Scalable Synthesis of 13C-Labeled Pyraoxystrobin for Advanced Agrochemical Analysis
The development of precise analytical standards is critical for modern agrochemical safety and regulatory compliance. Patent CN107674027B introduces a robust synthetic methodology for producing 13C-labeled pyraoxystrobin, a key fungicide belonging to the methoxyacrylate class. This innovation addresses the significant limitations of previous radioactive labeling techniques by utilizing stable carbon-13 isotopes, thereby enabling safer and more scalable production of reference materials. The disclosed route ensures high isotopic abundance and chemical purity, which are essential parameters for Isotope Dilution Mass Spectrometry (IDMS) applications used in detecting pesticide residues in food and environmental samples. By shifting from radioactive 14C to stable 13C labels, the industry gains a powerful tool for tracking metabolic pathways and environmental fate without the regulatory burdens associated with radioactivity.
This technical breakthrough is particularly relevant for laboratories and manufacturers requiring reliable agrochemical intermediate supplier capabilities for high-end analytical standards. The method described allows for the flexible introduction of the isotope label at multiple positions within the molecular framework, including the pyrazole ring and the methoxyacrylate side chain. Such versatility supports comprehensive metabolic studies and enhances the accuracy of quantitative analysis in complex matrices. Furthermore, the process avoids the use of exotic reagents, relying instead on established organic transformations that can be readily optimized for industrial throughput. This combination of safety, precision, and scalability represents a significant advancement in the field of stable isotope labeling synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of labeled pyraoxystrobin relied heavily on radioactive carbon-14 precursors, such as 14C-marked chlorobenzoic acid. While effective for trace-level research, these radioactive methods impose severe restrictions on operational scale and facility requirements. Handling radioactive materials necessitates specialized containment, rigorous safety protocols, and generates hazardous waste that is costly and difficult to dispose of. Consequently, these methods are unsuitable for batch production or routine quality control applications where larger quantities of the standard are needed. Additionally, alternative non-radioactive routes reported in literature often suffer from complex multi-step sequences, harsh reaction conditions, and low overall yields. These inefficiencies lead to significant impurity profiles that complicate downstream purification, ultimately reducing the reliability of the analytical standard produced.
The Novel Approach
The novel approach detailed in the patent overcomes these hurdles by employing a convergent synthesis strategy using stable 13C isotopes. This method utilizes readily available starting materials like chlorobenzene and methyl o-tolylacetate, introducing the label through controlled acylation or condensation steps. By avoiding radioactivity, the process eliminates the need for specialized radiation safety infrastructure, drastically simplifying the manufacturing workflow. The reaction conditions are mild, typically operating between 20°C and 100°C, which minimizes thermal degradation and side reactions. This results in a cleaner crude product that is easier to separate and purify, achieving chemical purity levels exceeding 98%. The strategic placement of the 13C label ensures that the internal standard behaves identically to the native analyte during extraction and ionization, correcting for matrix effects and recovery losses in LC-MS analysis.
Mechanistic Insights into Stable Isotope Incorporation
The core of this synthesis lies in the precise incorporation of the 13C isotope during the construction of the two main molecular fragments. In the first pathway, the pyrazole moiety is constructed starting with a Friedel-Crafts acylation of chlorobenzene. By using 13C-labeled acetyl chloride in the presence of aluminum trichloride, the carbonyl carbon of the resulting p-chloroacetophenone becomes labeled. This intermediate then undergoes a Claisen-type condensation with dimethyl carbonate under basic conditions, extending the carbon chain while preserving the isotopic label. Subsequent cyclization with methyl hydrazine forms the pyrazole ring, locking the label into the heterocyclic core. This sequence demonstrates high atom economy for the expensive labeled reagent, ensuring that the isotopic enrichment is maintained throughout the transformation without dilution from unlabeled carbon sources.
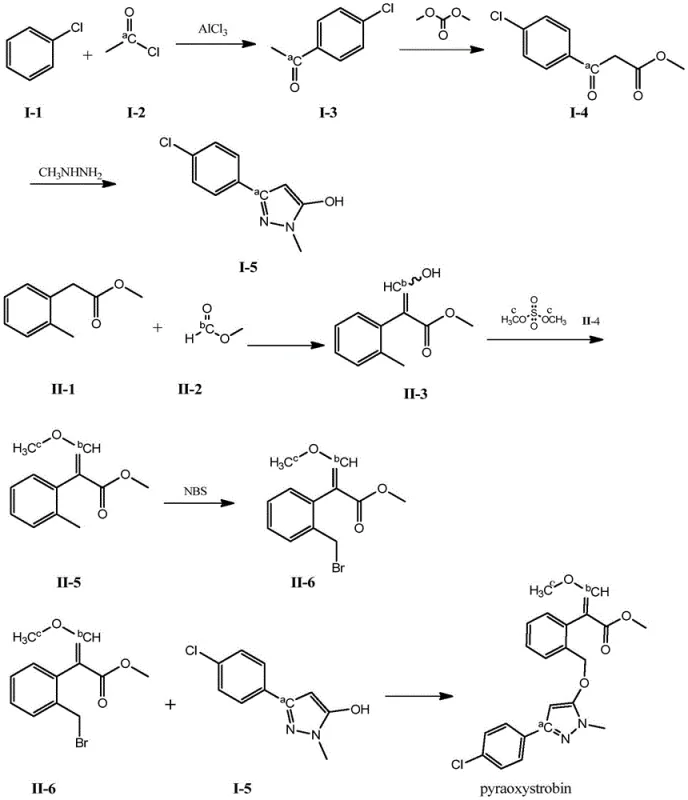
In the parallel pathway, the methoxyacrylate side chain is assembled from methyl o-tolylacetate. The introduction of the label here can occur at the formyl carbon via condensation with 13C-methyl formate or at the methoxy group via methylation with 13C-dimethyl sulfate. The condensation step generates a beta-hydroxy acrylate intermediate, which is subsequently methylated to protect the enol ether functionality. A critical step in this sequence is the radical bromination of the benzylic position using N-bromosuccinimide (NBS). This transformation activates the side chain for the final coupling reaction without disturbing the sensitive methoxyacrylate double bond geometry. The final convergence involves a nucleophilic substitution where the pyrazole alcohol attacks the benzylic bromide, forming the ether linkage that defines the pyraoxystrobin structure. This modular design allows for the synthesis of singly, doubly, or triply labeled variants depending on the specific analytical requirements.
How to Synthesize 13C-Labeled Pyraoxystrobin Efficiently
The synthesis of this high-value agrochemical intermediate requires strict control over reaction stoichiometry and temperature to maximize isotopic retention. The process is divided into the preparation of the pyrazole fragment and the acrylate fragment, followed by their coupling. Detailed operational parameters, including solvent choices like tetrahydrofuran and DMF, and base selection such as sodium hydride or potassium carbonate, are critical for success. The following guide outlines the standardized procedure derived from the patent examples to ensure reproducibility and high yield.
- Synthesize the pyrazole intermediate (I-5) by reacting chlorobenzene with 13C-labeled acetyl chloride via Friedel-Crafts acylation, followed by condensation with dimethyl carbonate and cyclization with methyl hydrazine.
- Prepare the bromide intermediate (II-6) by condensing methyl o-tolylacetate with 13C-labeled methyl formate, followed by methylation using 13C-labeled dimethyl sulfate and radical bromination with NBS.
- Couple the pyrazole alcohol (I-5) and the bromide intermediate (II-6) in an alkaline environment (e.g., NaH/DMF) to form the final 13C-labeled pyraoxystrobin with >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this stable isotope synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of radioactive precursors removes a major bottleneck in the supply chain, as sourcing 14C materials often involves long lead times and complex regulatory approvals. In contrast, 13C-labeled reagents like acetyl chloride and methyl formate are commercially available from multiple suppliers, enhancing supply security and reducing dependency on single-source vendors. This diversification of the raw material base significantly mitigates the risk of production delays caused by regulatory hold-ups or shortages of specialized radioactive isotopes. Furthermore, the simplified waste profile means that disposal costs are drastically reduced, contributing to a lower total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily through the high utilization rate of the stable isotope atoms. In many labeling syntheses, expensive labeled reagents are used in large excess or lost during purification, driving up costs. However, this method optimizes the molar ratios, such as using a 1:1.1 ratio of ketone to carbonate, to minimize waste of the premium labeled starting materials. Additionally, the avoidance of transition metal catalysts in the final coupling step eliminates the need for expensive metal scavenging processes, further streamlining the production budget. The high yield and purity reduce the volume of solvent and energy required for recrystallization and chromatography, leading to significant operational savings.
- Enhanced Supply Chain Reliability: By utilizing common organic solvents and reagents, the manufacturing process becomes far more resilient to market fluctuations. The reliance on standard chemicals like chlorobenzene and methyl o-tolylacetate ensures that production can be scaled rapidly to meet demand spikes without waiting for custom synthesis of exotic precursors. This robustness is crucial for maintaining continuity in the supply of analytical standards, which are often needed urgently for regulatory testing cycles. The ability to produce batch quantities rather than trace amounts also allows for better inventory management, enabling suppliers to stock finished goods and reduce lead times for customers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples. The reactions do not require extreme pressures or temperatures, making them compatible with standard glass-lined or stainless steel reactors found in most fine chemical plants. From an environmental perspective, the absence of radioactive waste simplifies compliance with environmental protection regulations. The process generates standard organic waste streams that can be treated using conventional incineration or recycling methods, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 13C-labeled pyraoxystrobin. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the advantages of using 13C-labeled pyraoxystrobin over 14C-labeled versions?
A: Unlike 14C-labeled compounds which are radioactive and require specialized handling facilities suitable only for trace research, 13C-labeled pyraoxystrobin is stable and safe. It allows for batch production and simplifies logistics while providing accurate mass spectrometry data for residue detection without radiation hazards.
Q: What is the chemical purity and isotopic abundance achievable with this method?
A: The synthetic method described in patent CN107674027B achieves a chemical purity of over 98% and an isotopic abundance exceeding 98%. This high level of purity ensures reliable performance as an internal standard in LC-MS analysis for pesticide residue detection.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the process utilizes common chemical raw materials like chlorobenzene and methyl o-tolylacetate, avoiding complex or hazardous reagents. The operation steps are simple, involving standard reactions like Friedel-Crafts acylation and nucleophilic substitution, making it highly suitable for pilot-scale and large-batch commercial preparation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyraoxystrobin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity analytical standards play in ensuring global food safety and environmental protection. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex labeled molecules can be manufactured with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced LC-MS and NMR instrumentation to verify isotopic abundance and chemical integrity. Our commitment to quality ensures that every batch of 13C-labeled pyraoxystrobin meets the demanding requirements of regulatory agencies and research institutions worldwide.
We invite you to collaborate with us to optimize your supply chain for agrochemical intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments for your next project. Let us help you secure a stable and cost-effective source of high-quality labeled compounds.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
