Advanced Manufacturing of Nilotinib Free Base: Overcoming Solvent Limitations for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for kinase inhibitors, particularly for oncology applications where supply chain reliability is paramount. Patent CN114853734A introduces a significant technological advancement in the preparation of Nilotinib free base, a critical intermediate for the tyrosine kinase inhibitor Tasigna. This innovation addresses long-standing challenges in the final coupling step, specifically targeting the limitations imposed by traditional high-boiling polar aprotic solvents. By shifting the reaction medium to a more volatile and manageable solvent system comprising toluene, tetrahydrofuran, dichloromethane, or acetonitrile, the disclosed method achieves superior yield and purity profiles. This technical breakthrough not only enhances the chemical efficiency of the amide bond formation but also streamlines the downstream processing, offering a compelling value proposition for manufacturers aiming to optimize their pharmaceutical intermediates production lines.
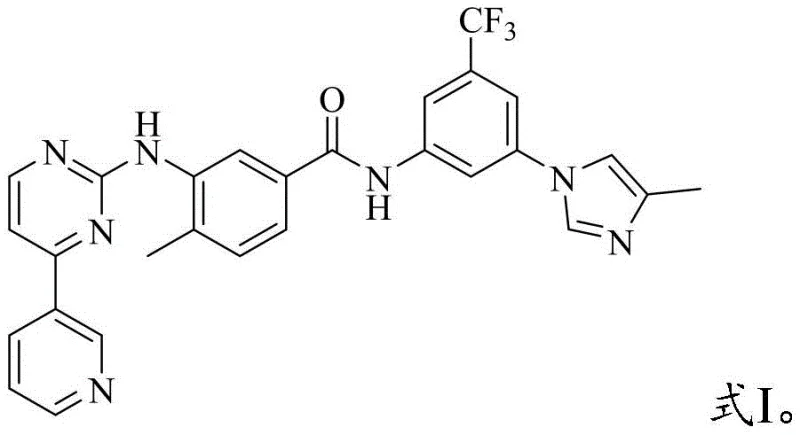
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex amides like Nilotinib has relied heavily on solvents such as N-Methyl-2-pyrrolidone (NMP) to ensure solubility of the bulky reactants. However, reliance on NMP introduces severe operational bottlenecks during the isolation phase. The high boiling point and strong solvating power of NMP often result in the product remaining in solution or forming oils, making filtration exceptionally difficult. Furthermore, reactions conducted in NMP are prone to generating colored impurities that are challenging to remove without extensive and yield-loss-inducing purification steps. These factors collectively drive up the cost of goods sold (COGS) and extend the manufacturing cycle time, creating friction in the supply chain for high-purity API intermediates.
The Novel Approach
The methodology outlined in CN114853734A circumvents these issues by employing a specific first organic solvent system, preferably tetrahydrofuran (THF) or toluene, for the critical coupling reaction between the aniline derivative (Formula II) and the acid chloride (Formula III). This solvent switch fundamentally alters the precipitation behavior of the product. As the reaction proceeds, the Nilotinib free base crystallizes more readily from these solvents compared to NMP, facilitating straightforward filtration. The result is a product with significantly improved color and physical form. This approach exemplifies how strategic solvent selection can resolve cost reduction in pharmaceutical intermediates manufacturing by eliminating the need for complex chromatographic purification or repeated recrystallizations typically required to meet stringent quality specifications.
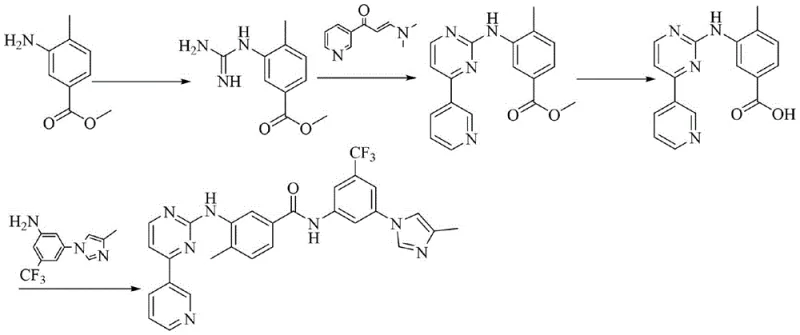
Mechanistic Insights into Optimized Amide Coupling
The core of this synthesis lies in the nucleophilic acyl substitution reaction between the amine group of Formula II and the acid chloride of Formula III. In the optimized protocol, the reaction is catalyzed by 4-Dimethylaminopyridine (DMAP), which acts as a nucleophilic catalyst to accelerate the formation of the acyl-pyridinium intermediate. This activated species is more susceptible to attack by the sterically hindered aniline nitrogen. The choice of THF as the solvent is mechanistically significant; it provides sufficient polarity to dissolve the ionic intermediates and the base (DIPEA or Potassium Carbonate) while maintaining a low enough dielectric constant to allow the neutral product to precipitate upon completion. This balance ensures that the reaction kinetics remain favorable while thermodynamically driving the equilibrium towards product formation through precipitation.
Impurity control is another critical aspect managed by this mechanism. In conventional NMP-based systems, side reactions such as hydrolysis of the acid chloride or over-acylation can be harder to suppress due to the difficulty in controlling local concentration and temperature gradients in the viscous solvent. The lower viscosity of THF allows for better mixing and heat transfer, ensuring that the pH remains within the optimal range of 8 to 9 throughout the reaction. This precise control minimizes the formation of hydrolysis byproducts and urea derivatives, leading to the observed high purity of over 99%. The subsequent workup, involving quenching with saturated sodium bicarbonate and crystallization in an ice-water bath, effectively removes residual catalysts and unreacted starting materials, ensuring a clean impurity profile suitable for downstream salt formation.
How to Synthesize Nilotinib Free Base Efficiently
The synthesis involves a convergent strategy where two key fragments are prepared independently before being joined in the final step. The first fragment, the imidazole-substituted aniline, is generated via a copper-catalyzed coupling, while the second fragment, the trifluoromethyl-benzoyl chloride, is formed via chlorination of the corresponding acid. The final coupling is the rate-determining step where process parameters must be tightly controlled. Detailed standardized operating procedures regarding stoichiometry, addition rates, and temperature ramps are essential for reproducibility. For a comprehensive guide on the exact molar ratios and specific handling of reagents to ensure safety and quality, please refer to the technical protocol below.
- Synthesize the imidazole-substituted aniline intermediate (Formula II) via copper-catalyzed coupling of 3-amino-5-bromotrifluorotoluene and 4-methylimidazole.
- Convert the benzoic acid derivative (Formula IV) into the corresponding acid chloride (Formula III) using thionyl chloride in toluene.
- Perform the final amide coupling between Formula II and Formula III in tetrahydrofuran (THF) with DMAP catalysis, followed by aqueous workup and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the solvent system described in this patent offers tangible logistical and financial benefits beyond simple yield improvements. The elimination of NMP removes a solvent that is increasingly scrutinized under environmental regulations due to its reproductive toxicity classification. Switching to THF or Toluene aligns the manufacturing process with greener chemistry principles, reducing the regulatory burden and waste disposal costs associated with hazardous solvent recovery. This shift simplifies the environmental compliance landscape, allowing for smoother audits and uninterrupted production schedules.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the isolation procedure. By avoiding the filtration difficulties associated with NMP, manufacturers can reduce the labor hours and equipment time required for product recovery. Furthermore, the improved yield means that less raw material is consumed per kilogram of final product, directly lowering the variable cost of production. The ability to achieve high purity without resorting to expensive column chromatography or multiple recrystallization cycles further contributes to substantial cost savings, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: Solvents like THF and Toluene are commodity chemicals with robust global supply chains, unlike specialized high-boiling solvents which may face availability fluctuations. Utilizing common solvents reduces the risk of supply disruption. Additionally, the milder reaction conditions (20-30°C) reduce the energy load on the facility and decrease the wear and tear on reactor jackets and cooling systems. This operational stability ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical partners who require rigorous consistency in their oncology API supply.
- Scalability and Environmental Compliance: The process is inherently scalable due to the exothermic nature of the coupling reaction being easily managed in lower viscosity solvents. Heat removal is more efficient in THF than in NMP, reducing the risk of thermal runaways during scale-up. From an environmental perspective, the lower boiling point of the new solvent system allows for more energy-efficient solvent recovery via distillation. This reduces the carbon footprint of the manufacturing process and lowers the volume of hazardous waste generated, supporting corporate sustainability goals and ensuring compliance with increasingly strict international environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific production needs. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation.
Q: Why is replacing NMP with THF critical in Nilotinib synthesis?
A: Replacing N-Methyl-2-pyrrolidone (NMP) with solvents like THF or Toluene significantly improves post-reaction filtration and product color. NMP often leads to difficult solid-liquid separation and darker impurities, whereas the optimized solvent system facilitates easier crystallization and higher purity.
Q: What is the expected purity profile of this method?
A: The optimized protocol described in patent CN114853734A demonstrates the capability to achieve purity levels exceeding 99%, with specific examples showing 99.394% purity, significantly outperforming comparative methods using traditional solvents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (20-30°C for the coupling step) and commercially available reagents. The simplified workup procedure involving standard aqueous washing and crystallization makes it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nilotinib Free Base Supplier
As the demand for targeted cancer therapies continues to grow, securing a stable supply of high-quality intermediates like Nilotinib free base is essential. NINGBO INNO PHARMCHEM stands ready to support your development and commercialization goals with our state-of-the-art manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in clinical trials or full-scale launch. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical grade materials.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized process can enhance your operational efficiency and reduce your overall cost of goods.
