Advanced Aqueous Synthesis of 1,3-Diol Monoethers for High-Performance Polymer Catalysts
Advanced Aqueous Synthesis of 1,3-Diol Monoethers for High-Performance Polymer Catalysts
The chemical industry is constantly seeking more sustainable and economically viable pathways for synthesizing critical intermediates, particularly those used in high-value applications like olefin polymerization. Patent CN1978416B introduces a groundbreaking methodology for the preparation of 1,3-diol monoether compounds, specifically addressing the structural formula (I) shown below. This innovation represents a significant departure from traditional hazardous synthetic routes by utilizing a benign aqueous or biphasic system. By shifting away from strictly anhydrous organic solvents and reactive metals, this technology offers a robust platform for producing electron donor components essential for Ziegler-Natta catalysts. The ability to synthesize these compounds safely and economically is paramount for manufacturers aiming to optimize their supply chains for polymer additives.
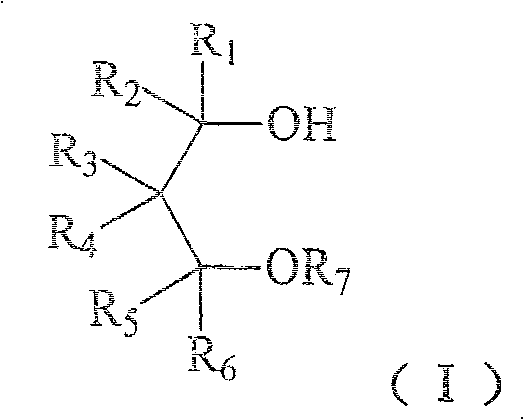
For R&D directors and process engineers, the implications of this patent extend beyond simple cost savings; it fundamentally alters the risk profile of ether synthesis. The traditional reliance on moisture-sensitive reagents often leads to batch inconsistencies and safety incidents. In contrast, the method described in CN1978416B leverages the stability of sulfate esters in alkaline water phases. This approach not only simplifies the operational protocol by removing the need for rigorous drying of starting materials but also enhances the reproducibility of the reaction. Consequently, this technology serves as a reliable foundation for the commercial scale-up of complex polymer additives, ensuring consistent quality for downstream polymerization processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-diol monoethers has been dominated by the Williamson ether synthesis, a method fraught with significant operational and safety challenges. This conventional approach typically necessitates the use of strong bases such as metallic sodium or sodium hydride in strictly anhydrous organic solvents like tetrahydrofuran or tert-butanol. The reaction between alcohols and these reactive metals is notoriously violent, generating flammable hydrogen gas which poses severe explosion hazards in large-scale manufacturing environments. Furthermore, the requirement for absolutely dry conditions mandates energy-intensive drying procedures for all reactants and solvents, adding substantial complexity and cost to the production workflow. Additionally, the alkylating agents traditionally employed, such as alkyl iodides or bromides, are often prohibitively expensive and contribute to a heavier environmental burden due to halogenated waste streams.
The Novel Approach
The methodology disclosed in CN1978416B offers a transformative solution by replacing hazardous reagents with safer, more economical alternatives within an aqueous framework. Instead of reactive metals, the process utilizes common inorganic bases like sodium hydroxide or potassium carbonate in water or water-oil biphasic systems. This shift eliminates the generation of explosive hydrogen gas and removes the stringent requirement for anhydrous conditions, thereby drastically simplifying the operational procedure. Crucially, the invention substitutes costly alkyl halides with sulfate esters, such as dimethyl sulfate or diethyl sulfate, which are significantly more affordable and readily available on the global market. The result is a streamlined process where the product naturally separates into an oil phase post-reaction, facilitating easier isolation and purification compared to the complex workups required by traditional methods.
Mechanistic Insights into Aqueous Phase Nucleophilic Substitution
From a mechanistic perspective, this synthesis relies on a nucleophilic substitution reaction where the hydroxyl group of the 1,3-diol acts as the nucleophile attacking the sulfur-bound alkyl group of the sulfate ester. The reaction is facilitated by the alkaline environment, which deprotonates the alcohol to form a more reactive alkoxide species in situ. In biphasic systems, the addition of phase transfer catalysts, such as quaternary ammonium salts, plays a critical role in shuttling the ionic alkoxide species into the organic phase where the reaction with the sulfate ester occurs efficiently. This interfacial catalysis ensures high reaction rates even at moderate temperatures, typically ranging from 20°C to 100°C, preventing the thermal decomposition of the sulfate ester which can occur at higher temperatures. The careful control of pH between 8 and 12 is essential to maintain the balance between alkoxide formation and the stability of the reactants.
Regioselectivity is another critical aspect managed by this mechanism, ensuring the formation of the mono-ether rather than the di-ether. By precisely controlling the molar ratio of the 1,3-diol to the sulfate ester, typically favoring an excess of the diol, the probability of a second alkylation event is statistically minimized. The steric hindrance provided by the substituents on the 1,3-diol backbone, such as the gem-dimethyl groups in 2,2-dimethyl-1,3-propanediol, further aids in directing the substitution to the desired position. This intrinsic selectivity reduces the formation of by-products, leading to a cleaner crude reaction mixture. For procurement managers, this high selectivity translates directly into reduced purification costs and higher overall yields, making the process highly attractive for cost reduction in polymer additive manufacturing.
How to Synthesize 1,3-Diol Monoether Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of mixing, heating, and separation steps that are easily adaptable to existing reactor infrastructure. The process begins with the dissolution of the 1,3-diol substrate in an aqueous medium, potentially supplemented with a co-solvent like toluene or petroleum ether to create a biphasic system. An inorganic base is then introduced to adjust the pH, followed by the controlled addition of the sulfate ester alkylating agent. The reaction mixture is maintained at an elevated temperature, generally between 50°C and 90°C, for a duration sufficient to drive the conversion to completion, typically ranging from 1 to 8 hours depending on the specific substrates involved.
- Dissolve the 1,3-diol starting material in water or a water-organic solvent mixture under alkaline conditions.
- Add the sulfate ester alkylating agent slowly while maintaining the pH between 8 and 12, optionally using a phase transfer catalyst.
- Separate the oil phase containing the product from the aqueous layer, followed by distillation to isolate the pure 1,3-diol monoether.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this aqueous synthesis technology presents a compelling value proposition centered on cost efficiency and risk mitigation. The elimination of expensive alkyl halides in favor of sulfate esters results in a direct reduction in raw material expenditure, which is a primary driver of the final product price. Moreover, the removal of the drying step for solvents and reactants significantly lowers energy consumption and shortens the overall cycle time per batch. This efficiency gain allows for higher throughput without the need for capital investment in new equipment, effectively increasing the capacity of existing facilities. The simplified workup procedure, where the product separates as an oil layer, reduces the demand for complex distillation columns and extensive solvent recovery systems, further lowering operational expenditures.
- Cost Reduction in Manufacturing: The substitution of high-cost alkyl halides with economical sulfate esters creates a substantial margin improvement for manufacturers. By avoiding the use of precious metal catalysts or reactive metals like sodium hydride, the process eliminates the need for specialized handling equipment and quenching protocols associated with hazardous materials. The reduction in solvent usage and the ability to use water as the primary reaction medium significantly decrease the volume of organic waste generated, leading to lower disposal fees. These cumulative savings allow suppliers to offer more competitive pricing for high-purity 1,3-diol monoethers while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide and dimethyl sulfate ensures a stable and resilient supply chain, as these materials are produced globally in massive quantities. Unlike specialized reagents that may suffer from supply disruptions, the inputs for this process are readily available from multiple vendors, reducing the risk of production stoppages. The robustness of the aqueous system also means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent output even when sourcing from different suppliers. This reliability is crucial for maintaining continuous production schedules for critical polymer catalyst components.
- Scalability and Environmental Compliance: The inherent safety of the aqueous process facilitates easier regulatory approval and scaling from pilot plants to multi-ton commercial production. The absence of flammable hydrogen gas generation removes a major safety barrier often encountered during scale-up, allowing for larger batch sizes without proportional increases in safety infrastructure. Furthermore, the reduced generation of halogenated waste aligns with increasingly stringent environmental regulations, minimizing the ecological footprint of the manufacturing process. This compliance advantage future-proofs the supply chain against tightening environmental laws, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-diol monoethers using this advanced methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains or product portfolios.
Q: Why is the aqueous sulfate ester method safer than traditional Williamson ether synthesis?
A: Traditional methods often utilize reactive metals like sodium or sodium hydride which generate flammable hydrogen gas and require strictly anhydrous conditions. The patented aqueous method eliminates pyrophoric reagents and explosive gas generation, significantly enhancing operational safety.
Q: How does this process impact the cost of producing polymer catalyst donors?
A: By replacing expensive alkyl halides with cost-effective sulfate esters and eliminating the need for energy-intensive solvent drying and recovery processes, the overall manufacturing cost is substantially reduced.
Q: Can this method achieve high regioselectivity for mono-etherification?
A: Yes, by carefully controlling the molar ratio of the diol to the sulfate ester and utilizing specific alkaline conditions, the process effectively suppresses di-ether formation, yielding high-purity mono-ether products suitable for sensitive catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Diol Monoether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of downstream polymerization catalysts. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global polymer industry. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our capability to implement the safe and efficient aqueous synthesis methods described in recent patents allows us to provide a stable supply of essential catalyst components.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can benefit your specific applications. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-performance intermediates into your production workflow.
