Advanced Synthesis of Piperonyl Ethylamine Salicylaldehyde for High-Purity Antitumor Intermediates
Advanced Synthesis of Piperonyl Ethylamine Salicylaldehyde for High-Purity Antitumor Intermediates
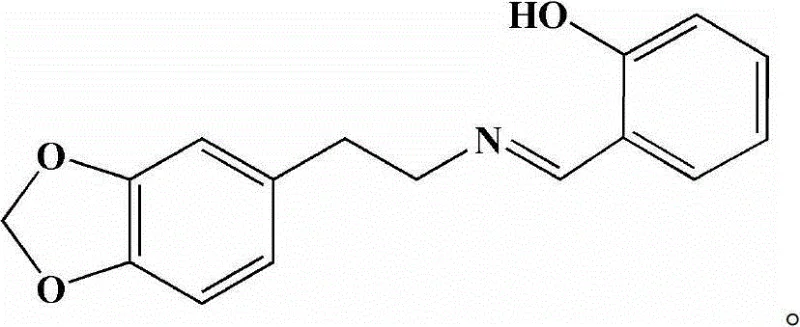
The pharmaceutical industry is constantly seeking robust and scalable pathways for novel antitumor agents, and patent CN102942554B presents a significant breakthrough in this domain with the introduction of piperonyl ethylamine salicylaldehyde. This novel Schiff base compound is synthesized through a highly efficient condensation reaction between salicylaldehyde and homopiperonylamine, offering a streamlined route that bypasses the complexities often associated with traditional heterocyclic synthesis. The structural integrity of this molecule, characterized by its distinctive imine linkage and functional groups, is critical for its observed biological activity, particularly its selectivity against specific human tumor cell lines. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis allows us to deliver high-purity materials that meet the rigorous demands of modern drug discovery pipelines. The following analysis details the technical merits and commercial viability of this patented methodology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing complex Schiff bases often suffer from苛刻 reaction conditions that necessitate the use of expensive transition metal catalysts or harsh acidic environments, which can lead to significant impurity profiles and difficult downstream purification processes. Many conventional routes require extended reaction times at elevated temperatures that risk thermal degradation of sensitive functional groups, thereby reducing the overall yield and compromising the quality of the final active pharmaceutical ingredient. Furthermore, the reliance on stoichiometric amounts of dehydrating agents that are difficult to remove can introduce toxic residues, creating substantial regulatory hurdles for clinical applications. The lack of selectivity in older methodologies often results in the formation of polymeric byproducts or oligomers, requiring multiple recrystallization steps that drastically increase production costs and waste generation. These inefficiencies create bottlenecks in the supply chain, making it challenging to secure consistent quantities of high-quality intermediates for large-scale manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102942554B utilizes a mild and direct condensation strategy that operates effectively at temperatures ranging from 40°C to the boiling point of the chosen organic solvent. By employing simple, commercially available solvents such as ethanol or dichloromethane in conjunction with molecular sieves for water removal, the process eliminates the need for complex catalytic systems while driving the equilibrium towards product formation. This approach not only simplifies the operational workflow but also significantly enhances the safety profile of the manufacturing process by avoiding hazardous reagents. The reaction proceeds to completion within a reasonable timeframe of 1 to 12 hours, as monitored by thin-layer chromatography, allowing for precise control over the reaction endpoint to minimize side reactions. The resulting product precipitates readily upon cooling, facilitating easy isolation through filtration and yielding a high-purity solid that requires minimal further processing.
Mechanistic Insights into Schiff Base Condensation
The core of this synthesis lies in the nucleophilic attack of the primary amine group of homopiperonylamine on the carbonyl carbon of salicylaldehyde, forming a tetrahedral intermediate that subsequently undergoes dehydration to establish the stable carbon-nitrogen double bond characteristic of Schiff bases. The presence of the ortho-hydroxyl group on the salicylaldehyde moiety plays a pivotal role in stabilizing the resulting imine through intramolecular hydrogen bonding, which contributes to the thermodynamic stability of the final product. The use of molecular sieves, specifically 4Å types, is mechanistically crucial as they sequester the water byproduct generated during the condensation, effectively shifting the chemical equilibrium towards the right according to Le Chatelier's principle. This dehydration step is vital for preventing the hydrolysis of the imine bond, ensuring that the reaction proceeds to high conversion rates without the need for excessive heat or pressure. Understanding this mechanism allows process chemists to optimize solvent choices and drying agent loading to maximize efficiency and reproducibility.
Impurity control in this system is primarily managed through the selection of high-purity starting materials and the optimization of the crystallization phase, where the specific solubility characteristics of the target compound are exploited. The patent data indicates that the product forms distinct yellow needle-like crystals, a morphological feature that suggests a high degree of lattice order and purity, which is essential for consistent biological performance. By controlling the cooling rate and the final temperature of the crystallization process, operators can influence the crystal size distribution, which in turn affects the filtration efficiency and the residual solvent content in the dried product. Rigorous quality control measures, including NMR and mass spectrometry, confirm the absence of unreacted aldehydes or amines, ensuring that the impurity profile remains well within the stringent limits required for pharmaceutical intermediates. This level of control is indispensable for maintaining the batch-to-batch consistency required by global regulatory agencies.
How to Synthesize Piperonyl Ethylamine Salicylaldehyde Efficiently
To achieve optimal results in the production of this valuable intermediate, it is essential to adhere to the standardized protocol derived from the patent examples, which emphasizes the importance of solvent dehydration and temperature control. The process begins with the precise weighing of equimolar quantities of salicylaldehyde and homopiperonylamine, ensuring that the stoichiometry favors the formation of the mono-condensed product without excess reagents that could complicate purification. Dissolving these reactants in a pre-dried organic solvent creates a homogeneous reaction medium where molecular collisions occur frequently enough to drive the kinetics forward at a practical rate. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve equimolar amounts of salicylaldehyde and homopiperonylamine in a dehydrated organic solvent such as ethanol or dichloromethane.
- Heat the mixture to a temperature between 40°C and the solvent's boiling point (preferably 60-70°C) and stir for 1 to 12 hours until TLC indicates completion.
- Cool the reaction mixture to 10-25°C to induce crystallization, filter the resulting yellow needle-like crystals, and vacuum dry at 25-50°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers substantial strategic benefits by leveraging widely available and cost-effective raw materials that are not subject to volatile market fluctuations. The elimination of precious metal catalysts removes a significant cost center associated with both the initial purchase of the catalyst and the subsequent validation of its removal from the final product. This simplification of the bill of materials translates directly into a more predictable cost structure, allowing procurement managers to negotiate better long-term contracts and secure supply continuity for critical drug development programs. Additionally, the use of common industrial solvents reduces the logistical complexity and safety risks associated with transporting and storing hazardous specialty chemicals. These factors combined create a resilient supply chain capable of withstanding external disruptions while maintaining competitive pricing.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing a catalyst-free system that relies on thermal energy and molecular sieves, which are inexpensive and regenerable consumables. By avoiding the use of expensive transition metals, the downstream processing costs related to metal scavenging and validation are completely eliminated, leading to a leaner manufacturing budget. The high yields reported, ranging from 70% to 90%, mean that less raw material is wasted per unit of product, further enhancing the overall economic efficiency of the production line. This economic advantage allows for more aggressive pricing strategies in the competitive landscape of pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as salicylaldehyde and ethanol ensures that the supply chain is not vulnerable to the shortages that often plague specialized reagents. Since the reaction conditions are mild and do not require exotic equipment, the process can be easily transferred between different manufacturing sites or contract organizations, providing flexibility in sourcing. The robustness of the crystallization step ensures that the final product is stable and easy to transport, reducing the risk of degradation during logistics. This reliability is crucial for maintaining the timelines of clinical trials and commercial launches where delays can be prohibitively expensive.
- Scalability and Environmental Compliance: The simplicity of the reaction setup allows for seamless scale-up from laboratory benchtop quantities to multi-ton commercial production without the need for complex engineering modifications. The reduced use of hazardous reagents and the ability to recover and recycle solvents align with green chemistry principles, minimizing the environmental footprint of the manufacturing process. Waste generation is minimized due to the high atom economy of the condensation reaction and the efficient recovery of the product through crystallization. This compliance with environmental standards facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of piperonyl ethylamine salicylaldehyde, based on the detailed data provided in the patent literature. These answers are designed to provide clarity on the feasibility of the process and the quality attributes of the resulting material for potential partners. We encourage stakeholders to review these insights to understand how this technology can be integrated into their existing workflows.
Q: What is the expected yield for the synthesis of piperonyl ethylamine salicylaldehyde?
A: According to patent CN102942554B, the synthesis method consistently achieves yields ranging from 70% to 90%, depending on the specific solvent system and reaction time employed.
Q: Does this compound show activity against cisplatin-resistant cancer cells?
A: Yes, biological testing indicates significant proliferation inhibitory activity against the SK-OV-3/DDP cisplatin-resistant ovarian cancer cell line, suggesting a mechanism distinct from cisplatin.
Q: What solvents are suitable for this condensation reaction?
A: The process is flexible and supports various organic solvents including dichloromethane, ethanol, methanol, and chloroform, provided they are adequately dehydrated using molecular sieves.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperonyl Ethylamine Salicylaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antitumor therapies, and we are fully equipped to support your needs with this advanced synthesis platform. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of piperonyl ethylamine salicylaldehyde meets the highest international standards for pharmaceutical use. Our commitment to technical excellence ensures that you receive a product that is consistent, reliable, and ready for immediate integration into your drug substance manufacturing.
We invite you to contact our technical procurement team to discuss how we can assist in optimizing your supply chain for this specific intermediate. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into tangible value for your organization. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to be your long-term strategic partner. Let us collaborate to bring this promising antitumor candidate from the laboratory to the clinic efficiently and effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
