Revolutionizing Acifluorfen Production: High-Efficiency Catalytic Synthesis for Global Supply Chains
Revolutionizing Acifluorfen Production: High-Efficiency Catalytic Synthesis for Global Supply Chains
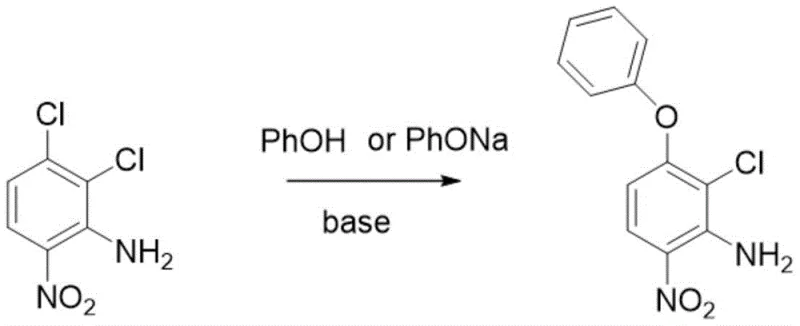
The global demand for high-performance herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, with acifluorfen standing out as a critical component in modern weed management strategies. Recent advancements documented in patent CN115108922A introduce a transformative one-step synthesis method utilizing 2,3-dichloro-6-nitroaniline as the primary raw material, marking a significant departure from traditional multi-stage processes. This technological breakthrough leverages the unique properties of quaternary ammonium salt catalysts to enhance reaction kinetics, ensuring that the conversion of starting materials proceeds with exceptional speed and selectivity. For R&D directors and technical leaders, this represents a pivotal opportunity to reevaluate existing manufacturing protocols, as the new method promises to resolve long-standing issues related to reaction completeness and impurity profiles. By integrating this advanced catalytic system, manufacturers can achieve a level of operational efficiency that was previously unattainable, setting a new benchmark for the production of high-purity agrochemical intermediates in a competitive market landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acifluorfen has been plagued by significant inefficiencies that hinder large-scale commercial viability and economic performance. Prior art methods, such as those described in CN103242178, rely heavily on dimethyl sulfoxide as a solvent and require extended reaction times of up to 20 hours at elevated temperatures, which drastically increases energy consumption and operational costs. Furthermore, the use of large quantities of carbonate systems in these traditional routes often leads to severe amplification effects during scale-up, where reaction times extend even further compared to laboratory trials, creating unpredictable bottlenecks in production schedules. The economic burden is compounded by the relatively low yields, typically hovering around 75%, which results in substantial material loss and increased waste generation that complicates environmental compliance. Additionally, alternative methods utilizing sodium phenolate, as seen in US4394159, introduce high raw material costs and purity challenges due to the difficulty in synthesizing sodium phenolate without generating troublesome impurities that degrade the final product quality.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach outlined in the patent introduces a streamlined one-step synthesis that fundamentally alters the reaction landscape through the strategic application of quaternary ammonium salt catalysts. This innovative method facilitates a rapid etherification reaction between 2,3-dichloro-6-nitroaniline and phenol, achieving completion within a mere 0.5 to 4.0 hours, which represents a drastic reduction in processing time compared to the 20-hour cycles of the past. The introduction of the catalyst not only accelerates the conversion rate but also enhances the selectivity of the reaction, ensuring that the desired product is formed with minimal side reactions and higher overall efficiency. By optimizing the solvent system to include recoverable organic solvents like N,N-dimethylformamide, the process allows for effective vacuum distillation and recycling, thereby minimizing waste and reducing the environmental footprint of the manufacturing operation. This shift towards a more catalytic and efficient process model provides a robust foundation for scaling production while maintaining strict control over product quality and cost structures.
Mechanistic Insights into Quaternary Ammonium Salt-Catalyzed Etherification
The core of this technological advancement lies in the mechanistic role played by the quaternary ammonium salt catalyst, which acts as a phase transfer agent to facilitate the nucleophilic substitution reaction between the aniline derivative and phenol. In the presence of a base such as potassium carbonate, the phenol is deprotonated to form a phenoxide ion, which is then effectively transported into the organic phase by the quaternary ammonium cation, significantly increasing its local concentration and reactivity towards the chlorinated aniline substrate. This phase transfer mechanism bypasses the solubility limitations often encountered in heterogeneous reaction mixtures, allowing the reaction to proceed homogeneously and rapidly at moderate temperatures ranging from 60°C to 100°C. The precise tuning of the catalyst loading, typically between 0.0001 to 0.1 mass ratio relative to the substrate, ensures that the reaction kinetics are optimized without introducing excessive amounts of foreign species that could complicate downstream purification. Understanding this catalytic cycle is crucial for R&D teams aiming to replicate these results, as it highlights the importance of interfacial chemistry in driving high-efficiency transformations in complex organic synthesis.
Beyond merely accelerating the reaction, this catalytic system plays a pivotal role in controlling the impurity profile of the final acifluorfen product, which is a critical parameter for regulatory compliance and end-user performance. The high selectivity of the quaternary ammonium catalyst minimizes the formation of bis-ether byproducts or other structural analogs that often arise from non-selective nucleophilic attacks under harsh conditions. By maintaining a controlled reaction environment with optimized molar ratios of base and phenol, the process ensures that the substitution occurs exclusively at the desired position on the aromatic ring, preserving the integrity of the nitro and amino functional groups. This precision in chemical transformation translates directly to a crude product with significantly higher purity, reducing the burden on subsequent recrystallization steps and minimizing the loss of valuable material during purification. For quality assurance teams, this means a more consistent impurity spectrum that is easier to characterize and control, ultimately leading to a final product that meets stringent international specifications for agrochemical active ingredients.
How to Synthesize Acifluorfen Efficiently
Implementing this advanced synthesis route requires a disciplined approach to process engineering, beginning with the precise charging of reactants into the reaction vessel to ensure optimal mixing and heat transfer. The protocol dictates the sequential addition of 2,3-dichloro-6-nitroaniline, the chosen organic solvent, and phenol, followed by the careful introduction of the base and catalyst under continuous stirring to prevent localized hot spots or concentration gradients. Once the reaction mixture is homogenized, the temperature is raised to the specified range, where the catalytic cycle initiates the rapid conversion of raw materials into the target ether product over a short duration. Following the reaction completion, the process moves to a solvent recovery phase via vacuum distillation, which is critical for both economic and environmental reasons, before the product is precipitated by the addition of water and isolated through filtration. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- Charge the reactor with 2,3-dichloro-6-nitroaniline, organic solvent (DMF/NMP), and phenol under stirring conditions.
- Add base (potassium carbonate) and quaternary ammonium salt catalyst, then heat to 60-100°C for 0.5 to 4 hours.
- Recover solvent via vacuum distillation, cool the mixture, add water to precipitate the crude product, and purify via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method offers compelling economic advantages that extend far beyond simple yield improvements, fundamentally reshaping the cost structure of acifluorfen manufacturing. The drastic reduction in reaction time from days to mere hours translates directly into increased asset utilization, allowing production facilities to turn over batches much faster and respond more agilely to market demand fluctuations without requiring additional capital investment in new reactors. Furthermore, the ability to recover and recycle the organic solvent multiple times significantly lowers the recurring cost of raw materials, which is often one of the largest variable expenses in fine chemical production, thereby enhancing the overall margin profile of the product. The elimination of expensive and difficult-to-handle reagents like sodium phenolate in favor of readily available phenol and catalytic amounts of quaternary salts simplifies the supply chain, reducing dependency on specialized vendors and mitigating the risk of raw material shortages. These factors combine to create a more resilient and cost-effective supply model that can withstand market volatility while delivering consistent value to downstream customers.
- Cost Reduction in Manufacturing: The implementation of this catalytic process drives substantial cost savings by eliminating the need for expensive solvents like dimethyl sulfoxide and reducing the overall consumption of base reagents through improved efficiency. By enabling the recycling of solvents such as DMF and NMP, the process minimizes waste disposal costs and reduces the purchase volume of fresh solvents, leading to a leaner operational expenditure model. Additionally, the higher yield and purity reduce the amount of raw material required per unit of finished product, effectively lowering the cost of goods sold and improving profitability margins for manufacturers. The simplified purification process also reduces energy consumption associated with extensive distillation or chromatography, further contributing to the overall economic benefits of the technology.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method enhances supply chain reliability by shortening the manufacturing lead time, allowing for quicker replenishment of inventory and faster response to urgent customer orders. The use of common and stable raw materials like 2,3-dichloro-6-nitroaniline and phenol ensures a steady supply flow, reducing the risk of disruptions caused by the scarcity of specialized reagents. Moreover, the scalability of the process means that production volumes can be increased seamlessly to meet surging demand without the need for complex process re-engineering or significant downtime. This reliability is crucial for maintaining long-term contracts with major agrochemical companies who prioritize consistent delivery schedules and uninterrupted supply continuity.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers significant advantages by drastically reducing the generation of three wastes, aligning with increasingly stringent global environmental regulations and sustainability goals. The ability to control the reaction precisely minimizes the formation of hazardous byproducts, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing site. The process is designed for easy scale-up, avoiding the amplification effects that often plague batch reactions, which ensures that pilot plant results can be translated to commercial production with high fidelity. This combination of environmental stewardship and scalable engineering makes the technology highly attractive for manufacturers looking to expand capacity while maintaining compliance with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced acifluorfen synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the patent data and practical considerations for industrial application, ensuring that decision-makers have accurate information regarding process capabilities and limitations. Understanding these details is essential for assessing the feasibility of integrating this method into existing production lines and for anticipating the operational changes required to maximize its benefits. We encourage technical teams to review these answers thoroughly to align their expectations with the proven performance metrics of the new catalytic system.
Q: What are the primary advantages of using quaternary ammonium salts in this synthesis?
A: The use of quaternary ammonium salts significantly accelerates the conversion rate of raw materials and improves reaction selectivity, leading to higher yields and purity compared to conventional carbonate-only systems.
Q: How does this process impact solvent recovery and waste generation?
A: The process allows for efficient vacuum distillation and recycling of organic solvents like DMF, drastically reducing the volume of three wastes and lowering overall material costs.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method eliminates complex amplification effects seen in prior art, offering a robust pathway for commercial scale-up with consistent reaction times between 0.5 to 4 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acifluorfen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain competitiveness in the global agrochemical market, and we are fully equipped to support the commercialization of this advanced acifluorfen production route. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle complex catalytic reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications to guarantee product quality that meets or exceeds international standards. We understand the nuances of scaling quaternary ammonium catalyzed reactions and have the engineering expertise to optimize solvent recovery systems and waste treatment protocols for maximum sustainability and cost efficiency.
We invite you to collaborate with us to leverage this technological breakthrough for your supply chain, offering a partnership that goes beyond simple manufacturing to include strategic process optimization and cost management. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new method can improve your bottom line. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to validate the performance of our acifluorfen intermediates against your internal benchmarks. By partnering with us, you gain access to a reliable source of high-quality agrochemical intermediates backed by a commitment to innovation, quality, and long-term supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
