Scalable Enzymatic Production of Paroxetine Intermediate Using Novel Marine Esterase PE8
Scalable Enzymatic Production of Paroxetine Intermediate Using Novel Marine Esterase PE8
The pharmaceutical industry constantly seeks robust and cost-effective pathways for synthesizing chiral intermediates, particularly for high-volume antidepressants like paroxetine. Patent CN103215238A introduces a groundbreaking biocatalytic solution utilizing a novel marine esterase, designated as PE8, derived from the marine bacterium Pelagibacterium halotolerans. This technology addresses critical bottlenecks in the production of (R)-3-(4-fluorophenyl) monomethyl glutarate, a key precursor for levo-paroxetine hydrochloride. By leveraging the unique genetic traits of marine microorganisms, this invention offers a sustainable alternative to traditional chemical synthesis and expensive commercial enzyme preparations. The patent details the cloning, expression, and application of this enzyme, highlighting its superior salt and alkali resistance which are vital for large-scale industrial fermentation and reaction processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure (R)-3-(4-fluorophenyl) glutaric acid monomethyl ester has faced significant hurdles regarding stereo-selectivity and cost efficiency. Early attempts using pig liver esterase resulted in the formation of the (S)-enantiomer, which is the incorrect configuration for synthesizing levo-paroxetine, rendering the process commercially unviable despite high yields. Furthermore, while immobilized lipases such as Novozym 435 from Candida antarctica can achieve the desired (R)-selectivity, their prohibitive cost makes them unsuitable for large-scale industrial manufacturing. Chemical methods often involve harsh conditions, heavy metal catalysts, and complex resolution steps that generate substantial waste and increase the environmental footprint. These legacy limitations create a pressing need for a biocatalyst that combines high stereoselectivity with economic feasibility and operational stability.
The Novel Approach
The introduction of esterase PE8 represents a paradigm shift in the biocatalytic production of this critical pharmaceutical intermediate. Isolated from a marine environment, this enzyme possesses inherent stability traits, such as salt tolerance and alkali resistance, which are rarely found in terrestrial counterparts. The patent demonstrates that PE8 can be highly and solubly expressed in E. coli Rosetta strains, achieving expression levels up to 23.1% of total protein, which drastically lowers the cost of the biocatalyst compared to purchasing commercial immobilized enzymes. By optimizing reaction parameters such as organic co-solvent concentration and pH, the process achieves high conversion rates and improved chiral selectivity. This approach not only ensures the production of the correct (R)-isomer but also simplifies the downstream processing due to the enzyme's robustness in varied buffer conditions.
Mechanistic Insights into Esterase PE8-Catalyzed Hydrolysis
The core of this technology lies in the specific hydrolytic mechanism of esterase PE8, which belongs to the family VI lipohydrolases. The enzyme selectively hydrolyzes one of the ester groups in 3-(4-fluorophenyl) dimethyl glutarate (3-DFG) to yield the mono-ester product. Phylogenetic analysis indicates low homology with existing esterases, suggesting a unique active site architecture that favors the (R)-configuration. The reaction proceeds efficiently in a biphasic or co-solvent system, where the enzyme maintains its structural integrity and catalytic turnover even in the presence of organic solvents like 1,4-dioxane. This solvent tolerance is crucial for dissolving the hydrophobic substrate while maintaining the aqueous environment required for enzyme activity. The mechanistic pathway avoids the formation of the di-acid byproduct until later stages, allowing for precise control over the reaction endpoint to maximize mono-ester yield.
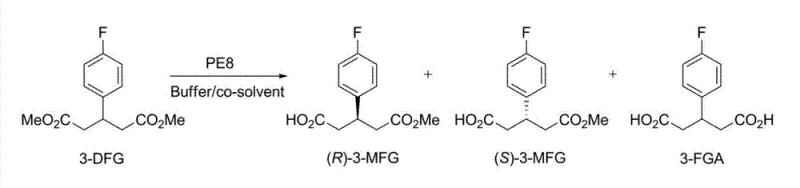
Impurity control is another critical aspect of the PE8 catalytic system, driven by the enzyme's specific substrate recognition. The patent data reveals that PE8 has strong activity towards short-chain esters but negligible activity towards long-chain esters, which minimizes side reactions with potential fatty acid impurities in the feedstock. Furthermore, the enzyme's stability at high pH values (up to pH 11) and high ionic strength (1M phosphate) allows manufacturers to operate under conditions that suppress microbial contamination and stabilize the product against non-enzymatic hydrolysis. The inhibition profile shows sensitivity to specific divalent cations like Zn2+ and Cu2+, indicating that water quality and buffer composition must be managed to prevent activity loss. However, its resilience against other ions ensures flexibility in buffer selection for scale-up. This combination of selectivity and stability creates a clean reaction profile, reducing the burden on purification units and ensuring a high-purity final product suitable for pharmaceutical applications.
How to Synthesize (R)-3-(4-fluorophenyl) monomethyl glutarate Efficiently
Implementing this enzymatic route requires a structured approach to biocatalyst preparation and reaction engineering to fully realize the benefits outlined in the patent. The process begins with the recombinant production of the enzyme, followed by the optimization of the hydrolysis reaction parameters to balance conversion and enantiomeric excess. Detailed standard operating procedures for strain construction, fermentation, and the specific hydrolysis protocol are essential for technology transfer. For a comprehensive guide on the exact step-by-step synthesis including reagent quantities and specific workup procedures, please refer to the standardized protocol section below.
- Clone the esterase gene pe8 from Pelagibacterium halotolerans into an expression vector like pET28b(+) and transform into E. coli Rosetta.
- Induce soluble expression of the recombinant esterase PE8 using IPTG at 25°C overnight to obtain high-activity crude enzyme powder.
- Perform selective hydrolysis of 3-(4-fluorophenyl) dimethyl glutarate in a buffer system containing 1,4-dioxane at pH 8.0 and 30°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of esterase PE8 offers tangible strategic advantages over traditional sourcing models. The primary benefit stems from the shift from purchasing expensive commercial immobilized enzymes to producing the biocatalyst in-house via fermentation. This vertical integration of the catalyst supply chain significantly reduces raw material costs and mitigates the risk of price volatility associated with proprietary commercial enzymes. Additionally, the enzyme's ability to function as a crude powder eliminates the need for costly purification steps during enzyme production, further driving down the cost of goods sold. The robust nature of the enzyme also translates to more reliable manufacturing schedules, as the process is less susceptible to batch failures caused by enzyme instability.
- Cost Reduction in Manufacturing: The elimination of expensive commercial lipases like Novozym 435 represents a major cost saving opportunity for large-scale production. By utilizing a recombinant system that yields high levels of soluble protein, manufacturers can produce the necessary biocatalyst at a fraction of the market price of imported enzymes. Furthermore, the ability to use crude enzyme powder directly in the reaction removes downstream purification costs associated with enzyme isolation. The process operates under mild temperatures around 30°C, which reduces energy consumption for heating and cooling compared to harsher chemical alternatives. These cumulative factors contribute to a substantially lower overall production cost for the paroxetine intermediate.
- Enhanced Supply Chain Reliability: Relying on a single source for specialized commercial enzymes creates a single point of failure in the supply chain. By cloning the gene and establishing an internal fermentation process, companies gain full control over the supply of the biocatalyst. The high expression levels in E. coli ensure that sufficient quantities of enzyme can be produced on demand to support continuous manufacturing campaigns. Moreover, the enzyme's stability under high salt and alkaline conditions means it has a longer shelf life and is more forgiving during transport and storage. This reliability ensures consistent availability of the key intermediate, preventing delays in the synthesis of the final active pharmaceutical ingredient.
- Scalability and Environmental Compliance: The enzymatic process aligns perfectly with green chemistry principles, which is increasingly important for regulatory compliance and corporate sustainability goals. The reaction avoids the use of heavy metal catalysts and toxic solvents often required in chemical resolution methods, simplifying waste treatment and disposal. The high selectivity of the enzyme reduces the formation of unwanted isomers and byproducts, leading to higher atom economy and less waste generation. Scalability is facilitated by the enzyme's tolerance to high substrate concentrations and its stability in the presence of organic co-solvents, allowing for efficient operation in large reactors. This makes the transition from pilot scale to commercial tonnage production smoother and more environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this marine esterase technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these nuances is critical for evaluating the feasibility of integrating this biocatalytic route into existing manufacturing frameworks.
Q: What are the stability characteristics of Esterase PE8?
A: Esterase PE8 exhibits exceptional stability under industrial conditions, maintaining high activity and chiral selectivity even at pH 11 and in 1M phosphate buffer, which facilitates robust process control.
Q: How does PE8 compare to commercial lipases like Novozym 435?
A: Unlike expensive immobilized commercial lipases, PE8 can be highly soluble expressed in E. coli, significantly reducing biocatalyst costs while providing the specific (R)-selectivity required for paroxetine synthesis.
Q: What is the substrate specificity of this marine esterase?
A: PE8 belongs to family VI lipohydrolases and shows strong catalytic activity towards short-chain esters, making it ideal for hydrolyzing dimethyl glutarate derivatives without affecting longer chain impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-(4-fluorophenyl) monomethyl glutarate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the PE8 esterase technology in streamlining the supply chain for antidepressant intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for global pharmaceutical markets. We are committed to leveraging advanced biocatalytic solutions like the one described in CN103215238A to deliver high-quality intermediates with consistent reliability.
We invite you to collaborate with us to optimize your supply chain for paroxetine precursors. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, scalable, and cost-efficient source of critical chiral building blocks, enabling you to focus on your core drug development goals while we manage the complexities of intermediate manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
