Advanced Benzotriazole-Hindered Amine Composite Photostabilizers for Superior Polymer Protection
The escalating demand for durable polymer materials in outdoor applications has driven significant innovation in the field of light stabilizers, particularly regarding the synergy between ultraviolet absorbers (UVA) and hindered amine light stabilizers (HALS). Patent CN103012843B discloses a groundbreaking class of benzotriazole-hindered amine composite photostabilizers that integrate both functional moieties into a single molecular architecture. Unlike traditional physical blends, this covalent bonding strategy ensures that the UV-absorbing benzotriazole segment and the radical-scavenging hindered amine segment work in concert without phase separation or differential migration. This technological leap addresses the critical failure mode of conventional stabilizers, which often suffer from premature depletion due to leaching or hydrolytic instability. By anchoring these protective groups via a robust sulfonamide linkage, the invention provides a reliable polymer additive supplier solution that maintains efficacy even under harsh environmental stressors such as acid rain and intense solar irradiation.
The development of these composite structures represents a paradigm shift in how we approach cost reduction in specialty chemical manufacturing for high-end coatings and textiles. Rather than relying on expensive physical mixtures that require complex dispersion technologies, this single-molecule approach simplifies the formulation process while enhancing performance. The patent details a versatile synthetic platform where substituents R1 can vary from hydrogen to halogens and alkoxy groups, allowing for fine-tuning of solubility and compatibility with diverse polymer matrices. This flexibility is crucial for R&D teams seeking to optimize the commercial scale-up of complex polymer additives without compromising on the purity or spectral characteristics of the final product. The ability to customize the steric and electronic properties of the benzotriazole core while retaining the proven efficacy of the piperidine-based HALS segment makes this technology a cornerstone for next-generation material protection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the field of composite photostabilizers has largely relied on carboxylate ester linkages to connect the UV-absorbing and radical-scavenging fragments. As illustrated in the structural formulas of previous generations, such as Compound IV and Compound V shown below, the ester bond serves as the molecular bridge. While these earlier iterations demonstrated improved light stability compared to single-function additives, they possess a fundamental chemical weakness: susceptibility to hydrolysis. In outdoor applications, materials are frequently exposed to moisture and fluctuating pH levels, including the acidic conditions found in acid rain. Under these circumstances, the ester linkage is prone to cleavage, leading to the dissociation of the stabilizer molecule. This hydrolytic degradation results in the leaching of the active components from the polymer matrix, drastically reducing the service life of the protected material and necessitating frequent re-application or replacement.
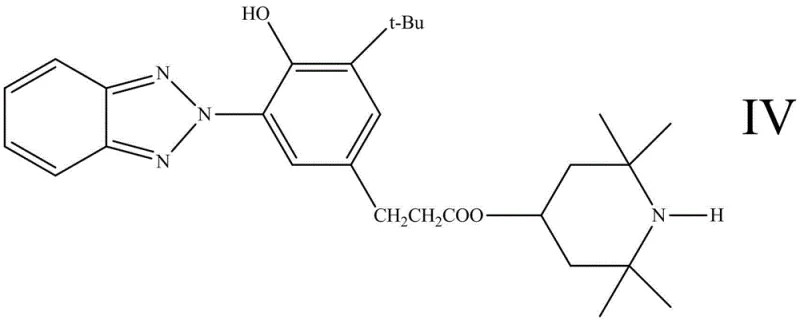
Furthermore, the hydrolysis products generated from ester-based stabilizers can sometimes be acidic themselves, potentially catalyzing further degradation of the host polymer, a phenomenon known as autocatalytic degradation. For procurement managers and supply chain heads, this instability translates into higher warranty claims and reputational risk for downstream manufacturers. The reliance on ester chemistry limits the application scope of these stabilizers to neutral or controlled environments, excluding them from many demanding industrial and agricultural uses. Consequently, there is a pressing need for a chemically inert bridging group that can withstand hydrolytic attack while maintaining the spatial proximity required for the synergistic interaction between the UVA and HALS components.
The Novel Approach
The novel approach presented in patent CN103012843B overcomes these limitations by replacing the labile ester bond with a highly stable sulfonamide or sulfonate ester linkage. As depicted in the general structure below, the sulfonamide group (-SO2-NH-) acts as a robust connector that is inherently resistant to both acidic and alkaline hydrolysis. This structural modification ensures that the composite molecule remains intact throughout the lifecycle of the polymer product, providing consistent protection against photo-oxidative degradation. The synthesis involves converting the hydroxyl group of the benzotriazole intermediate into a sulfonyl chloride, which is then reacted with a hindered amine. This pathway not only enhances chemical stability but also offers greater versatility in molecular design, allowing for the incorporation of various substituted piperidines to tailor the steric hindrance and basicity of the HALS segment.
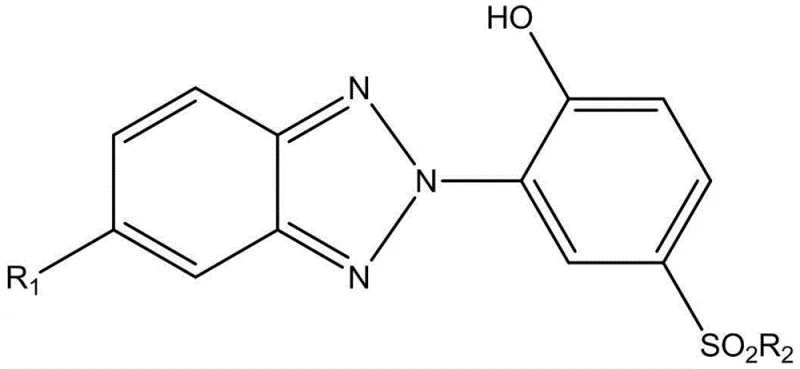
This strategic shift to sulfonamide chemistry significantly improves the durability of the photostabilizer in real-world conditions. Experimental data within the patent confirms that compounds utilizing this new linkage exhibit superior retention of UV absorption capacity after prolonged exposure to accelerated weathering tests compared to their ester-linked counterparts. For the end-user, this means extended product lifespans for textiles, automotive coatings, and agricultural films. From a manufacturing perspective, the new route utilizes readily available starting materials and standard unit operations, facilitating a smoother transition from laboratory scale to industrial production. This innovation effectively resolves the trade-off between stability and functionality, delivering a high-performance additive that meets the rigorous demands of modern material science.
Mechanistic Insights into Sulfonamide-Bridged Cyclization and Stabilization
The synthesis of these advanced photostabilizers relies on a sophisticated multi-step sequence that ensures high regioselectivity and purity. The process begins with the diazotization of an o-nitroaniline derivative, followed by coupling with p-hydroxybenzenesulfonic acid to form an azo dye intermediate. This intermediate is then subjected to a reductive ring-closure reaction using thiourea dioxide and sodium hydroxide. This specific reduction step is critical as it simultaneously reduces the nitro group and cyclizes the azo linkage to form the benzotriazole ring system, which is the core UV-absorbing chromophore. The use of thiourea dioxide as a reducing agent is particularly advantageous as it offers a cleaner reaction profile compared to traditional metal-based reducers, minimizing heavy metal contamination in the final product. Following cyclization, the phenolic hydroxyl group is activated via reaction with thionyl chloride to generate a reactive sulfonyl chloride intermediate, setting the stage for the final conjugation.
The final step involves the nucleophilic substitution of the sulfonyl chloride with a hindered amine, such as 1,2,2,6,6-pentamethylpiperidin-4-ol or 4-amino-2,2,6,6-tetramethylpiperidine. This reaction forms the stable sulfonamide or sulfonate bond that defines the invention. The resulting molecule, exemplified by Compound II shown below, possesses a dual-action mechanism. The benzotriazole moiety absorbs harmful UV radiation and dissipates it as harmless thermal energy through a rapid keto-enol tautomerism cycle. Simultaneously, the hindered amine segment intercepts free radicals generated by any UV radiation that penetrates the initial defense, breaking the chain reaction of photo-oxidation. This synergistic effect is far more efficient than the sum of the individual parts, providing comprehensive protection for the polymer substrate.
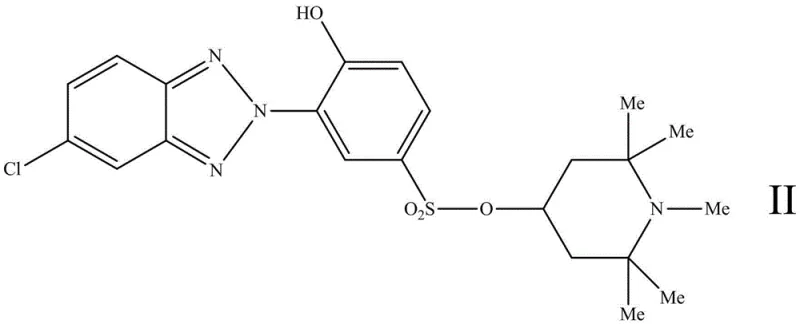
From an impurity control perspective, the synthetic route is designed to minimize side reactions. The use of specific molar ratios, such as 1:25~30 for the benzotriazole intermediate to thionyl chloride, ensures complete conversion to the acid chloride, preventing the formation of unreacted hydroxyl species that could compromise stability. Furthermore, the purification steps involving acid washing and recrystallization effectively remove inorganic salts and unreacted amines, ensuring a high-purity product suitable for sensitive applications like food packaging or medical devices. The robust nature of the sulfonamide bond also means that the final product does not degrade during the high-temperature processing of polymers, a common failure point for less stable additives. This mechanistic robustness is key to achieving the consistent quality required by global supply chains.
How to Synthesize Benzotriazole-Hindered Amine Composite Efficiently
The preparation of these high-value photostabilizers follows a logical progression of organic transformations that are well-suited for batch processing in standard chemical reactors. The protocol outlined in the patent emphasizes precise temperature control and stoichiometry to maximize yield and minimize byproduct formation. The initial diazo-coupling must be maintained at low temperatures (0-5°C) to prevent diazonium salt decomposition, while the subsequent reductive cyclization requires elevated temperatures (70-90°C) to drive the ring closure to completion. The final coupling with the hindered amine is conducted under mild conditions to preserve the integrity of the sensitive piperidine ring. Detailed standardized synthesis steps are provided in the guide below to assist process engineers in replicating this high-efficiency route.
- Perform diazo-coupling reaction between o-nitroaniline derivatives and p-hydroxybenzenesulfonic acid at 0-5°C to form the azo dye intermediate.
- Execute reductive ring-closure using thiourea dioxide and sodium hydroxide at 70-90°C to cyclize the intermediate into the benzotriazole UV absorber core.
- Convert the hydroxyl group to a sulfonyl chloride using thionyl chloride, followed by reaction with hindered amine light stabilizers (HALS) to form the final composite.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain managers, the adoption of this sulfonamide-bridged technology offers tangible strategic benefits beyond mere performance metrics. The primary advantage lies in the enhanced durability of the additive, which directly correlates to reduced consumption rates and lower total cost of ownership for the end customer. Because the stabilizer does not hydrolyze or leach out as rapidly as ester-based alternatives, manufacturers can achieve the same level of protection with lower loading levels or extend the warranty periods of their finished goods. This value proposition allows suppliers to command a premium position in the market while delivering actual cost savings to their clients through improved efficiency. Additionally, the synthetic route avoids the use of expensive transition metal catalysts, relying instead on commodity reagents like thionyl chloride and thiourea dioxide, which stabilizes raw material costs and mitigates supply volatility.
- Cost Reduction in Manufacturing: The elimination of unstable ester linkages removes the need for costly stabilization packages that are often required to protect the additive itself during storage and processing. By utilizing a chemically robust sulfonamide backbone, the manufacturing process becomes more forgiving and less prone to batch-to-batch variability caused by hydrolysis during production. This inherent stability simplifies quality control protocols and reduces the rejection rate of off-spec material. Furthermore, the avoidance of heavy metal catalysts in the reduction step eliminates the need for expensive and time-consuming metal scavenging processes, streamlining the downstream purification workflow and significantly lowering operational expenditures associated with waste treatment and catalyst recovery.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including o-nitroaniline derivatives and p-hydroxybenzenesulfonic acid, are widely available commodity chemicals with established global supply chains. This reduces the risk of supply disruptions that are often associated with specialized or proprietary intermediates. The synthetic steps involve standard unit operations such as filtration, distillation, and crystallization, which can be easily scaled up in existing multipurpose chemical plants without the need for specialized equipment. This manufacturability ensures that production can be ramped up quickly to meet surges in demand, providing a reliable source of high-purity polymer additives even during periods of market tightness. The robustness of the final product also simplifies logistics, as it does not require stringent moisture control during shipping and storage.
- Scalability and Environmental Compliance: The process is designed with environmental sustainability in mind, utilizing aqueous workups and avoiding chlorinated solvents where possible. The use of thiourea dioxide generates benign byproducts that are easier to treat in standard wastewater facilities compared to heavy metal sludge. This alignment with green chemistry principles facilitates regulatory compliance in regions with strict environmental standards, such as the EU and North America. The scalability of the route is proven by the use of common reagents and moderate reaction conditions, making it feasible to transition from kilogram-scale laboratory batches to multi-tonne commercial production with minimal process re-engineering. This ease of scale-up ensures a continuous supply of material to support long-term contracts and large-volume applications in the automotive and construction sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel photostabilizer technology. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the fit of this additive within your specific formulation requirements and supply chain strategy.
Q: Why is the sulfonamide bridge superior to ester bridges in photostabilizers?
A: Sulfonamide bridges offer significantly higher resistance to hydrolysis under acidic or alkaline conditions compared to carboxylate ester bridges, ensuring long-term durability in outdoor environments exposed to acid rain.
Q: What are the primary applications for these composite photostabilizers?
A: These stabilizers are designed for high-performance polymer materials, textiles, and coatings where simultaneous UV absorption and free radical scavenging are required to prevent photo-oxidative degradation.
Q: Does the introduction of the hindered amine segment affect UV absorption?
A: No, experimental data indicates that introducing the hindered amine fragment has negligible impact on the UV absorption wavelength and molar extinction coefficient of the benzotriazole core, maintaining high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzotriazole-Hindered Amine Photostabilizer Supplier
As the global demand for high-performance polymer additives continues to rise, partnering with a technically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in heterocyclic chemistry to deliver advanced solutions like the sulfonamide-bridged photostabilizers described in CN103012843B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the exacting standards required for critical applications in automotive, textile, and coating industries.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific formulation needs, demonstrating the economic benefits of switching to our hydrolytically stable additives. Contact us today to request specific COA data and route feasibility assessments, and let us help you build a more resilient and efficient supply chain for your light stabilization requirements.
