughput. Additionally, certain conventional routes generate undesirable by-products such as pyridinium salts, which complicate waste treatment and increase the environmental burden of manufacturing. These inefficiencies render older methods unsuitable for the high-volume, cost-sensitive demands of modern pest control agent manufacturing.
The Novel Approach
The inventive process detailed in CN109715606B overcomes these hurdles by introducing a streamlined two-step sequence that can be executed as a efficient one-pot reaction. By employing a metal base compound, such as sodium methoxide or sodium ethoxide, in conjunction with a trifluoroacetate ester, the acylation of the amino group at the 2-position of 2-aminopyridine proceeds with remarkable speed and selectivity. This novel approach eliminates the need for stoichiometric excesses of reagents found in prior art, utilizing merely 1.0 to 1.5 equivalents of ethyl trifluoroacetate. The subsequent alkylation with 2-chloro-5-chloromethylpyridine is facilitated by the use of polar aprotic solvents like N,N-dimethylformamide (DMF), ensuring high conversion rates. This strategic optimization allows for the production of the target compound in a short time with high yield, representing a paradigm shift in cost reduction in pest control agent manufacturing.
Mechanistic Insights into Trifluoroacetylation and Alkylation
The core of this synthetic strategy lies in the precise activation of the nucleophilic amino group on the pyridine ring. In Step 1, the metal base deprotonates the 2-aminopyridine or activates the trifluoroacetate, facilitating a nucleophilic attack that forms the stable amide bond in intermediate (B). The choice of solvent is paramount here; polar aprotic solvents like DMF, DMSO, or N-methyl-2-pyrrolidone stabilize the transition state and solubilize the ionic intermediates effectively. The reaction temperature is carefully controlled between 5°C and 55°C, with an optimal range of 25°C to 45°C, to prevent side reactions while maintaining kinetic energy for rapid conversion. This careful balance ensures that the intermediate (B) is formed with minimal impurities, setting the stage for the subsequent alkylation.
In Step 2, the nitrogen atom at the 1-position of the intermediate (B) undergoes alkylation with 2-chloro-5-chloromethylpyridine (C). The presence of the electron-withdrawing trifluoroacetyl group modulates the electronic density of the pyridine ring, influencing the nucleophilicity of the ring nitrogen. The reaction is typically conducted at elevated temperatures, preferably between 60°C and 70°C, to overcome the activation energy barrier for the substitution of the chloromethyl group. The use of DMF in this step is particularly advantageous as it supports the SN2 mechanism required for the alkylation. By avoiding the isolation of intermediate (B), the process minimizes material loss and exposure to atmospheric moisture, thereby preserving the integrity of the reactive species and ensuring the final product meets stringent purity specifications required for high-purity agrochemical intermediates.
How to Synthesize N-[1-((6-chloropyridin-3-yl)methyl)pyridin-2(1H)-ylidene]-2,2,2-trifluoroacetamide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the dissolution of 2-aminopyridine in a suitable solvent, followed by the controlled addition of the acylating agent and base. Critical to the success of this operation is the management of exothermic events during the base addition and the efficient removal of alcohol by-products (methanol or ethanol) generated during the acylation. Distilling off these alcohols under reduced pressure drives the equilibrium towards the formation of intermediate (B). Once the acylation is complete, the alkylating agent is introduced directly into the same vessel. This telescoping of steps not only saves time but also reduces the physical footprint required for production. For detailed operational parameters regarding stoichiometry and workup procedures, please refer to the standardized guide below.
- Acylation of 2-aminopyridine (Formula A) using ethyl trifluoroacetate and a metal base like sodium methoxide in DMF solvent.
- Optional distillation of alcohol by-products to drive the equilibrium forward before the next step.
- Alkylation of the intermediate (Formula B) with 2-chloro-5-chloromethylpyridine (Formula C) at elevated temperatures to yield Formula (I).
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the transition to this novel synthetic route offers tangible benefits beyond mere chemical yield. The ability to execute the synthesis in a single reaction vessel (one-pot method) drastically simplifies the manufacturing workflow, reducing the need for intermediate storage tanks and transfer operations. This consolidation of unit operations translates directly into lower capital expenditure (CAPEX) for reactor infrastructure and reduced operational expenditure (OPEX) related to labor and energy consumption. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which streamlines the purification process and reduces the volume of solvent waste generated. These factors collectively contribute to substantial cost savings and a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in reagent equivalents significantly lower the raw material bill of materials. By optimizing the stoichiometry of ethyl trifluoroacetate to near-equimolar ratios (1.0 to 1.5 equivalents), the process avoids the waste associated with large excesses typical in older methods. Additionally, the shortened reaction time—from days or 18 hours down to just a few hours—increases the asset turnover rate of the production facility, allowing for more batches to be produced within the same timeframe. This efficiency gain is a primary driver for overall cost reduction in agrochemical intermediate manufacturing, making the final product more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The starting materials for this process, including 2-aminopyridine, ethyl trifluoroacetate, and 2-chloro-5-chloromethylpyridine, are commercially available commodity chemicals with established supply chains. Unlike processes relying on exotic or custom-synthesized reagents, this method mitigates the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent grades, further ensures consistent production output even when minor variations in raw material quality occur. This reliability is crucial for maintaining continuous production schedules and reducing lead time for high-purity agrochemical intermediates, ensuring that downstream formulators receive their materials on time.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method through Example 22, where the process was successfully scaled to produce 12.60 kg of the target compound with a 93.8% yield. This proof of concept at the kilogram level indicates a smooth path to multi-ton commercial scale-up. Moreover, the process is environmentally friendlier as it avoids the generation of pyridinium salt waste, a common by-product in alternative routes. The use of recoverable solvents like DMF and the potential for solvent recycling further align the process with green chemistry principles and strict environmental regulations, reducing the liability and cost associated with waste disposal and environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN109715606B, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this method for their specific production needs. Whether the concern is regarding solvent selection, temperature sensitivity, or impurity profiles, the data supports the robustness of this new approach.
Q: What are the optimal reaction conditions for the acylation step?
A: The acylation step is optimally performed at temperatures between 25°C and 45°C using N,N-dimethylformamide (DMF) as the solvent and sodium methoxide or ethoxide as the base.
Q: Can this process be scaled for industrial production?
A: Yes, the patent demonstrates successful scale-up to kilogram levels (Example 22), achieving a 93.8% yield and 98.3% purity, confirming industrial viability.
Q: How does this method improve upon prior art?
A: Unlike prior methods requiring 18 hours or generating pyridinium salt waste, this method achieves high yields in significantly shorter times (e.g., 3 hours) using a one-pot approach.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-[1-((6-chloropyridin-3-yl)methyl)pyridin-2(1H)-ylidene]-2,2,2-trifluoroacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive agrochemical landscape. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN109715606B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with rigorous QC labs and advanced manufacturing facilities capable of meeting stringent purity specifications, ensuring that every batch of N-[1-((6-chloropyridin-3-yl)methyl)pyridin-2(1H)-ylidene]-2,2,2-trifluoroacetamide we deliver meets the highest industry standards. Our commitment to quality and consistency makes us the ideal partner for your long-term supply needs.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your transition to this high-efficiency production method is seamless and economically advantageous. Let us help you optimize your supply chain with our proven expertise in complex organic synthesis.
