Advanced Synthesis Of Ionone-Based Thiosemicarbazones For Pharma And Green Technology Applications
Advanced Synthesis Of Ionone-Based Thiosemicarbazones For Pharma And Green Technology Applications
The chemical landscape is continuously evolving with the discovery of multifunctional intermediates that bridge the gap between traditional medicinal chemistry and emerging green technologies. Patent CN102942514B introduces a sophisticated class of compounds known as ionone-based bis-chalcone thiosemicarbazones, which represent a significant advancement in organic synthesis. These molecules are characterized by their unique structural framework combining ionone derivatives with chalcone backbones and thiosemicarbazide moieties. The patent details a robust production method that leverages a direct condensation reaction, offering a streamlined pathway for generating high-value chemical entities. This technology is particularly noteworthy for its dual applicability, serving as potent bioactive agents in the pharmaceutical sector while simultaneously addressing critical challenges in the recycling of lithium-ion battery materials. For industry leaders seeking reliable pharmaceutical intermediate supplier partnerships, understanding the nuances of this synthesis is essential for securing supply chains in both healthcare and sustainable energy sectors.
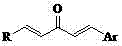
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for complex heterocyclic compounds often suffer from excessive step counts, requiring harsh reaction conditions that can degrade sensitive functional groups. In the context of chalcone derivatives, conventional methods frequently involve multiple protection and deprotection steps to manage the reactivity of the carbonyl and olefinic groups. Furthermore, older methodologies might rely on toxic solvents or expensive transition metal catalysts that introduce heavy metal impurities, necessitating costly purification processes to meet stringent regulatory standards for high-purity pharmaceutical intermediates. The lack of atom economy in these traditional pathways not only inflates production costs but also generates significant chemical waste, posing environmental compliance challenges for modern manufacturing facilities. Additionally, the scalability of such complex multi-step syntheses is often limited by low overall yields and difficult isolation procedures, creating bottlenecks in the supply chain for bulk chemical procurement.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by utilizing a direct condensation strategy between specific chalcone precursors and thiosemicarbazide. This approach eliminates the need for complex catalytic systems, relying instead on a mild acid-catalyzed mechanism in a benign ethanol solvent system. By carefully controlling the reaction parameters, specifically maintaining a pH of 5 with glacial acetic acid, the process achieves high selectivity for the desired thiosemicarbazone linkage without compromising the integrity of the ionone ring system. This simplification of the synthetic route drastically reduces the operational complexity, allowing for easier commercial scale-up of complex polymer additives and fine chemicals. The use of readily available starting materials and a single-step reaction vessel significantly lowers the barrier to entry for manufacturing, ensuring a more consistent and reliable supply of these specialized compounds for downstream applications in drug discovery and material science.
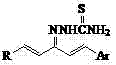
Mechanistic Insights into Acid-Catalyzed Condensation
The core of this synthesis lies in the nucleophilic addition-elimination mechanism facilitated by the acidic environment. In this reaction, the terminal amino group of the thiosemicarbazide acts as a nucleophile, attacking the electrophilic carbonyl carbon of the chalcone precursor. The presence of glacial acetic acid serves a dual purpose: it protonates the carbonyl oxygen to enhance its electrophilicity and helps in the subsequent dehydration step to form the stable C=N double bond characteristic of the thiosemicarbazone structure. The specific choice of ethanol as a solvent is critical, as it provides excellent solubility for both organic reactants while allowing for easy removal post-reaction via rotary evaporation. The reaction temperature, maintained at reflux, provides the necessary activation energy to overcome the kinetic barrier of the condensation without inducing thermal decomposition of the sensitive conjugated systems present in the ionone-chalcone backbone.
Impurity control in this process is inherently managed through the stoichiometry and the crystallization behavior of the product. The patent specifies a precise 1:1 molar ratio of reactants, which minimizes the formation of side products derived from excess reagents. Furthermore, the final purification step involves recrystallization from 95% ethanol, a technique that effectively removes unreacted starting materials and minor byproducts based on solubility differences. This physical purification method is superior to chromatographic techniques for large-scale production as it is cost-effective and scalable. The resulting solids, described as ranging from milky white to dark green depending on the substituents, exhibit sharp melting points, indicating high chemical purity. This level of purity is paramount for reducing lead time for high-purity pharmaceutical intermediates intended for biological testing, where trace impurities can skew assay results and compromise safety profiles.
How to Synthesize Ionone-based Bis-chalcone Thiosemicarbazone Efficiently
The synthesis protocol described offers a reproducible framework for generating these valuable compounds with consistent quality. The process begins with the precise weighing of chalcone and thiosemicarbazide, ensuring the stoichiometric balance required for optimal yield. The reaction mixture is then subjected to controlled heating under reflux conditions, allowing the condensation to proceed to completion over a defined period. Following the reaction, the workup procedure is designed to maximize recovery, utilizing cooling to induce precipitation followed by filtration. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Mix chalcone and thiosemicarbazide in a 1: 1 molar ratio within a three-necked flask.
- Add anhydrous ethanol at a 43: 1 molar ratio relative to chalcone and adjust pH to 5 using glacial acetic acid.
- Reflux the mixture for 4 hours, then remove solvent, cool to precipitate solids, and recrystallize from 95% ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis route offers substantial benefits regarding cost efficiency and supply chain resilience. The reliance on commodity chemicals such as ethanol and acetic acid, rather than specialized proprietary catalysts, insulates the production cost from volatile market fluctuations associated with rare earth metals or complex ligands. This stability allows for more accurate long-term budgeting and pricing strategies for cost reduction in pharmaceutical intermediates manufacturing. Moreover, the simplicity of the reaction setup means that production can be easily distributed across multiple manufacturing sites, reducing the risk of supply disruption due to localized events. The ability to produce these compounds in a single reactor vessel also minimizes the footprint required for manufacturing, optimizing facility utilization rates and lowering overhead costs associated with plant maintenance and operation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of a single solvent system significantly lower the raw material costs per kilogram of product. By avoiding complex purification steps like column chromatography and relying on simple recrystallization, the operational expenditure related to solvent consumption and waste disposal is drastically reduced. This lean manufacturing approach translates directly into improved margins for buyers, enabling more competitive pricing for end-user applications in both the pharmaceutical and agricultural sectors without compromising on the quality or purity specifications of the final active ingredient.
- Enhanced Supply Chain Reliability: The starting materials, specifically various substituted chalcones and thiosemicarbazide, are widely available from global chemical suppliers, ensuring a robust upstream supply chain. This accessibility mitigates the risk of raw material shortages that often plague specialty chemical production. Furthermore, the reaction conditions are mild and do not require specialized high-pressure equipment or cryogenic cooling, meaning that production can be ramped up quickly in response to surges in demand. This flexibility is crucial for maintaining continuity of supply for critical drug development programs and industrial processes that depend on timely delivery of key intermediates.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous ethanol streams that can be easily treated or recycled, aligning with modern green chemistry principles. The absence of heavy metals simplifies the environmental compliance burden, reducing the costs and time associated with effluent treatment and regulatory reporting. This environmental friendliness enhances the sustainability profile of the supply chain, appealing to corporate social responsibility goals. Additionally, the straightforward nature of the chemistry ensures that scaling from pilot plant to full commercial production involves minimal re-engineering, facilitating a smoother transition to large-volume manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized thiosemicarbazone derivatives. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for integrating these materials into your specific R&D pipelines or manufacturing processes effectively.
Q: What are the primary applications of ionone-based bis-chalcone thiosemicarbazones?
A: These compounds exhibit significant biological activities including antibacterial, antiviral, and antitumor properties, making them valuable pharmaceutical intermediates. Additionally, specific derivatives serve as highly effective flotation agents for recovering lithium cobalt oxide from waste batteries.
Q: How does the pH level affect the condensation reaction yield?
A: Maintaining a pH of 5 using glacial acetic acid is critical for optimizing the nucleophilic attack of thiosemicarbazide on the chalcone carbonyl group, ensuring high conversion rates without degrading the sensitive ionone moiety.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes common solvents like ethanol and standard reflux conditions, avoiding expensive catalysts or extreme pressures, which facilitates straightforward scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ionone-based Bis-chalcone Thiosemicarbazone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug discovery and advancing material science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ionone-based thiosemicarbazone meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent material performance, whether you are conducting initial biological screening or preparing for clinical trial manufacturing.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and purity requirements. Please contact us today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner dedicated to driving efficiency and innovation in the fine chemical industry, ensuring your projects proceed without interruption.
