Advanced Hydroformylation Strategy for High-Purity 3,3-Diarylpropylamines Manufacturing
The pharmaceutical industry continuously seeks more efficient pathways to synthesize complex amine structures, particularly those serving as critical intermediates for treating chronic conditions such as urinary incontinence. Patent CN1441771A introduces a groundbreaking methodology for producing 3,3-diarylpropylamines of formula (I), utilizing a sophisticated hydroformylation and subsequent reductive amination sequence. This technical advancement represents a significant departure from traditional multi-step syntheses, offering a streamlined approach that enhances both atomic economy and process safety. By leveraging transition metal catalysis, specifically rhodium-based systems, this method facilitates the direct conversion of 1,1-diarylethylene derivatives into valuable amine products with high precision. The strategic integration of hydroformylation allows for the construction of the propylamine backbone in a single operational phase, drastically reducing the environmental footprint and operational complexity typically associated with building such sterically hindered molecular architectures. For R&D teams focused on optimizing synthetic routes for API intermediates, this patent provides a robust framework for achieving superior purity profiles while minimizing waste generation.
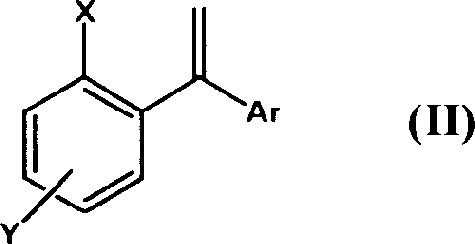
The structural versatility of the substrates accepted by this novel method is extensive, accommodating various substituted or unsubstituted aryl radicals and functional groups such as hydroxyl, alkoxy, or halogen substituents. This flexibility is crucial for medicinal chemists who require diverse analog libraries to explore structure-activity relationships without being constrained by rigid synthetic limitations. The ability to tolerate sensitive functional groups during the high-pressure hydroformylation step underscores the mildness and selectivity of the catalytic system employed. Furthermore, the resulting 3,3-diarylpropylamines serve as pivotal precursors for a wide range of therapeutic agents, extending beyond urological treatments to potential applications in central nervous system disorders. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented technologies enables us to offer clients not just materials, but strategic synthesis solutions that align with their long-term development goals. The adoption of this technology signifies a move towards more sustainable and economically viable manufacturing practices in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-diarylpropylamine derivatives has been plagued by inefficient multi-step protocols that often involve hazardous reagents and cumbersome purification processes. Traditional routes frequently rely on the alkylation of amines with corresponding halides or the reduction of amides, methods that suffer from poor atom economy and the generation of substantial stoichiometric waste. Moreover, achieving the desired stereochemistry in these conventional pathways often necessitates costly resolution steps or the use of chiral pool starting materials, which significantly drives up the cost of goods sold. The requirement for enantiomer separation in many prior art methods introduces additional unit operations, increasing the risk of yield loss and extending the overall production lead time. These inefficiencies create bottlenecks in the supply chain, making it difficult to scale production to meet commercial demand without compromising on quality or cost. Additionally, the use of strong bases or aggressive reducing agents in older methodologies poses safety risks and complicates waste disposal, further burdening the manufacturing infrastructure with regulatory compliance challenges.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct hydroformylation of 1,1-diarylethylenes followed by reductive amination, effectively collapsing multiple synthetic transformations into a more cohesive workflow. This strategy eliminates the need for pre-functionalized alkylating agents, thereby reducing the raw material costs and simplifying the logistics of precursor sourcing. By employing a transition metal catalyst, the reaction proceeds under controlled conditions that favor the formation of the desired linear or branched isomers with high selectivity, minimizing the formation of difficult-to-remove byproducts. The integration of the amination step allows for the direct introduction of the nitrogen functionality, bypassing the need for intermediate isolation and handling. This telescoped process not only accelerates the timeline from laboratory bench to pilot plant but also enhances the overall safety profile of the manufacturing operation. For procurement managers, this translates into a more stable supply of high-purity intermediates with reduced vulnerability to raw material price fluctuations associated with complex multi-step syntheses.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation
The core of this innovative synthesis lies in the intricate catalytic cycle driven by rhodium complexes in the presence of specific phosphine ligands. The reaction initiates with the coordination of the 1,1-diarylethylene substrate to the active rhodium species, followed by the insertion of carbon monoxide and hydrogen to form an acyl-rhodium intermediate. This step is critical as it determines the regioselectivity of the hydroformylation, influencing whether the formyl group adds to the terminal or internal position of the double bond. The choice of ligand, such as BINAP or tributylphosphine, plays a pivotal role in modulating the electronic and steric environment around the metal center, thereby steering the reaction towards the desired product distribution. Subsequent hydrogenolysis of the acyl intermediate releases the aldehyde, which then undergoes reductive amination with the added amine component. This mechanistic pathway ensures that the carbon-carbon bond formation and carbon-nitrogen bond formation are seamlessly integrated, maximizing the efficiency of the transformation. Understanding these mechanistic details is essential for process chemists aiming to optimize reaction parameters such as temperature, pressure, and gas ratios for maximum throughput.
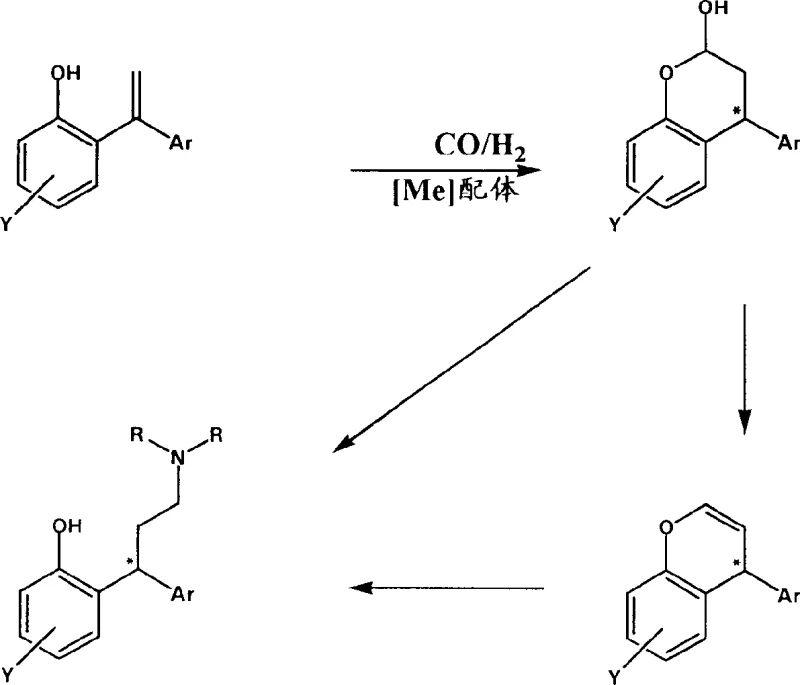
Impurity control is another critical aspect where this mechanistic understanding provides significant advantages. By carefully tuning the catalyst system and reaction conditions, the formation of side products such as hydrogenated alkenes or over-carbonylated species can be suppressed to negligible levels. The patent highlights the use of specific ligand-to-metal ratios to maintain catalyst stability throughout the reaction duration, preventing the decomposition of the active species which could lead to metal contamination in the final product. For R&D directors concerned with impurity profiles, this level of control is paramount, as it reduces the burden on downstream purification units like chromatography or crystallization. The ability to produce optically active compounds through ligand control or substrate control further enhances the value proposition, allowing for the direct synthesis of chiral intermediates without the need for external resolution. This precision in mechanistic execution ensures that the final API intermediate meets the stringent quality standards required by global regulatory bodies.
How to Synthesize 3,3-Diarylpropylamines Efficiently
The practical implementation of this synthesis route involves preparing a reaction mixture containing the 1,1-diarylethylene substrate, a rhodium catalyst precursor, and a suitable amine in an anhydrous solvent. The system is then pressurized with a specific ratio of carbon monoxide and hydrogen gas and heated to temperatures ranging from 100°C to 140°C to drive the hydroformylation and amination sequence. Detailed standardized synthesis steps see below guide.
- Prepare the 1,1-diarylethylene substrate and mix with a rhodium catalyst precursor and suitable phosphine ligand in an anhydrous solvent.
- Introduce a carbon monoxide and hydrogen gas mixture into the pressurized vessel and heat to 100-140°C to initiate hydroformylation.
- Perform subsequent reductive amination directly in the same vessel or after isolation of the oxo-intermediate to yield the final diarylpropylamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this hydroformylation technology offers profound benefits for procurement and supply chain management teams seeking to optimize their sourcing strategies. The reduction in synthetic steps directly correlates to a decrease in manufacturing overhead, as fewer unit operations mean lower energy consumption and reduced labor requirements. This streamlining of the process inherently leads to substantial cost savings, making the final intermediate more competitive in the global market without sacrificing quality. Furthermore, the reliance on readily available starting materials such as styrenes and benzophenones ensures a robust supply chain that is less susceptible to disruptions caused by scarce reagents. For supply chain heads, this reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical manufacturers. The scalability of the high-pressure hydroformylation process is well-established in the chemical industry, allowing for seamless transition from pilot scale to full commercial production.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps significantly lowers the operational expenditure associated with solvent usage and waste treatment. By avoiding the use of expensive chiral resolving agents or harsh alkylating reagents, the overall material cost is drastically reduced, providing a clear economic advantage over legacy synthetic routes. This efficiency allows for better margin management and the ability to offer more competitive pricing to partners while maintaining high profitability. The simplified process flow also reduces the capital investment required for specialized equipment, further enhancing the return on investment for manufacturing facilities adopting this technology.
- Enhanced Supply Chain Reliability: Utilizing common industrial feedstocks like syngas and simple olefins ensures that the production of these intermediates is not bottlenecked by the availability of exotic or regulated chemicals. This accessibility translates to shorter lead times and greater flexibility in responding to sudden increases in market demand. The robust nature of the rhodium catalyst system means that batch-to-batch consistency is high, reducing the risk of production delays due to failed quality control tests. For procurement managers, this reliability minimizes the need for safety stock and allows for leaner inventory management strategies, freeing up working capital for other strategic initiatives.
- Scalability and Environmental Compliance: The process is designed to be inherently scalable, utilizing standard high-pressure reactors that are common in fine chemical manufacturing plants. This compatibility with existing infrastructure facilitates rapid technology transfer and scale-up without the need for extensive retrofitting. Additionally, the improved atom economy and reduced waste generation align with modern environmental, social, and governance (ESG) goals, making it easier to comply with increasingly stringent environmental regulations. The lower environmental footprint not only mitigates regulatory risk but also enhances the brand reputation of the manufacturing entity as a sustainable partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,3-diarylpropylamines using this advanced hydroformylation technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of this synthetic route for potential partners. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their development pipelines.
Q: What are the primary advantages of this hydroformylation route over traditional alkylation?
A: This method significantly reduces the number of synthetic steps by combining hydroformylation and reductive amination, eliminating the need for harsh alkylating agents and complex purification sequences associated with conventional multi-step syntheses.
Q: Can this process achieve high stereoselectivity for chiral drug intermediates?
A: Yes, by utilizing chiral phosphine ligands such as BINAP or BINAPHOS during the catalytic cycle, the process allows for precise stereocontrol, enabling the production of specific enantiomers required for high-efficacy pharmaceutical applications.
Q: Is this technology scalable for commercial API production?
A: The process operates under standard high-pressure industrial conditions using readily available syngas and robust rhodium catalysts, making it highly suitable for large-scale commercial manufacturing with consistent quality and supply reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Diarylpropylamines Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. Our technical team is adept at implementing complex catalytic processes like the rhodium-mediated hydroformylation described in CN1441771A, guaranteeing stringent purity specifications and rigorous QC labs oversight for every batch. We understand the critical nature of API intermediates in the drug development timeline and are committed to delivering materials that meet the highest international standards. Our facility is equipped to handle high-pressure reactions safely and efficiently, providing a secure environment for the manufacture of sensitive pharmaceutical compounds. Partnering with us means gaining access to a wealth of chemical expertise and a dedication to quality that supports your regulatory filings and commercial success.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By engaging with us early in your development process, you can leverage our process optimization capabilities to secure specific COA data and route feasibility assessments that de-risk your supply chain. Let us demonstrate how our advanced manufacturing capabilities can support your goal of bringing life-saving medications to market faster and more cost-effectively. Reach out today to discuss how we can become your trusted partner in the synthesis of high-value pharmaceutical intermediates.
