Advanced Catalytic Synthesis of Terephthaloyl Chloride for High-Performance Polymer and Pharma Applications
Advanced Catalytic Synthesis of Terephthaloyl Chloride for High-Performance Polymer and Pharma Applications
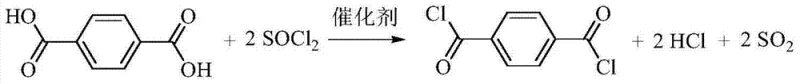
The production of high-purity terephthaloyl chloride represents a critical bottleneck in the supply chain for advanced polyamides, polyesters, and pharmaceutical intermediates. Traditional methods often struggle with impurity profiles that compromise the mechanical properties of downstream polymers or the safety profiles of drug substances. Patent CN103694113A introduces a transformative approach to this synthesis, utilizing specific nitrogenous organic bases to catalyze the reaction between terephthalic acid and thionyl chloride. This method addresses the longstanding challenges of catalyst stability and product separation, offering a pathway to yields reaching 96% and purity levels exceeding 99%. For R&D directors and procurement managers, this technology signifies a shift towards more robust, scalable, and economically viable manufacturing processes that eliminate the need for complex purification steps traditionally required to remove catalyst residues and by-products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chlorination of terephthalic acid has relied heavily on reagents like DMF or pyridine, which present significant operational and chemical drawbacks in an industrial setting. The use of DMF, for instance, often leads to the formation of Vilsmeier reagents upon reaction with thionyl chloride, creating unstable intermediates that complicate the isolation of the final acyl chloride product. This instability necessitates rigorous and repeated recrystallization or multiple vacuum distillation cycles to achieve the requisite purity of 99%, drastically increasing energy consumption and processing time. Furthermore, conventional phase transfer catalysts, such as quaternary ammonium salts, while effective in improving yield to around 90%, are often costly to synthesize and difficult to recover, leading to higher operational expenditures and environmental waste burdens that modern supply chains can no longer sustain efficiently.
The Novel Approach
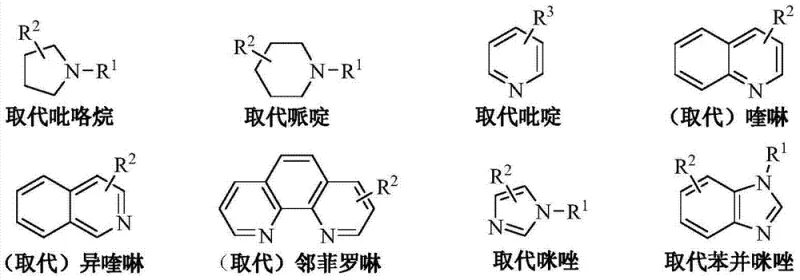
The innovative methodology described in the patent data circumvents these issues by employing a class of substituted nitrogenous organic bases that exhibit superior stability and selectivity under chlorination conditions. By utilizing catalysts such as substituted pyrrolidines, piperidines, or quinolines, the reaction system avoids the formation of difficult-to-remove side products, thereby streamlining the downstream purification process. The process operates effectively at moderate temperatures between 70°C and 120°C, reducing thermal stress on the equipment and minimizing the risk of thermal degradation of the sensitive acyl chloride functionality. This approach not only simplifies the operational workflow but also enhances the overall mass balance of the reaction, allowing for the direct recovery of excess thionyl chloride and the catalyst itself, which can be recycled for subsequent batches, creating a closed-loop system that aligns with green chemistry principles.
Mechanistic Insights into Nitrogenous Organic Base Catalysis
The efficacy of this synthesis relies on the dual role played by the nitrogenous organic base in facilitating both proton abstraction and phase transfer within the reaction medium. Terephthalic acid is notoriously difficult to dissolve in thionyl chloride, creating a heterogeneous solid-liquid system that limits reaction kinetics. The added base acts as a proton acceptor, removing hydrogen ions from the carboxylic acid groups to form organonitrogen cations. These cations then form ion pairs with the carboxylate anions, significantly enhancing the solubility of the terephthalic acid derivative in the polar thionyl chloride solvent. This solubilization effect transforms the reaction from a slow surface-limited process into a rapid homogeneous or pseudo-homogeneous reaction, accelerating the conversion rate and ensuring complete utilization of the starting material without the need for excessive thermal input.
Furthermore, the electronic properties of the catalyst substituents play a pivotal role in determining the reaction outcome, as evidenced by the comparative data in the patent. Electron-donating groups on the pyridine ring, such as methyl or ethyl groups, enhance the basicity of the nitrogen center, thereby improving its ability to abstract protons and catalyze the formation of the acyl chloride. Conversely, strong electron-withdrawing groups can diminish catalytic activity, while specific configurations like 3-cyanopyridine demonstrate that electronic effects are nuanced and dependent on the specific resonance structures involved. Understanding these mechanistic subtleties allows process chemists to fine-tune the catalyst selection to match specific batch requirements, ensuring consistent quality and minimizing the formation of mono-acyl chloride impurities that can act as chain terminators in subsequent polymerization steps.
How to Synthesize Terephthaloyl Chloride Efficiently
Implementing this catalytic route requires precise control over stoichiometry and thermal parameters to maximize the benefits of the nitrogenous base system. The process begins with the careful mixing of terephthalic acid and thionyl chloride, followed by the introduction of the catalyst at a loading of 0.5% to 2% by mass. The reaction mixture is then heated to the optimal range of 70°C to 120°C, where it is maintained for a duration of 4 to 16 hours depending on the specific catalyst activity and desired conversion level. Detailed standard operating procedures regarding the specific distillation pressures and temperatures required for the simultaneous recovery of the solvent and catalyst are critical for maintaining the economic viability of the process and are outlined in the technical guide below.
- Mix terephthalic acid and thionyl chloride at a mass ratio of 2: 1 to 5:1 in a reaction vessel equipped with stirring and temperature control.
- Add 0.5% to 2% by mass of a nitrogenous organic base catalyst such as substituted pyridine or quinoline relative to the terephthalic acid.
- Heat the mixture to 70-120°C for 4-16 hours, then recover excess thionyl chloride and catalyst via distillation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers substantial advantages in terms of cost structure and supply chain resilience for buyers of fine chemical intermediates. The ability to recover and reuse both the thionyl chloride solvent and the nitrogenous organic base catalyst drastically reduces the consumption of raw materials per kilogram of product produced. This circular usage model mitigates the impact of volatile raw material pricing and reduces the volume of hazardous waste requiring disposal, leading to significant operational cost savings that can be passed down through the supply chain. Additionally, the simplified purification process eliminates the need for multiple recrystallization steps, shortening the overall production cycle time and increasing the throughput capacity of existing manufacturing assets without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove catalysts like DMF reduces the complexity of the workup procedure, directly lowering labor and utility costs associated with prolonged distillation and purification cycles. By avoiding the formation of Vilsmeier reagents, the process minimizes product loss during purification, thereby improving the effective yield and reducing the cost per unit of high-purity terephthaloyl chloride. The recyclability of the catalyst further ensures that the variable cost of production remains stable and predictable, shielding the procurement budget from fluctuations in specialty chemical pricing.
- Enhanced Supply Chain Reliability: The robustness of the nitrogenous base catalysts against hydrolysis and thermal degradation ensures consistent batch-to-batch quality, reducing the risk of production delays caused by off-spec material. The use of readily available and stable catalysts means that supply disruptions are less likely compared to methods relying on specialized phase transfer agents that may have limited supplier bases. This reliability is crucial for maintaining continuous production schedules for downstream polymer manufacturers who depend on just-in-time delivery of high-quality monomers to meet their own customer commitments.
- Scalability and Environmental Compliance: The process generates gaseous by-products of HCl and SO2 which can be efficiently scrubbed using standard water and caustic absorption systems, ensuring compliance with strict environmental regulations regarding acidic emissions. The moderate reaction temperatures and the ability to recover solvents under vacuum make the process highly scalable from pilot plant to multi-ton commercial production without encountering the heat transfer limitations often seen in highly exothermic chlorination reactions. This scalability ensures that suppliers can rapidly ramp up production to meet surges in demand for high-performance materials used in aerospace and automotive applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic synthesis route for terephthaloyl chloride. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring that the information is grounded in verified chemical principles rather than marketing speculation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing workflows or for procurement professionals assessing the long-term viability of this supply source.
Q: What are the advantages of using nitrogenous organic bases over DMF in this synthesis?
A: Unlike DMF, which can form unstable Vilsmeier reagents complicating purification, nitrogenous organic bases remain stable against thionyl chloride, allowing for simpler workup and higher product purity exceeding 99% without repeated recrystallization.
Q: Can the catalyst be reused in the terephthaloyl chloride production process?
A: Yes, the patent data confirms that the nitrogenous organic base catalyst has a lower boiling point than the product and can be recovered via vacuum distillation along with excess thionyl chloride for circular reuse, significantly reducing raw material costs.
Q: What is the expected yield and purity for this catalytic method?
A: Experimental data from the patent indicates that optimized conditions using specific substituted pyridines or quinolines can achieve a separation yield of up to 96% with a final product purity greater than 99% as verified by GC-MS and NMR.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terephthaloyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a synthesis route is only as valuable as its ability to be executed reliably at scale. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this catalytic method are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of terephthaloyl chloride meets the exacting standards required for high-performance polymer synthesis and pharmaceutical applications, providing our partners with the confidence to innovate without supply chain constraints.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can drive efficiency in your specific operations. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that align with your strategic goals for cost reduction and quality assurance in the competitive fine chemicals market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
