Advanced Nickel-Catalyzed Synthesis of 1-Benzyloxy-4-Bromo-5-Ethyl-2-Fluorobenzene for Pharma
Advanced Nickel-Catalyzed Synthesis of 1-Benzyloxy-4-Bromo-5-Ethyl-2-Fluorobenzene for Pharma
The pharmaceutical industry's relentless pursuit of potent Janus Kinase (JAK) inhibitors has placed immense pressure on the supply chain for high-quality intermediates, specifically demanding robust and cost-effective synthetic routes for complex fluorinated aromatics. Patent CN114181055A introduces a groundbreaking preparation method for 1-benzyloxy-4-bromo-5-ethyl-2-fluorobenzene, a critical building block in the development of therapies for rheumatoid arthritis, myelofibrosis, and various malignancies. This innovation addresses the longstanding economic and technical bottlenecks associated with traditional palladium-catalyzed methodologies by leveraging a more abundant and economical nickel-based catalytic system. For R&D directors and procurement strategists, this patent represents a pivotal shift towards sustainable manufacturing, offering a pathway that not only simplifies operational complexity but also drastically reduces the dependency on precious metal catalysts. The disclosed route transforms p-bromofluorobenzene into the target molecule through a sequence of lithiation, protection, cross-coupling, and bromination, demonstrating exceptional potential for commercial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific fluorinated intermediate has been plagued by reliance on prohibitively expensive reagents and catalysts that strain project budgets and complicate waste management protocols. One prevalent prior art method, as illustrated in the reaction scheme below, relies heavily on the use of organozinc reagents coupled with palladium complexes to install the ethyl group onto the aromatic ring. This approach suffers from significant drawbacks, including the high cost of diethylzinc, the sensitivity of organozinc species to moisture and air which necessitates rigorous inert atmosphere handling, and the inherent expense of palladium catalysts which often require complex removal steps to meet stringent residual metal specifications in pharmaceutical products.
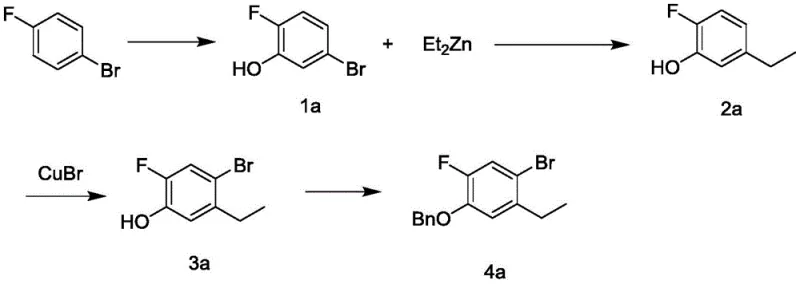
Furthermore, alternative synthetic strategies have attempted to construct the carbon skeleton starting from p-fluoroacetylene derivatives, followed by a reduction step to generate the ethyl side chain. However, this pathway introduces substantial risks regarding process safety and selectivity control, as the reduction of alkynyl groups can be difficult to manage on a large industrial scale without generating unwanted byproducts or over-reduced species. The combination of expensive starting materials and the operational hazards associated with alkyne chemistry renders these conventional methods suboptimal for the high-volume production required by the global demand for JAK inhibitors, creating a clear market opportunity for a more efficient alternative.
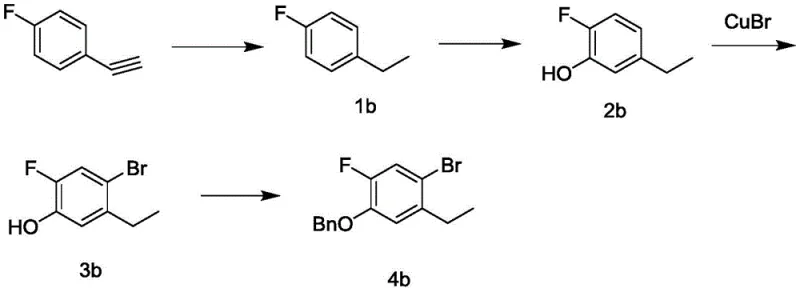
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN114181055A utilizes a nickel-complex catalyzed cross-coupling reaction that fundamentally alters the economic landscape of producing this intermediate. By substituting the costly palladium and organozinc reagents with a nickel catalyst and ethyl magnesium chloride (a Grignard reagent), the process achieves a dramatic reduction in raw material costs while maintaining high reaction efficiency and selectivity. The synthetic route begins with the conversion of p-bromofluorobenzene into a phenol intermediate, followed by protection, nickel-catalyzed ethylation, and a final regioselective bromination using copper bromide, as comprehensively depicted in the following reaction scheme.
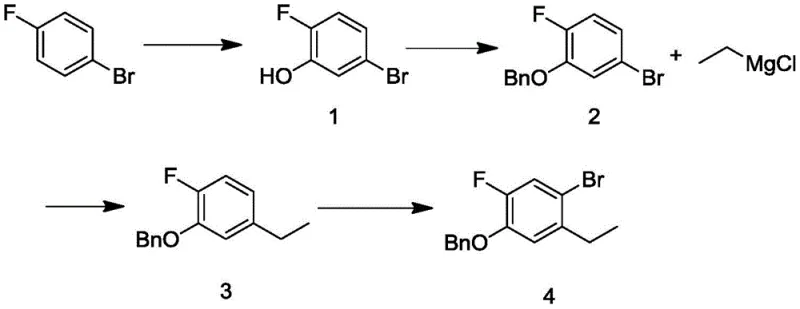
This strategic substitution not only lowers the direct cost of goods sold (COGS) but also simplifies the downstream purification processes, as nickel residues are generally easier to manage and remove compared to palladium traces in many organic matrices. The use of ethyl magnesium chloride offers a robust and widely available source of the ethyl group, eliminating the need for specialized organometallic reagents that require custom synthesis or importation. Consequently, this novel approach provides a reliable pharmaceutical intermediate supplier with a distinct competitive advantage, enabling the delivery of high-purity materials at a fraction of the cost associated with traditional palladium-mediated pathways.
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling and Bromination
The core of this technological advancement lies in the mechanistic efficiency of the nickel-catalyzed Kumada-type cross-coupling reaction, which facilitates the formation of the carbon-carbon bond between the aryl halide and the alkyl Grignard reagent. The nickel complex catalyst, such as 1,3-bis(diphenylphosphino)propane nickel dichloride [NiCl2(dppp)], undergoes a catalytic cycle involving oxidative addition of the aryl bromide, transmetallation with the ethyl magnesium chloride, and reductive elimination to release the ethyl-substituted product. This cycle is particularly advantageous for fluorinated substrates, as the specific ligand environment around the nickel center helps to preserve the sensitive carbon-fluorine bond while selectively activating the carbon-bromine bond for coupling, thereby preventing defluorination side reactions that could compromise the integrity of the final API.
Following the coupling step, the process employs a highly regioselective bromination using copper(II) bromide (CuBr2) to install the final bromine atom at the desired position on the aromatic ring. This step is critical for ensuring the correct substitution pattern required for subsequent coupling reactions in the JAK inhibitor synthesis. The mechanism likely involves an electrophilic aromatic substitution or a radical pathway mediated by the copper species, which directs the bromine to the position ortho to the benzyloxy group and meta to the fluorine, driven by the electronic effects of the existing substituents. This precise control over regiochemistry is essential for minimizing the formation of isomeric impurities, which are notoriously difficult to separate and can act as genotoxic alerts if carried through to the final drug substance, thus underscoring the importance of this specific bromination protocol for quality assurance.
How to Synthesize 1-Benzyloxy-4-Bromo-5-Ethyl-2-Fluorobenzene Efficiently
The practical execution of this synthesis involves a streamlined four-step sequence that balances reaction yield with operational simplicity, making it ideally suited for transfer from laboratory bench to pilot plant and eventually to commercial manufacturing scales. The process initiates with the lithiation of p-bromofluorobenzene using butyllithium at cryogenic temperatures, followed by quenching with trimethyl borate and oxidative workup to yield the phenol precursor, which is then protected as a benzyl ether to prevent interference during the subsequent metal-catalyzed steps. The critical nickel-catalyzed coupling is performed in tetrahydrofuran (THF) under inert atmosphere, where temperature control is maintained to ensure optimal conversion and minimize homocoupling byproducts, followed by a straightforward aqueous workup and solvent exchange for the final bromination step.
- Perform lithiation of p-bromofluorobenzene followed by borylation and oxidation to generate the phenol intermediate.
- Protect the phenol group via benzylation using benzyl bromide under alkaline conditions to form the ether.
- Execute a nickel-catalyzed cross-coupling reaction with ethyl magnesium chloride to introduce the ethyl group.
- Conduct regioselective bromination using copper(II) bromide to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple unit price reductions, impacting the overall resilience and agility of the pharmaceutical supply network. By eliminating the dependence on palladium catalysts and organozinc reagents, manufacturers can insulate their production costs from the volatile pricing fluctuations characteristic of precious metal markets, ensuring more stable long-term contracting and budget forecasting for key stakeholders. Furthermore, the use of commodity chemicals like ethyl magnesium chloride and copper bromide enhances supply chain security, as these reagents are produced in vast quantities globally, reducing the risk of shortages that frequently plague specialized fine chemical supply chains dependent on niche vendors.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with nickel complexes results in a substantial decrease in catalyst loading costs, which is a major component of the overall manufacturing expense for complex intermediates. Additionally, the avoidance of organozinc reagents removes the need for costly custom synthesis or specialized handling infrastructure, further driving down the operational expenditure associated with raw material procurement and storage. This economic efficiency allows for a more competitive pricing structure for the final API, enabling pharmaceutical companies to allocate resources towards clinical development and market expansion rather than being burdened by excessive material costs.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as p-bromofluorobenzene and benzyl bromide ensures a robust supply base that is less susceptible to geopolitical disruptions or single-source vendor failures. The simplified reaction conditions, which do not require extreme pressures or temperatures, also reduce the likelihood of unplanned plant shutdowns due to equipment stress or safety incidents, thereby guaranteeing consistent delivery schedules for downstream customers. This reliability is paramount for maintaining continuous clinical trial supplies and commercial inventory levels, preventing costly delays in drug launch timelines that can arise from intermediate shortages.
- Scalability and Environmental Compliance: The process utilizes common organic solvents like THF, ethyl acetate, and acetonitrile, which are well-understood in terms of recovery and recycling, facilitating compliance with increasingly stringent environmental regulations regarding solvent emissions and waste disposal. The high yields reported in the patent examples indicate a material-efficient process that minimizes waste generation per kilogram of product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing operation. This scalability ensures that the process can be seamlessly expanded from kilogram-scale development batches to multi-ton commercial production without the need for significant process re-engineering or capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity for technical teams evaluating the feasibility of adopting this new methodology for their supply chains. These insights are derived directly from the experimental data and technical disclosures within the patent, offering a realistic perspective on the capabilities and limitations of the described process for industrial application.
Q: Why is the nickel-catalyzed route preferred over palladium for this intermediate?
A: The nickel-catalyzed route significantly reduces raw material costs by avoiding expensive palladium complexes and organozinc reagents, while maintaining high yields suitable for industrial scale-up.
Q: What are the critical purity controls in this synthesis?
A: Critical controls include the regioselectivity during the initial lithiation and the final bromination step to prevent isomer formation, ensuring high purity for downstream JAK inhibitor synthesis.
Q: Is this process scalable for commercial production?
A: Yes, the process utilizes mild reaction conditions and commercially available solvents like THF and ethyl acetate, making it highly adaptable for large-scale manufacturing from 100 kgs to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyloxy-4-Bromo-5-Ethyl-2-Fluorobenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation JAK inhibitors, and we are uniquely positioned to leverage this patented technology to serve your manufacturing needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1-benzyloxy-4-bromo-5-ethyl-2-fluorobenzene meets the exacting standards required for pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply chain to drive significant value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements, along with access to specific COA data and route feasibility assessments tailored to your project timeline. Partnering with us ensures not only a reliable source of this critical intermediate but also a collaborative relationship focused on continuous improvement and mutual success in the competitive pharmaceutical landscape.
