Advanced Cyclophosphazene Flame Retardant Synthesis for Scalable Polymer Additive Manufacturing
Advanced Cyclophosphazene Flame Retardant Synthesis for Scalable Polymer Additive Manufacturing
The development of high-performance flame retardant materials has become a critical priority for the global polymer industry, driven by increasingly stringent safety regulations and environmental standards. Patent CN100484946C introduces a groundbreaking approach to synthesizing cyclophosphazene compounds that function as reactive flame retardants, addressing the longstanding limitations of traditional additive-type agents. This technology leverages the inherent thermal stability of the phosphorus-nitrogen inorganic backbone while introducing polymerizable organic side chains, ensuring that the flame retardant properties are chemically bonded to the matrix rather than physically blended. For R&D directors and procurement specialists seeking a reliable flame retardant supplier, this patent represents a significant leap forward in material science, offering a pathway to produce additives that do not migrate, volatilize, or leach out of the final polymer product over time.
Unlike conventional phosphorus-nitrogen compounds that often suffer from compatibility issues and poor dispersion within polymer matrices, the cyclophosphazene derivatives described in this intellectual property are designed to participate directly in the curing or polymerization process. The molecular architecture features a cyclic phosphazene core substituted with aromatic groups containing unsaturated bonds, such as acrylate or methacrylate functionalities. This structural design not only enhances the char-forming capability during combustion but also improves the mechanical integrity of the cured resin. By shifting from additive to reactive mechanisms, manufacturers can achieve permanent flame retardancy with lower loading levels, thereby preserving the physical properties of the base polymer while meeting rigorous fire safety codes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of reactive phosphazene-based flame retardants has been plagued by severe technical challenges that hindered their widespread commercial adoption. Prior art, such as the methods disclosed in Japanese patent JP 70-95426, relied on reaction conditions that were excessively harsh, often requiring extreme temperatures or pressures that complicated process control and increased energy consumption. Furthermore, the resulting compounds frequently exhibited unstable performance characteristics, with inconsistent curing times and limited versatility in terms of applicable curing mechanisms. These deficiencies created significant bottlenecks for supply chain heads, as the unpredictability of the synthesis led to batch-to-batch variability and potential delays in production schedules. Additionally, the reliance on complex purification steps to remove toxic byproducts or unreacted chlorines added substantial cost and environmental burden to the manufacturing process.
The Novel Approach
The innovative methodology presented in CN100484946C overcomes these historical barriers by redesigning the molecular substitution pattern to exploit the enhanced reactivity of benzyl positions. Instead of direct substitution on the phosphorus atom which can be sterically hindered or electronically deactivated, the process utilizes intermediates where the reactive sites are located on the benzyl carbon atoms, either as halogens or hydroxyl groups. This strategic modification allows the subsequent esterification or etherification reactions to proceed under much milder conditions, typically below 100°C, significantly reducing thermal stress on the sensitive phosphazene ring. 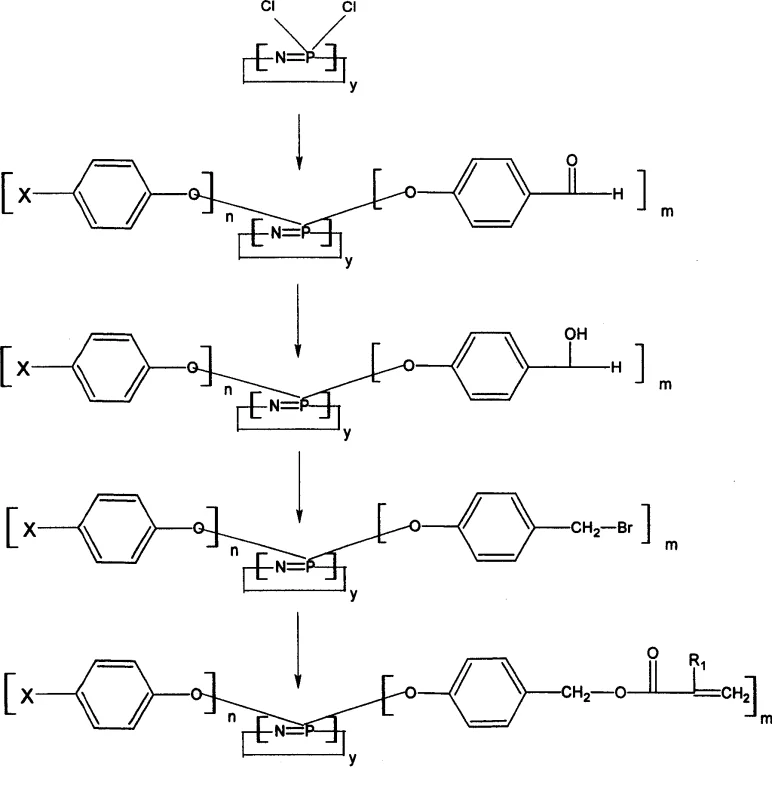 As illustrated in the reaction scheme, the pathway involves a logical sequence of functional group transformations that are robust and scalable, enabling the production of high-purity cyclophosphazene derivatives with consistent quality. This novel approach not only simplifies the operational complexity but also drastically shortens the curing time of the final resin, making it highly attractive for high-throughput industrial applications.
As illustrated in the reaction scheme, the pathway involves a logical sequence of functional group transformations that are robust and scalable, enabling the production of high-purity cyclophosphazene derivatives with consistent quality. This novel approach not only simplifies the operational complexity but also drastically shortens the curing time of the final resin, making it highly attractive for high-throughput industrial applications.
Mechanistic Insights into Nucleophilic Substitution and Esterification
The core chemical transformation driving this synthesis is a series of nucleophilic substitutions centered around the hexachlorocyclotriphosphazene starting material. The initial step involves the displacement of chlorine atoms on the phosphorus ring by the phenoxide anion derived from p-hydroxybenzaldehyde, facilitated by bases such as potassium carbonate or sodium hydride in tetrahydrofuran solvent. This reaction establishes the stable P-O-C linkage that anchors the organic functionality to the inorganic core, creating a hybrid structure that combines the thermal resistance of the P-N ring with the reactivity of the aromatic aldehyde. The subsequent reduction of the aldehyde group to a hydroxymethyl group using sodium borohydride is a critical mechanistic step that prepares the molecule for further functionalization without affecting the integrity of the phosphazene cycle or the aromatic rings.
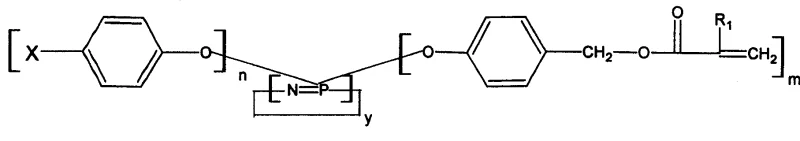 The final stage of the mechanism involves converting the benzylic hydroxyl group into a leaving group, typically a bromide, or directly esterifying it with acryloyl chloride to introduce the polymerizable vinyl group. This step is crucial for imparting the reactive nature of the flame retardant, allowing it to copolymerize with resins like epoxy or unsaturated polyester. From an impurity control perspective, the use of phase transfer catalysts and specific solvent systems ensures that side reactions, such as homopolymerization of the acrylate groups, are minimized. The rigorous washing and recrystallization protocols described in the patent effectively remove inorganic salts like potassium bromide or chloride, ensuring that the final product meets the stringent purity specifications required for electronic or aerospace grade materials.
The final stage of the mechanism involves converting the benzylic hydroxyl group into a leaving group, typically a bromide, or directly esterifying it with acryloyl chloride to introduce the polymerizable vinyl group. This step is crucial for imparting the reactive nature of the flame retardant, allowing it to copolymerize with resins like epoxy or unsaturated polyester. From an impurity control perspective, the use of phase transfer catalysts and specific solvent systems ensures that side reactions, such as homopolymerization of the acrylate groups, are minimized. The rigorous washing and recrystallization protocols described in the patent effectively remove inorganic salts like potassium bromide or chloride, ensuring that the final product meets the stringent purity specifications required for electronic or aerospace grade materials.
How to Synthesize Cyclophosphazene Flame Retardant Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing these advanced materials, emphasizing reproducibility and ease of scale-up. The process begins with the preparation of the aldehyde-functionalized intermediate, followed by reduction and final functionalization, with each step optimized for maximum yield and minimal waste generation. Detailed standard operating procedures regarding molar ratios, solvent volumes, and temperature profiles are essential for maintaining product consistency, particularly when transitioning from laboratory bench scale to pilot plant operations.
- Perform nucleophilic substitution of hexachlorocyclotriphosphazene with p-hydroxybenzaldehyde in THF using K2CO3 or NaH as base at reflux temperatures.
- Reduce the resulting aldehyde-functionalized cyclophosphazene to the corresponding hydroxymethyl derivative using sodium borohydride in a methanol/THF mixture.
- Convert the hydroxyl group to a bromide using HBr/H2SO4, followed by reaction with potassium methacrylate or acryloyl chloride to introduce the polymerizable unsaturated bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple material performance. The elimination of harsh reaction conditions translates directly into reduced energy costs and lower capital expenditure on specialized high-pressure or high-temperature reactor equipment. Furthermore, the use of common, commercially available reagents such as tetrahydrofuran, sodium borohydride, and potassium methacrylate ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or controlled precursors. This accessibility allows for cost reduction in polymer additive manufacturing by streamlining the procurement process and reducing lead times for raw material acquisition.
- Cost Reduction in Manufacturing: The simplified purification workflow, which relies on standard recrystallization and aqueous washing rather than expensive chromatographic separation, significantly lowers the operational expenditure per kilogram of product. By avoiding the use of transition metal catalysts that require complex removal steps to meet regulatory limits, the process inherently reduces downstream processing costs and waste treatment liabilities. The high yields reported in the examples indicate efficient atom economy, meaning less raw material is wasted, which directly contributes to substantial cost savings over large production volumes.
- Enhanced Supply Chain Reliability: The robustness of the chemical steps, which tolerate minor variations in reaction parameters without compromising product quality, ensures consistent output even in large-scale manufacturing environments. This reliability is critical for maintaining continuous production schedules for downstream customers who depend on just-in-time delivery of flame retardant additives. The ability to synthesize the material using standard glass-lined or stainless steel reactors further enhances supply security, as it does not require specialized metallurgy that might be subject to long lead times or maintenance downtime.
- Scalability and Environmental Compliance: The process operates at temperatures generally below 100°C for most steps, reducing the thermal load on cooling systems and minimizing the risk of thermal runaway incidents. The solvents used, such as THF and acetone, are widely recycled in the chemical industry, facilitating closed-loop systems that align with green chemistry principles and reduce volatile organic compound (VOC) emissions. This environmental compatibility simplifies regulatory compliance and permitting processes, accelerating the time-to-market for new flame retardant formulations based on this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclophosphazene synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making.
Q: What is the thermal stability of the synthesized cyclophosphazene resin?
A: According to TGA analysis data provided in the patent, the cured resin exhibits exceptional thermal stability, showing only 20% weight loss at 390°C and retaining 48% of its mass even at 750°C, confirming its suitability for high-performance flame retardant applications.
Q: How does the benzyl modification improve reactivity compared to traditional methods?
A: The synthetic strategy utilizes the high reactivity of the benzyl position (either via halogen or hydroxyl groups) to facilitate substitution reactions under milder conditions, overcoming the harsh synthesis requirements and instability issues associated with earlier phosphazene derivatives.
Q: What purification methods are employed to ensure high product purity?
A: The process employs straightforward purification techniques including aqueous washing, solvent extraction, and recrystallization using solvents like ethyl acetate, ethanol, or acetone/petroleum ether mixtures, effectively removing inorganic salts and byproducts without complex chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclophosphazene Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cyclophosphazene flame retardant technology disclosed in CN100484946C and are fully equipped to bring this innovation to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are designed to handle the specific solvent systems and reaction conditions required for phosphazene chemistry, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards for polymer additives.
We invite you to collaborate with us to optimize this synthesis route for your specific application requirements, whether for epoxy curing agents, coating resins, or high-performance composites. Our technical team is ready to provide a Customized Cost-Saving Analysis that evaluates the economic feasibility of adopting this reactive flame retardant in your current formulation. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable competitive advantage through advanced material science.
