Advanced Catalytic Reduction for High-Purity 3,3'-Diaminodiphenyl Sulfone Production
The chemical industry is constantly evolving towards greener, more efficient synthetic pathways, particularly for high-value intermediates like 3,3'-diaminodiphenyl sulfone (DDS). A pivotal advancement in this domain is documented in Chinese Patent CN101654422A, which details a novel preparation method utilizing hydrazine hydrate reduction catalyzed by iron oxide hydroxide. This technology represents a significant departure from traditional methods, addressing critical pain points regarding environmental compliance and process economics. For R&D directors and procurement strategists, understanding this shift is vital, as it offers a route to high-purity products with substantially reduced downstream processing requirements. The patent outlines a robust protocol where 3,3'-dinitrodiphenyl sulfone serves as the starting material, undergoing reduction in a saturated low-grade fatty alcohol solvent system. This approach not only simplifies the operational workflow but also ensures that the final product meets stringent quality specifications necessary for demanding applications in polysulfonamide fibers and epoxy curing systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines like 3,3'-DDS has relied heavily on two primary methodologies: iron powder reduction and catalytic hydrogenation. While the iron powder reduction method is technically mature and easy to control, it suffers from severe environmental and efficiency drawbacks. The process generates massive quantities of iron mud and amine-containing wastewater, creating a substantial burden for waste treatment facilities and increasing the overall cost of production due to complex post-treatment steps. Furthermore, the isolation of water-soluble low arylamines is notoriously difficult, often leading to reduced yields and compromised product purity. On the other hand, catalytic hydrogenation, while offering high conversion rates and fewer side reactions, requires significant capital investment in high-pressure equipment and precious metal catalysts like platinum or palladium. This makes it less economically viable for small-batch production or facilities lacking specialized high-pressure infrastructure, thereby limiting supply chain flexibility and increasing the barrier to entry for manufacturers.
The Novel Approach
The methodology presented in CN101654422A offers a compelling alternative by leveraging hydrazine hydrate as a hydrogen donor in conjunction with an inexpensive iron oxide hydroxide catalyst. This novel approach effectively bypasses the heavy metal sludge issues of iron powder reduction and the high capital expenditure of catalytic hydrogenation. By operating under mild reaction conditions, specifically within a temperature range of 60-70°C, the process minimizes energy consumption and reduces the risk of thermal runaway or safety incidents associated with high-pressure hydrogen gas. The use of saturated lower aliphatic alcohols as solvents further enhances the sustainability profile, as these solvents are readily available and can be efficiently recovered. This combination of factors results in a streamlined process that is not only environmentally friendlier with fewer "three wastes" but also highly adaptable for varying production scales, from pilot batches to commercial manufacturing, ensuring a reliable supply of high-purity intermediates.
Mechanistic Insights into Iron Oxide Hydroxide-Catalyzed Hydrazine Reduction
The core of this technological breakthrough lies in the specific interaction between the iron oxide hydroxide catalyst and the hydrazine hydrate reducing agent. Unlike traditional heterogeneous catalysis that might require extreme pressures, this system operates through a surface-mediated transfer hydrogenation mechanism. The iron oxide hydroxide, prepared by adjusting ferric chloride to a specific pH and aging the precipitate, possesses a high specific surface area ranging from 50 to 500 m²/g. This extensive surface area provides abundant active sites for the adsorption and activation of hydrazine molecules. Upon heating, the hydrazine decomposes on the catalyst surface to release active hydrogen species, which are then transferred to the nitro groups of the 3,3'-dinitrodiphenyl sulfone substrate. This stepwise reduction converts the nitro groups first to nitroso and hydroxylamine intermediates before finally yielding the primary amine groups. The mild alkalinity maintained during catalyst preparation ensures the stability of the active iron species, preventing premature deactivation or aggregation that could hinder reaction kinetics.
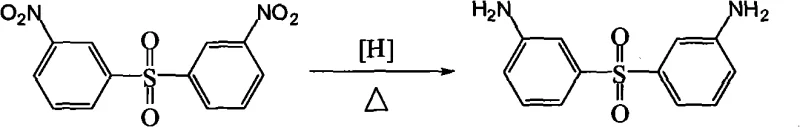
Furthermore, the impurity control mechanism inherent in this process is superior to conventional methods. In iron powder reductions, the formation of azo and azoxy byproducts is common due to condensation reactions between intermediate species. However, the controlled release of active hydrogen in the hydrazine system, moderated by the iron oxide hydroxide catalyst, favors the direct reduction pathway to the amine. The use of alcohol solvents also plays a crucial role in solubilizing the organic substrate while maintaining the catalyst in a suspended state, ensuring uniform contact and minimizing localized hot spots that could lead to degradation. Post-reaction, the catalyst can be separated via simple filtration, and the product is isolated through crystallization from the mother liquor after solvent evaporation. This simplicity in work-up significantly reduces the potential for introducing contaminants during purification, thereby consistently achieving HPLC purity assays of 99% as demonstrated in the patent examples.
How to Synthesize 3,3'-Diaminodiphenyl Sulfone Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the stoichiometry of the reagents to maximize yield and purity. The process begins with the in-situ or ex-situ generation of the iron oxide hydroxide catalyst, followed by the suspension of the dinitro precursor in the alcoholic solvent. The reaction is initiated by the controlled addition of hydrazine hydrate under reflux conditions. Detailed standard operating procedures regarding exact mixing times, addition rates, and crystallization parameters are critical for reproducibility.
- Preparation of Iron Oxide Hydroxide Catalyst: Adjust ferric chloride solution to pH 6-10 with base, heat to 60-70°C, age, filter, and dry to obtain the catalyst.
- Reduction Reaction: Suspend 3,3'-dinitrodiphenyl sulfone and the catalyst in saturated lower aliphatic alcohol, heat to 60-70°C, and add hydrazine hydrate.
- Isolation and Purification: Reflux, filter hot, decolorize with activated carbon, evaporate solvent, and crystallize the product from the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrazine-based reduction technology translates into tangible strategic advantages beyond mere technical feasibility. The shift away from precious metal catalysts and high-pressure hydrogenation equipment drastically lowers the barrier for manufacturing, allowing for a more diversified and resilient supplier base. This decentralization of production capability reduces the risk of supply disruptions caused by bottlenecks at specialized high-pressure facilities. Moreover, the simplified waste profile means that manufacturers face lower regulatory compliance costs and reduced liabilities associated with hazardous waste disposal. The ability to operate at atmospheric pressure and moderate temperatures also enhances plant safety, potentially lowering insurance premiums and operational downtime related to safety audits. These factors collectively contribute to a more stable and cost-effective supply chain for critical polymer monomers and epoxy curing agents.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts such as platinum or palladium, which are subject to volatile market pricing, leads to significant raw material cost savings. Additionally, the avoidance of high-pressure reactor vessels reduces capital depreciation costs and maintenance expenses associated with complex hydrogenation infrastructure. The recyclability of the iron oxide hydroxide catalyst further extends the economic benefits, as the catalyst load can be minimized over multiple batches without compromising activity. This reduction in both fixed and variable costs allows for more competitive pricing structures in the final polymer additive market.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including ferric chloride, hydrazine hydrate, and lower aliphatic alcohols, are commodity chemicals with robust global supply chains. This contrasts sharply with the reliance on specialized catalysts or high-purity hydrogen gas, which can be subject to logistical constraints. The mild reaction conditions also mean that production can be scaled up or down rapidly in response to market demand without requiring extensive requalification of equipment. This flexibility ensures that buyers can secure consistent volumes of high-purity 3,3'-DDS even during periods of market volatility, safeguarding their own production schedules for downstream polymer applications.
- Scalability and Environmental Compliance: The process generates significantly less solid waste (iron mud) and wastewater compared to traditional iron powder reduction, simplifying the environmental permitting process for manufacturing sites. The reduced volume of hazardous waste lowers the cost and complexity of effluent treatment, making the process more sustainable and aligned with modern green chemistry principles. Furthermore, the simplicity of the unit operations—mixing, heating, filtering, and crystallizing—facilitates easy scale-up from laboratory to commercial production. This scalability ensures that the technology can meet growing global demand for polysulfonamide fibers and advanced epoxy systems without encountering the engineering challenges typical of high-pressure chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,3'-diaminodiphenyl sulfone using this advanced reduction method. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this material into your specific application workflows.
Q: What are the primary advantages of the hydrazine reduction method over traditional iron powder reduction?
A: The hydrazine reduction method described in CN101654422A eliminates the generation of large quantities of iron mud and amine-containing wastewater associated with iron powder reduction, significantly simplifying post-treatment and reducing environmental burden.
Q: Can the iron oxide hydroxide catalyst be recycled in this process?
A: Yes, the patent highlights that the iron oxide hydroxide catalyst is cheap, easily obtained, and capable of repeated recycling, which contributes to lower operational costs and reduced waste generation.
Q: What purity levels can be achieved with this synthesis route?
A: According to the experimental data in the patent, the process yields 3,3'-diaminodiphenyl sulfone with an HPLC purity assay reaching up to 99% and reaction yields up to 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diaminodiphenyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of advanced polymers and specialty chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 3,3'-diaminodiphenyl sulfone that meets stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify every batch. Our capability to implement the efficient hydrazine reduction technology described in CN101654422A allows us to offer a product that balances exceptional quality with commercial viability, catering to the exacting needs of the global polymer and electronics industries.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals. Let us be your partner in driving innovation and efficiency in your chemical manufacturing processes.
