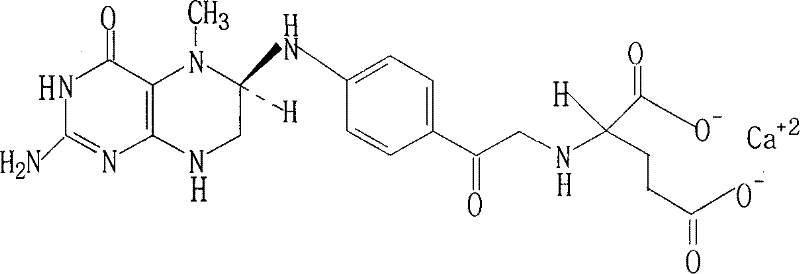
Advanced Resolution and Salification Technology for High-Purity (6S)-5-MTHF Calcium Salt Production
The pharmaceutical and nutritional industries have long recognized the critical biological role of (6S)-5-methyltetrahydrofolate ((6S)-5-MTHF) as the primary active form of folate in human circulation. Unlike synthetic folic acid, this bioactive form bypasses metabolic conversion steps, making it essential for treating conditions ranging from megaloblastic anemia to potential neurodegenerative disorders. However, the commercial production of high-purity (6S)-5-MTHF has historically been hindered by the difficulty of separating the biologically active S-enantiomer from the inactive R-enantiomer. Patent CN101143863B, filed in 2010, introduces a groundbreaking resolution and salification methodology that addresses these longstanding challenges. By utilizing readily available organic bases such as alpha-phenylethylamine for chiral splitting and alkaline earth hydroxides for salt formation, this technology offers a robust pathway to produce (6S)-5-MTHF calcium salt with exceptional optical purity. This report analyzes the technical merits of this patent, providing strategic insights for R&D directors seeking process optimization and procurement leaders aiming for cost-effective supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of (6S)-5-MTHF from its racemic mixture presented significant economic and technical barriers. Early literature suggested that chemical separation of the 6R and 6S enantiomers was nearly impossible, forcing manufacturers to rely on extraction from biological tissues, a method plagued by low yields and exorbitant costs. Subsequent synthetic approaches attempted to use specialized resolving agents like N-ethyl-2-aminomethylpentazane; however, these reagents are not only prohibitively expensive but also difficult to source in large quantities, creating a bottleneck for commercial scale-up. Furthermore, traditional salification processes often employed calcium chloride to generate the stable calcium salt. This approach introduced a critical quality defect: the inevitable retention of chloride ions in the final crystal lattice. For pharmaceutical and food-grade applications, removing these residual chlorides to meet regulatory limits requires additional, costly purification steps, thereby eroding profit margins and complicating the manufacturing workflow.
The Novel Approach
The methodology described in Patent CN101143863B fundamentally reengineers the production landscape by substituting scarce reagents with commodity chemicals. The core innovation lies in the use of alpha-phenylethylamine, a ubiquitous and affordable chiral amine, as the resolving agent. This switch drastically lowers the raw material cost basis while maintaining high stereoselectivity. Additionally, the patent replaces calcium chloride with calcium hydroxide or oxide for the salification step. This modification is chemically elegant; it ensures that the counter-ion introduced is hydroxide, which reacts cleanly without leaving behind halogenated impurities. The process further optimizes crystallization by employing 95% ethanol as an anti-solvent, facilitating the precipitation of the calcium salt from aqueous solutions where it is otherwise difficult to crystallize. This holistic approach transforms a complex, multi-step purification challenge into a streamlined, economically viable operation suitable for industrial manufacturing.
Mechanistic Insights into Alpha-Phenylethylamine Mediated Resolution
The success of this synthesis hinges on the precise thermodynamic control of diastereomeric salt formation. When racemic (6R,S)-5-MTHF is introduced to an aqueous solution containing alpha-phenylethylamine, the chiral amine interacts differently with the two enantiomers of the folate derivative. Due to the spatial arrangement of the pteroyl and glutamyl moieties relative to the chiral center at the 6-position, the (6S)-enantiomer forms a less soluble diastereomeric salt complex with the amine compared to the (6R)-complex. By carefully controlling the temperature profile—heating the mixture to between 20°C and 80°C to ensure complete dissolution and interaction, followed by slow cooling to approximately 20°C—the system leverages solubility differences to selectively precipitate the desired 6S-salt. The insoluble 6R-salt remains in the mother liquor or is filtered off earlier depending on the specific isomer of the amine used, allowing for the physical separation of the enantiomers through simple filtration.
Following the resolution, the conversion to the calcium salt involves a delicate acid-base neutralization and metathesis reaction. The isolated 6S-amine salt is first converted to the sodium salt using sodium hydroxide, rendering the folate moiety soluble and free from the resolving agent. Subsequently, calcium hydroxide is introduced under controlled thermal conditions (not exceeding 40°C) to prevent thermal degradation of the sensitive tetrahydropteridine ring. The calcium ions coordinate with the carboxylate groups of the glutamic acid side chain and the pteroic acid moiety. The addition of 95% ethanol reduces the dielectric constant of the solvent medium, dramatically decreasing the solubility of the resulting calcium salt and driving the equilibrium toward precipitation. This mechanism ensures that the final product is not only chemically pure but also possesses the correct hydration state, typically forming a pentahydrate structure that is stable for storage and formulation.
How to Synthesize (6S)-5-Methyltetrahydrofolate Calcium Salt Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and solvent ratios as critical process parameters. The procedure begins with the preparation of an aqueous suspension of the racemic starting material, followed by the stoichiometric addition of the resolving agent. Operators must monitor the dissolution closely, ensuring that the reaction mixture reaches the specified temperature range to maximize the formation of the diastereomeric complex. Once the resolution is complete and the crystals are harvested, the subsequent salification steps require precise pH monitoring to ensure complete conversion to the calcium species without degrading the folate core. For a detailed breakdown of the specific reagent quantities, stirring rates, and drying conditions required to replicate the high yields reported in the patent examples, please refer to the standardized synthesis guide below.
- Dissolve racemic (6R,S)-5-MTHF in water and add the resolving agent alpha-phenylethylamine, heating to 20-80°C to form the diastereomeric salt.
- Cool the solution to approximately 20°C to induce crystallization of the desired 6S-isomer salt, then filter and separate the crystals from the mother liquor containing the 6R-isomer.
- Suspend the isolated 6S-salt in water, adjust pH with sodium hydroxide, add calcium hydroxide, and precipitate the final product using 95% ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN101143863B represents a significant opportunity to optimize the cost structure of folate-based product lines. The shift from proprietary, high-cost resolving agents to commodity-grade alpha-phenylethylamine directly impacts the bill of materials, reducing the dependency on single-source suppliers for niche chemicals. Furthermore, the elimination of chloride-based salification reagents simplifies the downstream processing requirements. By avoiding the introduction of chloride ions, manufacturers can bypass expensive ion-exchange chromatography or extensive recrystallization cycles typically needed to meet pharmacopeial limits for halogens. This streamlining of the purification train not only reduces utility consumption and waste generation but also shortens the overall batch cycle time, enhancing the responsiveness of the supply chain to market demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the substitution of expensive chiral auxiliaries with alpha-phenylethylamine, a bulk chemical available from multiple global vendors. This diversification of the supply base mitigates price volatility and ensures consistent availability. Additionally, the use of calcium hydroxide instead of calcium chloride eliminates the need for costly chloride removal steps, significantly lowering the operational expenditure associated with purification. The high recovery yield of the calcium salt precipitation step, driven by the ethanol anti-solvent technique, maximizes the throughput of the active pharmaceutical ingredient per batch, further diluting fixed overhead costs across a larger volume of saleable product.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates like (6S)-5-MTHF is paramount for continuous pharmaceutical production. This process enhances reliability by utilizing reagents that are staples of the fine chemical industry, reducing the risk of supply disruptions caused by the scarcity of specialized resolving agents. The robustness of the crystallization process, which tolerates minor variations in temperature and concentration while still delivering high optical purity, ensures consistent batch-to-batch quality. This consistency reduces the rate of batch failures and reworks, stabilizing the production schedule and ensuring that delivery commitments to downstream formulators are met without delay.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process offers distinct advantages. The aqueous-ethanol solvent system is relatively benign compared to processes requiring chlorinated solvents or heavy metal catalysts, simplifying waste treatment and disposal protocols. The absence of heavy metals and the reduction of halogenated waste streams align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. Moreover, the unit operations involved—heating, cooling, filtration, and precipitation—are standard in chemical engineering and scale up linearly from kilogram to tonne scales without requiring specialized equipment, facilitating rapid capacity expansion to meet growing global demand for active folate supplements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (6S)-5-MTHF calcium salt using the resolution method detailed in Patent CN101143863B. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's applicability to your specific manufacturing needs. Understanding these nuances is crucial for technical teams assessing process transfer feasibility and quality assurance protocols.
Q: Why is alpha-phenylethylamine preferred over other resolving agents for 5-MTHF?
A: Alpha-phenylethylamine is selected because it is a commercially available, cost-effective organic base compared to expensive alternatives like N-ethyl-2-aminomethylpentazane. It effectively forms diastereomeric salts with high optical purity (>=98.5%) without introducing toxic impurities.
Q: How does this process avoid chloride contamination in the final calcium salt?
A: Unlike traditional methods that use calcium chloride for salification, this patent utilizes calcium hydroxide or oxide. This eliminates the risk of residual chloride ions, ensuring the final product meets stringent pharmaceutical limits for halogen impurities.
Q: What is the expected optical purity of the resolved (6S)-5-MTHF?
A: According to the experimental data in Patent CN101143863B, the process consistently achieves an optical purity of greater than 98.5%, with specific examples demonstrating purity levels reaching 99.0%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (6S)-5-Methyltetrahydrofolate Calcium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality oversight. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the alpha-phenylethylamine resolution process are fully realized in a GMP-compliant environment. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying optical purity levels exceeding 99%, guaranteeing that every batch of (6S)-5-MTHF calcium salt meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite forward-thinking organizations to collaborate with us to leverage this advanced technology for their product portfolios. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. We encourage you to contact us today to request specific COA data from our recent pilot batches and to discuss route feasibility assessments that can accelerate your time-to-market for next-generation folate-based therapies.
