Advanced Photochlorination Technology for High-Purity o-Chloromethyl Benzoyl Chloride Production
Advanced Photochlorination Technology for High-Purity o-Chloromethyl Benzoyl Chloride Production
The global demand for high-efficiency herbicides, particularly bensulfuron-methyl, has necessitated a rigorous re-evaluation of key intermediate supply chains, specifically focusing on the synthesis of o-chloromethyl benzoyl chloride. Patent CN110818557A introduces a transformative manufacturing methodology that replaces traditional thermal catalysis with a continuous photochlorination process, fundamentally altering the safety and efficiency profile of this critical agrochemical building block. This technological leap addresses the longstanding industry challenges associated with hazardous catalyst residues and low selectivity in benzylic chlorination reactions. By leveraging ultraviolet irradiation within a circulating reactor system, the process achieves exceptional control over reaction kinetics, ensuring that the primary chlorination event dominates while suppressing undesirable over-chlorination pathways. For R&D directors and procurement strategists, this patent represents a viable pathway to secure a more stable, high-purity supply of intermediates essential for next-generation crop protection agents. The integration of reaction and separation units further exemplifies modern process intensification strategies aimed at maximizing atom economy and minimizing waste generation in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of o-chloromethyl benzoyl chloride has relied heavily on thermal initiation using catalysts such as azodiisobutyronitrile (AIBN) or organic peroxides, a approach fraught with significant safety and quality control liabilities. These thermal initiators are inherently unstable compounds that can undergo violent decomposition at elevated temperatures, creating substantial explosion and ignition hazards within large-scale reactor vessels. Furthermore, the decomposition products of these catalysts often remain as impurities within the crude reaction mixture, complicating downstream purification efforts and potentially interfering with subsequent coupling reactions in herbicide synthesis. The conventional batch process typically struggles to halt the reaction at the monochloro stage, leading to the formation of o-dichloromethyl benzoyl chloride as a persistent by-product that reduces overall yield to approximately 78 percent. Additionally, the presence of catalyst residues and by-products makes effective vacuum rectification difficult, often resulting in a final product content of only 75 percent, which is suboptimal for high-value pharmaceutical or agrochemical applications requiring stringent impurity profiles.
The Novel Approach
The innovative method disclosed in the patent data circumvents these issues by employing ultraviolet light with a wavelength range of 315 nm to 400 nm as the sole initiator for the free radical chain reaction. This photochemical activation eliminates the need for any chemical catalyst, thereby removing the risk of thermal runaway explosions and ensuring that no foreign organic residues contaminate the final product stream. The process operates continuously within a specialized circulating reactor where o-methylbenzoyl chloride and chlorine gas are intimately mixed under controlled temperature conditions of 60-80 ℃, allowing for precise management of the reaction progression. By coupling the chlorination reactor directly with a gas-removing tower and a vacuum rectification column, the system enables the immediate separation of hydrogen chloride and unreacted chlorine, driving the equilibrium forward while protecting the product from degradation. Most critically, the unreacted raw material recovered from the top of the rectification column is recycled back into the chlorination reactor, creating a closed-loop system that maximizes raw material utilization and pushes the overall yield to an impressive 95 percent with a purity content reaching 97 percent.
Mechanistic Insights into UV-Initiated Free Radical Chlorination
The core chemical transformation relies on the homolytic cleavage of the chlorine molecule (Cl2) induced by photon absorption, generating highly reactive chlorine radicals (Cl•) that propagate the chain reaction without the need for thermal energy input. Upon exposure to UV light, the chlorine gas dissociates into two chlorine radicals, which then abstract a hydrogen atom from the benzylic methyl group of the o-methylbenzoyl chloride substrate. This abstraction generates a resonance-stabilized benzylic radical intermediate, which subsequently reacts with another chlorine molecule to form the desired o-chloromethyl benzoyl chloride product while regenerating a chlorine radical to sustain the chain. The absence of thermal initiators means the reaction rate is governed primarily by light intensity and chlorine concentration rather than temperature-dependent decomposition kinetics, offering a much flatter and more controllable reaction profile. This mechanistic clarity allows operators to fine-tune the photon flux to match the feed rate of reactants, ensuring that the concentration of chlorine radicals remains optimal for monochlorination without promoting excessive radical density that could lead to non-selective attack.
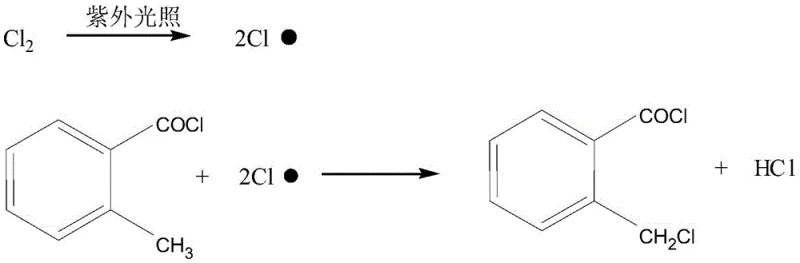
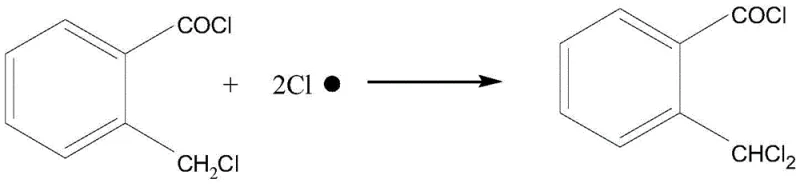
A critical aspect of this mechanism is the suppression of the secondary chlorination reaction, where the newly formed benzylic chloride could theoretically undergo a second hydrogen abstraction to form the dichloro by-product. The patent data highlights that the selectivity is maintained by strictly controlling the residence time and the molar ratio of reactants within the circulating loop, ensuring that the concentration of the monochloro product does not build up to levels where it becomes the dominant substrate for further chlorination. By maintaining the reaction mixture composition at roughly 50-55 percent monochloro product and 45-50 percent starting material, the probability of a chlorine radical encountering a monochloro molecule is statistically minimized relative to encountering the more abundant starting material. This kinetic control is visually represented by the competing reaction pathways, where the goal is to maximize the flux through the primary pathway while effectively shutting down the secondary pathway through process engineering rather than chemical inhibition. The result is a crude mixture containing less than 0.1 percent of the dichloro impurity, drastically simplifying the burden on the downstream distillation columns.
How to Synthesize o-Chloromethyl Benzoyl Chloride Efficiently
Implementing this synthesis route requires a specialized setup capable of handling corrosive chlorine gas and integrating photochemical reactors with high-vacuum distillation units. The process begins with the continuous pumping of o-methylbenzoyl chloride into a chlorination circulating reactor equipped with high-intensity UV lamps, where the temperature is meticulously maintained between 60 ℃ and 80 ℃ to balance reaction rate and selectivity. Chlorine gas is introduced at a controlled flow rate to maintain a specific molar excess, ensuring that the light-driven radical generation is the rate-limiting step rather than chlorine availability. As the reaction proceeds, the effluent is continuously monitored for component ratios, and once the target conversion index is reached, the stream is diverted to a gas-stripping tower to remove dissolved gases before entering the fractionation column. For a detailed breakdown of the operational parameters, equipment specifications, and safety protocols required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Continuously pump o-methylbenzoyl chloride and chlorine gas into a circulating reactor equipped with a UV light source (315-400 nm) at 60-80°C.
- Monitor the reaction mixture composition to maintain 50-55% monochloro product and 45-50% raw material, minimizing dichloro by-products.
- Transfer the mixture to a gas-removing tower to eliminate HCl and excess chlorine, followed by vacuum rectification to separate the product and recycle unreacted raw material.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this photochlorination technology offers profound advantages in terms of supply chain resilience and total cost of ownership for agrochemical manufacturers. The elimination of hazardous thermal catalysts not only reduces the cost of raw materials but also significantly lowers the expenses associated with waste disposal and environmental compliance, as there are no toxic catalyst residues to treat. The continuous nature of the process, combined with the internal recycling of unreacted starting materials, ensures a highly efficient use of feedstock, which translates to substantial cost savings in raw material procurement over long production runs. Furthermore, the improved safety profile of the UV-based method reduces insurance premiums and operational downtime risks associated with handling explosive initiators, providing a more reliable and consistent supply stream for downstream herbicide production facilities. The ability to achieve high purity directly from the reactor reduces the load on purification units, leading to lower energy consumption and faster batch cycle times, which collectively enhance the overall throughput capacity of the manufacturing plant.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous catalysts like AIBN eliminates a significant line item from the bill of materials while simultaneously reducing the complexity of the workup procedure. Without catalyst residues to remove, the distillation process becomes more energy-efficient and requires less maintenance, leading to lower utility costs and extended equipment lifespan. The high yield of 95 percent means that less raw material is wasted as by-products or lost during purification, directly improving the mass balance and reducing the cost per kilogram of the final active intermediate. Additionally, the recycling loop ensures that virtually all purchased o-methylbenzoyl chloride is eventually converted to product, maximizing the return on investment for every ton of feedstock acquired by the procurement team.
- Enhanced Supply Chain Reliability: The continuous flow design of this process allows for scalable production that can be easily ramped up to meet fluctuating market demands for bensulfuron-methyl precursors without the bottlenecks typical of batch processing. Since the process does not rely on catalysts that have limited shelf lives or require cold storage, the supply chain is simplified, and the risk of production stoppages due to reagent degradation is effectively nullified. The robustness of the UV initiation system ensures consistent product quality across different batches, reducing the likelihood of rejected shipments and fostering stronger trust between suppliers and multinational agrochemical clients. This reliability is crucial for maintaining just-in-time inventory levels and ensuring that downstream formulation plants never face shortages of this critical intermediate.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, as the only major by-product is hydrogen chloride gas, which can be easily captured and sold as hydrochloric acid or neutralized safely. The absence of heavy metal or organic peroxide contaminants simplifies the wastewater treatment process, helping manufacturers meet increasingly stringent environmental regulations without costly upgrades to effluent treatment plants. The modular nature of the photochlorination reactors allows for linear scale-up, meaning that capacity can be increased by adding parallel reactor units rather than building massive single vessels, reducing capital expenditure risks. This environmental and operational flexibility positions manufacturers using this technology as preferred partners for global companies seeking sustainable and compliant supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature to provide accurate guidance for technical evaluators. Understanding these details is essential for assessing the feasibility of integrating this technology into existing production lines or for qualifying new suppliers who utilize this advanced method.
Q: Why is UV light preferred over AIBN for this chlorination?
A: UV light eliminates the need for hazardous thermal initiators like AIBN or peroxides, which pose explosion risks at high temperatures and leave residues that complicate purification. The photochemical method ensures a cleaner product profile suitable for sensitive agrochemical applications.
Q: How does the process control over-chlorination to the dichloro by-product?
A: The process utilizes a continuous circulation system where the conversion rate is strictly controlled. By maintaining the reaction mixture at approximately 50-55% monochloro product and recycling unreacted raw material, the residence time is optimized to prevent the secondary reaction that forms o-dichloromethyl benzoyl chloride.
Q: What are the purity and yield specifications of this new method?
A: According to patent CN110818557A, this optimized photochlorination route achieves a product yield of 95% and a purity content of 97%, significantly outperforming traditional thermal methods which typically yield around 78% with 75% content.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Chloromethyl Benzoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of agrochemical and pharmaceutical development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of o-chloromethyl benzoyl chloride meets the exacting standards required for herbicide synthesis. Our commitment to process safety and environmental stewardship aligns perfectly with the advanced photochlorination techniques described in recent patents, allowing us to offer a product that is both economically competitive and environmentally responsible.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique supply chain needs.
