Advanced Synthesis Of Hydrazide-Containing Hindered Amines For Industrial Polymer Stabilization
The chemical industry is constantly evolving, driven by the need for materials that offer superior performance and longevity in demanding environments. Patent CN108586321B introduces a significant breakthrough in the field of light stabilizers, specifically focusing on a novel hindered amine compound containing a hydrazide group. This innovation addresses the limitations of traditional Hindered Amine Light Stabilizers (HALS) by modifying the core chemical structure to enhance oxidation resistance and aging resistance. The patent details a robust synthetic route that begins with the dehydration condensation of diacid dihydrazide and triacetoneamine, followed by a critical hydrogenation step. This technical advancement is not merely an academic exercise but represents a tangible opportunity for manufacturers of polymer additives to improve the durability of polyolefins, ABS resins, and other high-grade materials. By integrating a hydrazide moiety, the resulting compound exhibits unique basicity properties that outperform conventional stabilizers in rigorous testing scenarios, marking a pivotal shift in how we approach polymer stabilization chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing hindered amine light stabilizers often rely on esterification reactions or the substitution of amino groups on piperidine rings, which can present significant challenges in terms of cost and efficiency. For instance, the production of well-known stabilizers like Tinuvin 770 involves transesterification reactions that require high temperatures and specific catalysts, potentially leading to higher energy consumption and complex post-processing requirements. Furthermore, conventional routes may struggle with the balance between alkalinity and stability; reducing alkalinity to prevent salt formation in polymers can inadvertently compromise the light-stabilizing efficacy, as seen in certain commercial products where reduced basicity led to diminished performance. The reliance on expensive catalysts in older patents, such as those utilizing specific alkali metal salts, adds to the production cost, while the formation of by-products during high-temperature reactions can complicate purification processes, ultimately affecting the purity and consistency of the final additive supplied to the market.
The Novel Approach
In contrast, the methodology outlined in CN108586321B leverages the high reactivity and stability of diacid dihydrazide compounds to create a more efficient synthesis pathway. By reacting diacid dihydrazide with triacetoneamine under acid catalysis, the process achieves a dehydration condensation that yields an acylhydrazone intermediate with minimal by-product formation. This approach avoids the harsh conditions associated with traditional transesterification, operating at moderate temperatures that preserve the integrity of the reactants. The subsequent hydrogenation step is equally optimized, utilizing readily available catalysts to reduce the imine groups effectively. This novel route not only simplifies the operational workflow but also enhances the overall yield, with the patent reporting condensation yields exceeding 93 percent. The structural innovation of introducing a hydrazide group allows for a fine-tuning of the compound's basicity, ensuring that it maintains high light-stabilizing performance without the drawbacks associated with earlier generations of HALS chemistry.
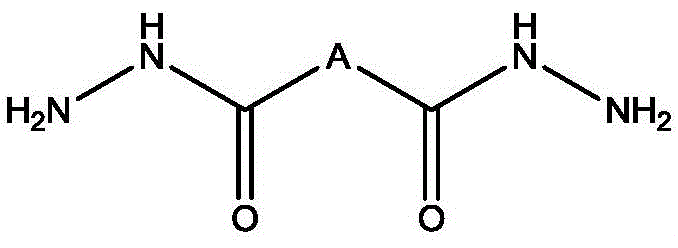
Mechanistic Insights into Acid-Catalyzed Condensation and Hydrogenation
The core of this technological advancement lies in the precise mechanistic control of the condensation and reduction steps. The reaction begins with the nucleophilic attack of the hydrazide nitrogen on the carbonyl carbon of the triacetoneamine, facilitated by an acid catalyst such as p-toluenesulfonic acid or acetic acid. This acid catalysis is crucial for activating the carbonyl group and promoting the elimination of water, which is continuously removed via azeotropic distillation to drive the equilibrium towards the product side. The choice of solvent, ranging from toluene to xylene, is strategically selected to ensure immiscibility with water, allowing for efficient phase separation and water removal. This meticulous control over the reaction environment minimizes side reactions and ensures that the acylhydrazone intermediate is formed with high selectivity. The structural integrity of the hindered amine skeleton is preserved throughout this process, setting the stage for the final reduction step where the imine bond is converted to a stable amine linkage.
Following the condensation, the hydrogenation mechanism plays a pivotal role in defining the final product's purity and performance. The acylhydrazone intermediate is subjected to catalytic hydrogenation using supported noble metals like Pd/C or skeletal metals like Raney Nickel. This step requires careful control of pressure and temperature to ensure complete reduction without over-hydrogenation or degradation of the sensitive hydrazide functionality. The patent specifies conditions such as 0.5 to 1.0 MPa pressure and temperatures between 50 to 80 degrees Celsius, which are optimal for activating the catalyst while maintaining safety and efficiency. The resulting compound, as depicted in the structural formula below, features a unique arrangement where the hydrazide group is flanked by sterically hindered piperidine rings. This specific architecture is responsible for the enhanced antioxidant properties, as the secondary amine groups introduced by the hydrazide moiety contribute to a more effective radical scavenging mechanism compared to standard HALS structures.
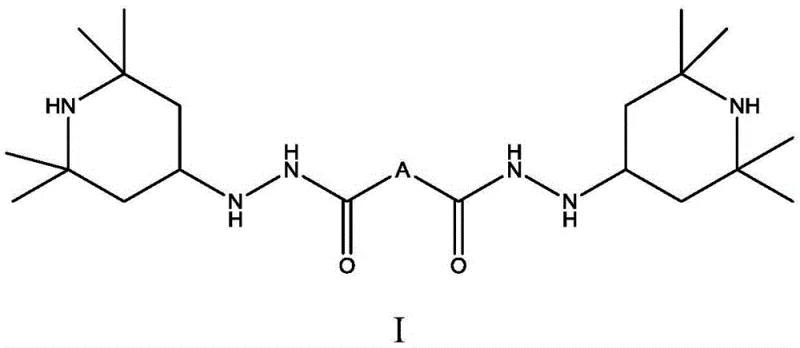
How to Synthesize N,N'-2-(2,2,6,6-Tetramethylpiperidine) Adipic Acid Dihydrazide Efficiently
The synthesis of this high-performance light stabilizer is designed to be scalable and reproducible, making it an attractive candidate for industrial adoption. The process begins with the precise mixing of diacid dihydrazide and triacetoneamine in a solvent system, followed by the addition of a catalytic amount of acid to initiate the condensation. The reaction mixture is then heated to facilitate the azeotropic removal of water, a critical step that ensures high conversion rates. Once the acylhydrazone intermediate is formed, it undergoes a purification step involving cooling and filtration to remove any unreacted starting materials or impurities. The detailed standardized synthesis steps for this specific compound are provided in the guide below, outlining the exact molar ratios, temperature profiles, and workup procedures necessary to achieve the reported purity levels of over 99wt percent.
- Perform dehydration condensation between diacid dihydrazide and triacetoneamine using an acid catalyst in an organic solvent with azeotropic water removal.
- Purify the intermediate acylhydrazone compound via cooling crystallization and filtration to ensure high purity before hydrogenation.
- Conduct catalytic hydrogenation using Pd/C or Raney Nickel under controlled pressure and temperature to reduce the imine group to the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages rooted in process efficiency and material stability. The use of diacid dihydrazide as a starting material provides a more stable and reactive feedstock compared to the amines used in conventional methods, which translates to a more consistent supply of raw materials and reduced variability in production batches. The high yields reported in the patent indicate a significant reduction in waste generation, which directly correlates to lower disposal costs and a smaller environmental footprint for the manufacturing facility. Furthermore, the ability to use common industrial solvents and catalysts means that the supply chain is not dependent on exotic or hard-to-source chemicals, thereby mitigating the risk of supply disruptions. This robustness in the supply chain is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global polymer manufacturers.
- Cost Reduction in Manufacturing: The synthesis route eliminates the need for expensive and specialized catalysts often required in traditional HALS production, replacing them with more cost-effective acid catalysts and standard hydrogenation catalysts like Raney Nickel. The high conversion efficiency of the condensation reaction means that less raw material is wasted, leading to substantial cost savings on input materials. Additionally, the simplified post-processing steps, such as the straightforward filtration and crystallization of the intermediate, reduce the energy and labor costs associated with complex purification procedures. These cumulative efficiencies result in a lower cost of goods sold, allowing for more competitive pricing in the global market for polymer additives without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, such as diacid dihydrazides and triacetoneamine, are commercially available and stable, ensuring a reliable supply chain that is less susceptible to market fluctuations. The process does not rely on single-source or proprietary reagents that could create bottlenecks, thereby enhancing the overall resilience of the manufacturing operation. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or scaled up without significant re-engineering, providing flexibility in production planning. This reliability is a key factor for supply chain heads who need to guarantee uninterrupted delivery of high-purity additives to their customers in the automotive, packaging, and construction industries.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and conditions that are easily adaptable from pilot scale to full commercial production. The use of azeotropic distillation for water removal is a well-established technique that can be efficiently managed in large-scale operations, ensuring consistent product quality across batches. From an environmental perspective, the high selectivity of the reaction reduces the formation of hazardous by-products, simplifying waste treatment and compliance with increasingly stringent environmental regulations. The ability to recover and recycle solvents further enhances the sustainability profile of the process, aligning with the corporate social responsibility goals of modern chemical enterprises and appealing to eco-conscious downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel hindered amine synthesis technology. These answers are derived directly from the technical specifications and performance data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for R&D teams evaluating the feasibility of adoption and for procurement specialists assessing the value proposition of this new additive class.
Q: What distinguishes this hindered amine from conventional HALS like Tinuvin 770?
A: This novel compound incorporates a hydrazide group into the hindered amine structure, providing stronger basicity and superior oxidation resistance compared to traditional ester-based or simple amino-based HALS.
Q: How does the synthesis process ensure high product purity?
A: The process utilizes a specific purification step for the intermediate acylhydrazone involving low-temperature crystallization and washing, which removes impurities before the final hydrogenation step, resulting in purity exceeding 99wt%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize common industrial solvents and catalysts like Raney Nickel or Pd/C, and the high yields reported in the patent indicate a robust process amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hindered Amine Light Stabilizer Supplier
The technical potential of the hydrazide-containing hindered amine described in CN108586321B represents a significant opportunity for enhancing the performance of polymer products globally. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the market. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of light stabilizer meets the highest industry standards. We understand the critical nature of supply chain continuity and are committed to providing a reliable source of high-performance additives that can withstand the rigors of industrial application.
We invite you to explore how this advanced synthesis route can optimize your product formulations and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments that will demonstrate the tangible benefits of partnering with us. By leveraging our expertise in process optimization and scale-up, we can help you secure a competitive edge in the market for high-purity polymer additives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
